Team:University College London/LabBook/Week8
From 2012.igem.org



Contents |
Monday 30.6.12
Aim: Repeat the gel from Expt 7.3 (week 7) with a smaller ladder and higher agarose percentage in order to detect the very small inserts of BBa_J23119 (35bp) and BBa_B0034 (12hp). Previous ladder used was 10037 bp, and this time we used a 25bp ladder.
Step 1 - Thawing cells: Thaw all materials on ice
Step 2 - Adding Ingredient: Add the following ingredients to autoclaved/sterile eppendorf tubes
| Component | Amount (ul) (one enzyme used) | Amount (ul) (two enzymes used) |
|---|---|---|
| dH20 | 2.5 | 1.5 |
| Buffer 1x | 1 | 1 |
| DNA template | 5 | 5 |
| BSA | 0.5 | 0.5 |
| Enzyme 1 | 1 | 2 |
| Enzyme 2 | N/A | 1 |
Step 3 - Addition of BioBrick: Flick contents gently and centrifuge.
Step 4 - Centrifuge:
RPM: 14000
Time: 1 minute
Temperature: 18oC
Step 5 - Digest Program: Place the samples on a thermocycler under the following conditions:
RPM: 550
Time: 2.5 hours
Temperature: 37oC
Step 6 - Denaturing Enzymes: If you are not running the samples on a gel immediately, denature the restriction enzymes by running the samples on a thermocycler under the following conditions:
RPM: 550
Time: 25 minutes
Temperature: 65oC
Step 2 - Setting up Digests and Controls: The protocol describes the recipe for (i) Digested Plasmid and (ii) Uncut Control. The table below indicates that an uncut and an Xba1/Spe1 digested sample be set up for each BioBrick. Set up Eppendorfs as follows
| Samples | Recipe | Enzymes | |
|---|---|---|---|
| BioBrick | BBa_J23119 | Digested Plasmid | Xba1 & Spe1 |
| Undigested Plasmid (Control) | None | ||
| BBa_B0034 | Digested Plasmid | Xba1 & Spe1 | |
| Undigested Plasmid (Control) | None | ||
Preparing the Gel
Step 1: Within a conical flask, add 3ml 50X TAE, 1.5g Agarose, and 150ml RO water
Step 2: Heat for 1 min in microwave. Swirl. Heat again for 30s. If solution is clear stop. Else repeat.
Step 3: Cool solution under running cold water.
Step 4: Add 20ul ethidium bromide (normal concentration of EB solution is 500ug/ul)
Step 5: Pour into a sealed casting tray in a slow steady stream, ensuring there are no bubbles
Running a gel
Step 6: Add 1 part loading buffer to five parts of loading sample
Step 7: Position the gel in the tank and add TAE buffer, enough to cover the gel by several mm
Step 8: Add 5ul of DNA ladder to lane 1
Step 9: Add samples to the remaining wells
Step 10: Run at 100 volts for 1hour and 15 minutes
Imaging the Gel
Step 11: Place gel in GelDoc 2000 chamber
Step 12: Turn GelDoc 2000 chamber on
Step 13: From computer: Quantity One > Scanner > Gel_Doc_Xr>Manuqal Acquire
Step 14: Alter the exposure/settings to give a clear image.
TAE - Tris-acetate-EDTA
EDTA - ethylenediamine tetraacetic acid
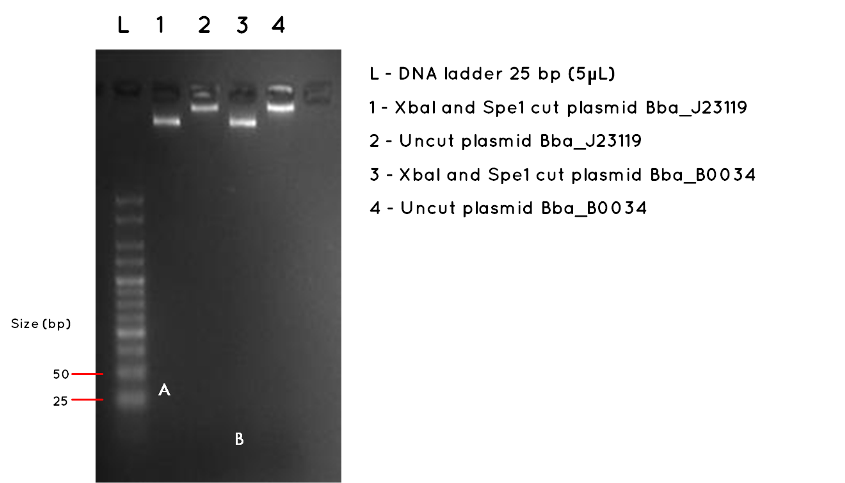
Results:The image below shows a 3.5% Agarose Gel of an Analytical Restriction Enzyme Digest for Expt 7.3. The high agarose concentration of the gel was intended to slow the progression of DNA fragments to enable us to detect the small inserts of BBa_J23119 and BBa_B0034. The table below indicates the expected sizes of BioBricks and Plasmid Backbones. Accordingly, we used a the smallest available ladder, with fragments ranging from 25bp to 500bp. At the top of the gel it is possible to see the large fragments we detected on a 1000bp gel in Expt 7.3 Week 7. However, we had expected this gel to also demonstrate the BBa_J23119 insert (35bp) corresponding to letter A in Lane 1, and the BBa_B0034 insert, corresponding to letter B in Lane 3. No product was found. As expected, there was no product measurable against the 25bp ladder for the uncut plasmids in Lane 2 and Lane 4.
| BioBrick | Expected BioBrick Size (bp) | Plasmid Backbone | Plasmid Expected Size (bp) | Combined Size |
|---|---|---|---|---|
| BBa_J23119 | 35 | PSB1A2 | 2079 | 2114 |
| BBa_B0034 | 12 | PSB1A2 | 2079 | 2091 |
Conclusion:No product was detected for either BioBrick insert. However, we do no feel this necessarily proves the transformation failed, especially as we detected the correct plasmid backbone for each in week 7 (Expt 7.3). The modest nanodrop concentration obtained last week suggests that the concentration was not high enough to allow such a small fragment to be visualised. We are considering alternatives such as running a polyacrylamide gel, which is more sensitive to small fragments, or amplifying the fragment using PCR

Monday 30.7.12
Aim - Picking colonies: Colonies were picked for BBa_I750016, which demonstrated colony formation in Expt 7.4 on Friday 27.7.12.
Step 2 - Inoculating Colonies into a Selective Broth:: Add Yul of antibiotic to reach desired antibiotic concentration.
(For Ampicillin this is 50ug/ml, For Kanamycin it is 25ug/ml, for Tetracycline it is 15ug/ml, and for Chloramphenicol it is 25ug/ml)
Step 4 – Selecting a Colony: Select a clear, isolated colony and using an inoculation hoop scoop up a colony onto the tip. Deposit in the falcon tube
Step 5 - Culture: Culture your falcon tubes overnight at a temperature of 37 oC. Leave for no longer than 16 hours.
Step 2 – Inoculating Colonies into a Selective Broth: The table below indicates the volume of broth and the concentration of antibiotic required for each BioBrick.
| Samples | Volume Inoculated | Broth (ml) | Antibiotic (ug/ml) | |
|---|---|---|---|---|
| BioBrick | BBa_I750016 | 10ul | Lysogeny Broth (5) | Ampicillin(50ug/ml) |
| 90ul | ||||
Tuesday 31.7.12
Aim – Results from Colony Picking
Results: The table below indicates whether there was growth for BBa_I1750016
| Samples | Volume Inoculated | Colony Formation | |
|---|---|---|---|
| BioBrick | BBa_I1750016 | 10μl | Yes |
| 90μl | Yes | ||
Conclusion: We can proceed onto Miniprep, Analytical Digest, and Nanodrop.
Method
Miniprep:
Step 1 - Pellet Cells: Pellet 1.5-5ml bacterial culture containing the plasmid by centrifugation g = 6000
Time = 2 min
Temperature = (15-25oC)
Step 2 - Resuspend Cells: Add 250ul S1 to the pellet and resuspend the cells completely by vortexing or pipetting. Transfer the suspension to a clean 1.5ml microcentrifuge tube.
Step 3 - Puncturing Cell Membrane: Add 250ul S2 gently mix by inverting the tube 4-6 times to obtain a clear lysate. Incubate on ice or at room temperature for NOT longer than 5 min.
Step 4 - Neutralising S2: Add 400ul Buffer NB and gently mix by inverting the tube 6-10 times, until a white precipitate forms.
Step 5 - Centrifuge:
g: 14000
Time:10 minutes
Temperature: (15-25oC)
Step 6 - Centrifuge: Transfer the supernatant into a column assembled in a clean collection tube (provided. Centrifuge:
g = 10,000
Time: 1 minute
Temperature: (15-25oC)
Step 7 - Wash Column: Discard the flow-through and wash the spin column by adding 700ul of Wash Buffer. Centrifuge:
g - 10,000
Time - 1 minute
Temperature: (15-25oC)
Step 8 - Remove residual ethanol: Centrifuge:
g - 10,000
Time - 1 minute
Temperature: (15-25oC)
Step 9 - Elute DNA: Place the column into a clean microcentrifuge tube. Add 50-100ul of Elution Buffer, TE buffer or sterile water directly onto column membrane and stand for 1 min. Centrifuge:
g - 10,000
Time - 1 minute
Temperature: (15-25oC)
Step 10 - Storage: Store DNA at 4oC or -20oC
Restriction Digest:
Step 1 - Thawing cells: Thaw all materials on ice
Step 2 - Adding Ingredient: Add the following ingredients to autoclaved/sterile eppendorf tubes
| Component | Amount (ul) (one enzyme used) | Amount (ul) (two enzymes used) |
|---|---|---|
| dH20 | 2.5 | 1.5 |
| Buffer 1x | 1 | 1 |
| DNA template | 5 | 5 |
| BSA | 0.5 | 0.5 |
| Enzyme 1 | 1 | 2 |
| Enzyme 2 | N/A | 1 |
Step 3 - Addition of BioBrick: Flick contents gently and centrifuge.
Step 4 - Centrifuge:
RPM: 14000
Time: 1 minute
Temperature: 18oC
Step 5 - Digest Program: Place the samples on a thermocycler under the following conditions:
RPM: 550
Time: 2.5 hours
Temperature: 37oC
Step 6 - Denaturing Enzymes: If you are not running the samples on a gel immediately, denature the restriction enzymes by running the samples on a thermocycler under the following conditions:
RPM: 550
Time: 25 minutes
Temperature: 65oC
Step 2 - Setting up Digests and Controls: The protocol describes the recipe for (i) Digested Plasmid and (ii) Uncut Control. The table below indicates that an uncut and an Xba1/Spe1 digested sample be set up for each BioBrick. Set up Eppendorfs as follows
| Samples | Recipe | Enzymes | |
|---|---|---|---|
| BioBrick | BBa_I1750016 | Digested Plasmid | Xba1 & Spe1 |
| Undigested Plasmid (Control) | None | ||

Tuesday 31.7.12
Aim - Transformation of previously failed BioBricks: BBa_C0040 was transformed in Expt 7.4, but appeared to be contaminated. BBa_R0040, as part of the same experiment, did not form colonies at all after transformation. Therefore both were reattempted.
Method:
Step 1 - Addition of BioBrick: To the still frozen competent cells, add 1 - 5 µL of the resuspended DNA to the 2ml tube.
Step 4 - Incubation: Close tube and incubate the cells on ice for 45 minutes.
Step 5 - Heat Shock: Heat shock the cells by immersion in a pre-heated water bath at 37ºC for 10 minutes.
Step 6 - Incubation: Incubate the cells on ice for 2 minutes.
Step 7 - Add media: Add 1.5ml of Lysogeny Broth and transfer to a falcon tube.
Step 8 - Incubation: Incubate the cells at 37ºC for 1 hour at RPM 550.
Step 9 - Transfer: transfer the solution back into a 1.5ml Eppendorf and centrifuge
RPM: 14000
Time: 2 minutes
Temperature (18-25oC)
Step 10 - Resuspend:Remove supernatant and resuspend in 100ul LB
Step 11 - Plating: Spread the resuspended cell solution onto a selective nutrient agar plate. Place the plates in a 37°C static incubator, leave overnight (alternatively a 30°C static incubator over the weekend)
Step 1 – Thawing Cells: Use W3100 cell line created in Week 2 (Expt 2.1)
Step 3 – Addition of BioBrick: To each 2ml eppendorf, add 1ul of the following BioBricks. Include an extra tube as a control, with no BioBrick added
| Function | Module | ||
|---|---|---|---|
| BioBrick | BBa_C0040 | Tetracycline Repressor | Buoyancy |
| BBa_R0040 | TetR Repressible Promoter | Buoyancy | |
| Control | Positive (one for each of the above BioBricks) | ||
| Negative (No BioBrick) | |||
Step 9 – Plating samples on Agar Plates: The table below indicates the chosen inoculation volume (two for each BioBrick) and the correct gel antibiotic concentration for all samples.(Extra caution was taken to allow agar to cool before adding Ampicillin, in case this is the cause of difficulty).
| Samples | Volume Inoculated | Antibiotic in Gel (ug/ml) | |
|---|---|---|---|
| BioBrick | BBa_C0040 | 10ul | Ampicillin(50ug/ml) |
| 90ul | |||
| BBa_R0040 | 10ul | ||
| 90ul | |||
| Control | Positive (Contains BioBrick BBa_C0040) | 36ul | No Antibiotic |
| Negative (No BioBrick) | 36ul | 1x Ampicillin(50ug/ml) | |

8-2

8-3

8-4

8-5
 "
"