Team:Groningen/Modeling
From 2012.igem.org
(→Result) |
(→Result) |
||
| Line 31: | Line 31: | ||
Great headway towards implementing a transcription-regulation network into the FBA model. | Great headway towards implementing a transcription-regulation network into the FBA model. | ||
| - | # Identified sources for the network (DBTBS,RegPrecise). | + | # Identified sources for the network ([http://dbtbs.hgc.jp/ DBTBS],[http://regprecise.lbl.gov/RegPrecise/collection_taxspec.jsp?collection_id=5 RegPrecise]). |
| - | # Identified sources for expression data (KEGG,BSORF). | + | # Identified sources for expression data ([http://www.genome.jp/kegg/expression/ KEGG],[http://bacillus.genome.ad.jp/ BSORF]). |
| - | # Analyzed the article's script implementing PROM in MATLAB to understand the influence of current environmental cues on the algorithm. Result-No effect (Link) | + | # Analyzed the article's script implementing PROM in MATLAB to understand the influence of current environmental cues on the algorithm. Result-No effect ([http://www.pnas.org/content/107/41/17845 Link]) |
=== References === | === References === | ||
Revision as of 11:52, 3 August 2012








Contents |
Overview
The modeling portion of the project focuses on three major initiatives:
- Estimation of the allowed initial medium composition which would:
- Germinate the spores
- Raise the population to reporter-required levels within the smallest possible spoilage time-frame
- Maintain an active population at that level for a prolonged period of time, avoiding the onset of dormancy or sporulation
- The creation of a dynamic model for B. subtilis suitable for flux balance analysis which responds to environmental cues.
- The creation of a succinct explanation of TnrA activation, according to current literature.
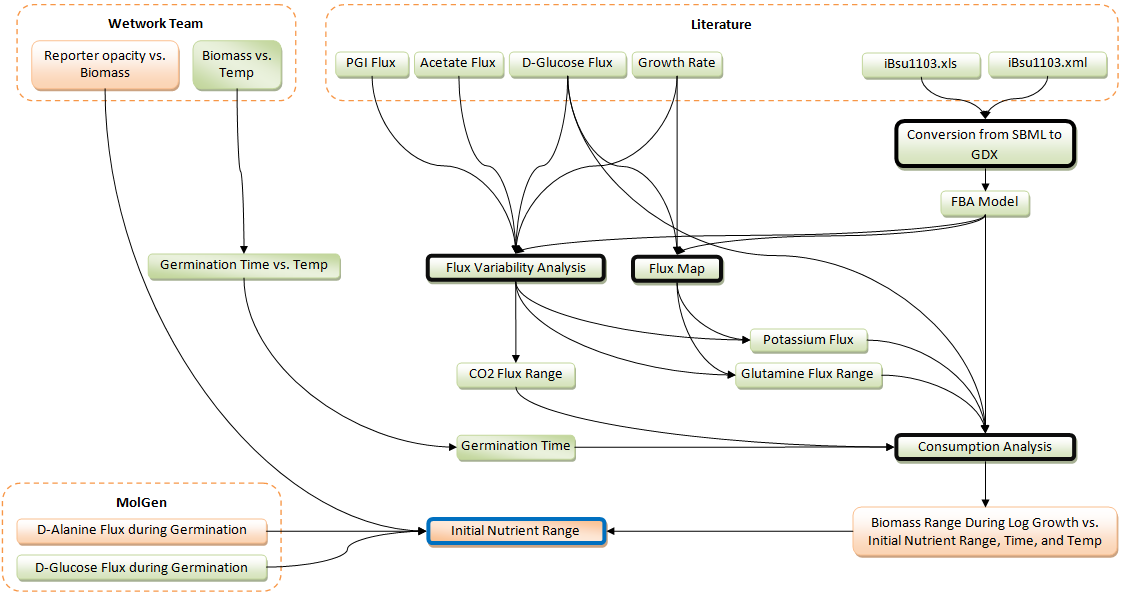
Initial Medium Composition
Purpose
The B. subtilis within the containment unit must go through distinct phases under defined time constraints. The only way to control the amount of biomass, and its behavior, is through the initial medium concentration. The containment unit does not contain any time-release capsules for providing a controlled level of nutrients.
Result
This is a work in progress. For now here is a chart showing the status of the involved steps:
Dynamic Model
Purpose
For our purposes, the dynamic model seeks to provide an explanation for the gene expression observed in the microarray experiment. However, for the scientific community in general, a dynamic model would be enable phenotype prediction over time under varying environmental conditions. Such a model would also be able to predict phenotypes for knock-out mutant strains.
Probabilistic integrative modeling (PROM) uses gene expression data across a wide variety of environmental conditions to quantify the link between the transcriptional-regulatory network and the stoichiometric metabolite-reaction matrix. In other words, PROM modulates the reaction fluxes by looking at the likelihood a given reaction is active given the current state of gene expression. This method has been shown to accurately predict growth phenotypes in knock-out strains of E. coli and M. tuberculosis.
Unfortunately, PROM does not consider the current environmental cues. It considers a multitude of prior environmental cues through the gene expression data. As such, it is really only useful for predicting fluxes in knock-out strains. Despite this limitation, it is still a great starting point for building a dynamic model as it considers both the transcriptional-regulatory network and a constraint-based stoichiometric model. It does not rely on the oft-inaccurate kinetic parameters used in ODE models.
Result
Great headway towards implementing a transcription-regulation network into the FBA model.
- Identified sources for the network ([http://dbtbs.hgc.jp/ DBTBS],[http://regprecise.lbl.gov/RegPrecise/collection_taxspec.jsp?collection_id=5 RegPrecise]).
- Identified sources for expression data ([http://www.genome.jp/kegg/expression/ KEGG],[http://bacillus.genome.ad.jp/ BSORF]).
- Analyzed the article's script implementing PROM in MATLAB to understand the influence of current environmental cues on the algorithm. Result-No effect ([http://www.pnas.org/content/107/41/17845 Link])
References
- N. E. Lewis, H. Nagarajan, and B. O. Palsson, “Constraining the metabolic genotype–phenotype relationship using a phylogeny of in silico methods,” Nature Reviews Microbiology, vol. 10, no. 4, pp. 291–305, Apr. 2012.
- S. Chandrasekaran and N. D. Price, “Probabilistic integrative modeling of genome-scale metabolic and regulatory networks in Escherichia coli and Mycobacterium tuberculosis,” PNAS, vol. 107, no. 41, pp. 17845–17850, Oct. 2010.
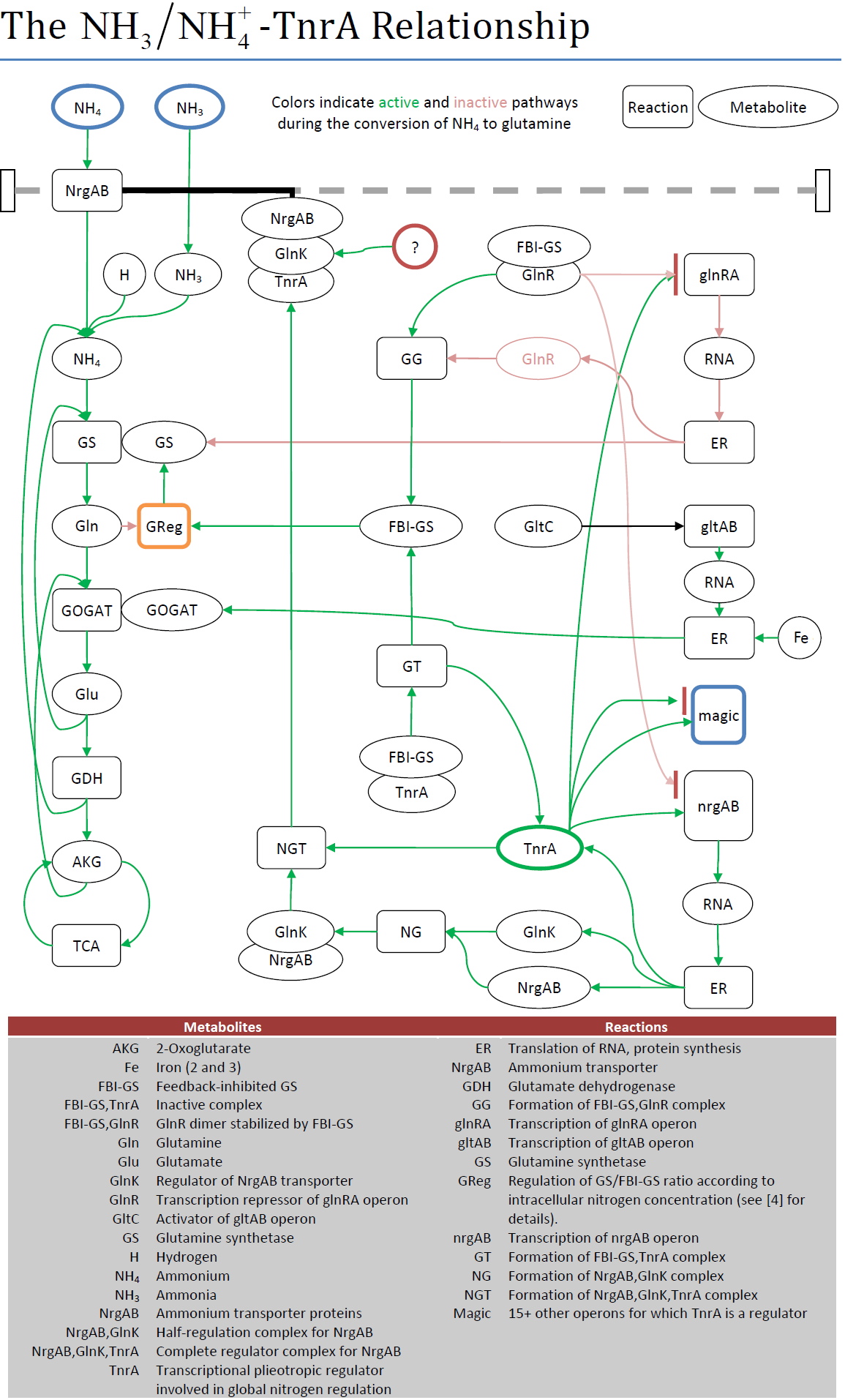
TnrA Activation
Purpose
Prior to the successful microarray experiment conducted by the wetwork team, the proposed volatile sensing mechanism tied reporter activation to the metabolism of ammonium/ammonia (NH4/NH3+). The nitrogen metabolism in B. subtilis is a convoluted mesh of reactions and (in)activation complexes mostly controlled by the TnrA transcription factor. In order to observe the effect of NH4 uptake on the TnrA, it was necessary to have a concise behavioral diagram. Unfortunately, no such diagram existed. The diagram for nitrogen metabolism on the KEGG database identified most of what was involved, but did so in an unclear manner.
Result
The figure below used the behavioral information from literature to highlight the active and inactive pathways during ammonium uptake. In terms of the project, the creation of this diagram brought to light a critical problem for using TnrA to sense extracellular NH4/NH3+; TnrA is only active when glutamine synthetase (GS) is actively converting NH4 to glutamine. This in turn is regulated by cell growth, not the amount of NH4/NH3+ present. However, in one article it was observed that GS is also active under conditions of nitrogen limitation. If the medium contained the precise amount of glutamine necessary to support the initial growth phase, then the glutamine would be depleted at the sensing phase. GS would activate to try and create more glutamine. GS would only deactivate when the amount of extracellular NH4/NH3+ reached a level to remove the lack of nitrogen as factor limiting further growth.
The real result? If this sensing pathway was selected, then the wetwork team would have to find the necessary levels of glutamine to exploit the edge of nitrogen limitation. Possible, but not exactly the easiest mechanism to implement for the consumer market.
| Fig. m1. Reactions involved between ammonium uptake and TnrA. |
References
- KEGG Database, "Nitrogen Metabolism - Bacillus subtilis", Kanehisa Laboratories, Last Modified: July 23, 2010. http://www.kegg.jp/kegg-bin/show_pathway?org_name=bsu&mapno=00910
- SubtiWiki, http://subtiwiki.uni-goettingen.de/wiki/index.php
- K. Gunka and F. M. Commichau, “Control of glutamate homeostasis in Bacillus subtilis: a complex interplay between ammonium assimilation, glutamate biosynthesis and degradation,” Molecular Microbiology, vol. 85, no. 2, pp. 213–224, 2012.
- N. Doroshchuk, M. Gelfand, and D. Rodionov, “Regulation of nitrogen metabolism in gram-positive bacteria,” Molecular Biology, vol. 40, no. 5, pp. 829–836, 2006.
- Study Guide, Chem153C, University of California, Los Angeles. http://vohweb.chem.ucla.edu/voh/classes%5Cspring10%5C153CID28%5C11AminoAcidBiosynthesisSQA.pdf
 "
"