Team:Calgary/Project/OSCAR/CatecholDegradation
From 2012.igem.org


Hello! iGEM Calgary's wiki functions best with Javascript enabled, especially for mobile devices. We recommend that you enable Javascript on your device for the best wiki-viewing experience. Thanks!
Catechol Degradation
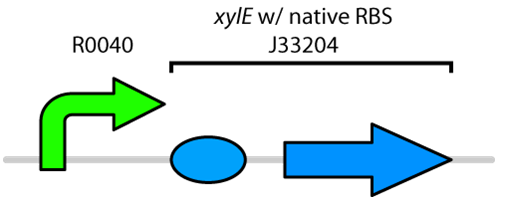
Catechol, a by-product of poly aromatic hydrocarbon metabolism, is a toxic compound found in tailings ponds. This toxic compound can be degraded by Catechol 2,3-dioxygenase, which is coded for by the gene XylE found on the Tol plasmid of Pseudomonas putida (Nakai et al., 1983). Previously there were only two available biobricks of this gene. One contained the XylE gene with its native RBS site and the other part had an added Pcst promoter, which was glucose-repressible. To tackle to problem of XylE being repressed, when the transformed E.coli was grown in media containing glucose, a TetR promoter was added to the part containing only the gene.
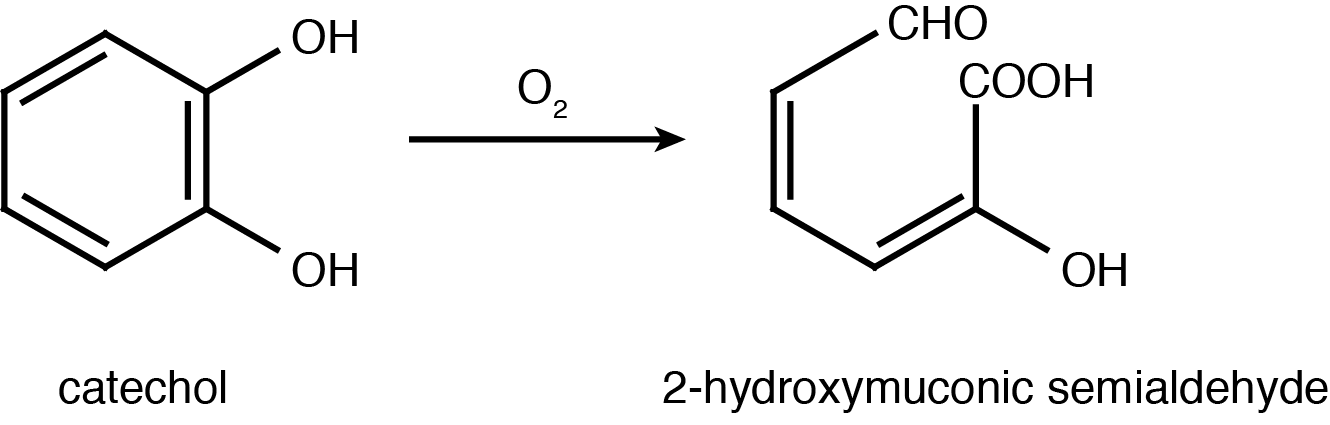
Catechol 2,3-dioxygenase is an extradiol dioxygenase which cleaves catechol adjacent to the two hydroxyl groups. When this occurs 2-hydroxymuconic semialdehyde is produced, which is yellow in colour. This colour change allows for easy assays to be conducted which show the enzymes activity.
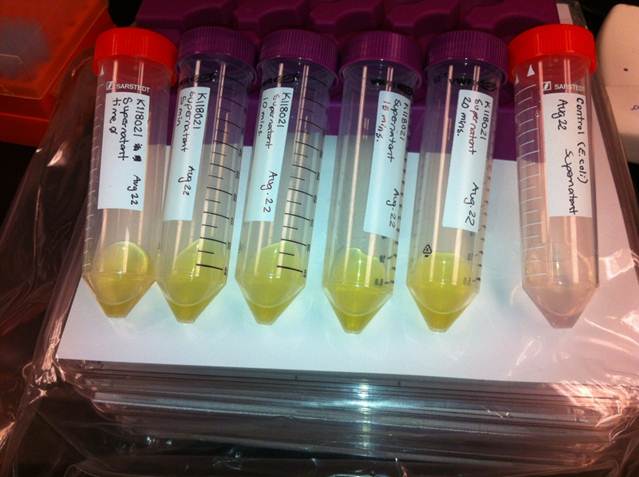
The colour change assays were performed with the newly constructed part by bringing the supernatant of an overnight culture to a concentration of 0.1 M of catechol. The catechol was added to the supernatant because the reaction takes place outside of the cell. Within minutes of the addition of catechol to the supernatant, the solution turned from the pale yellow of LB to a bright yellow. This assay was completed by following the previous assay done by the 2008 Edinburgh iGEM team.
References
Nakai, C., Kagamiyama, H., and Nozaki, M. 1983. Complete nucleotide sequence of the metapyrocatechase gene on the TOL plasmid of Pseudomonas putida. The Journal of Biological Chemistry 258: 2923-2928.
Shu, L., Chiou, Y., Orville, A.M., Miller, M.A., Lipscomb, J.D., and Que, L. 1995. X-ray absorption spectroscopic studies of the Fe(II) active site of catechol 2,3-dioxygenase. Implications for the extradiol cleavage mechanism. Biochemistry 34: 6649-6659.
 "
"