Team:Bielefeld-Germany/Results/Summary
From 2012.igem.org
KevinJarosch (Talk | contribs) (→BPUL - Laccase from Bacillus pumilus DSM 27 (ATCC7061)) |
KevinJarosch (Talk | contribs) (→BPUL - Laccase from Bacillus pumilus DSM 27 (ATCC7061)) |
||
| Line 128: | Line 128: | ||
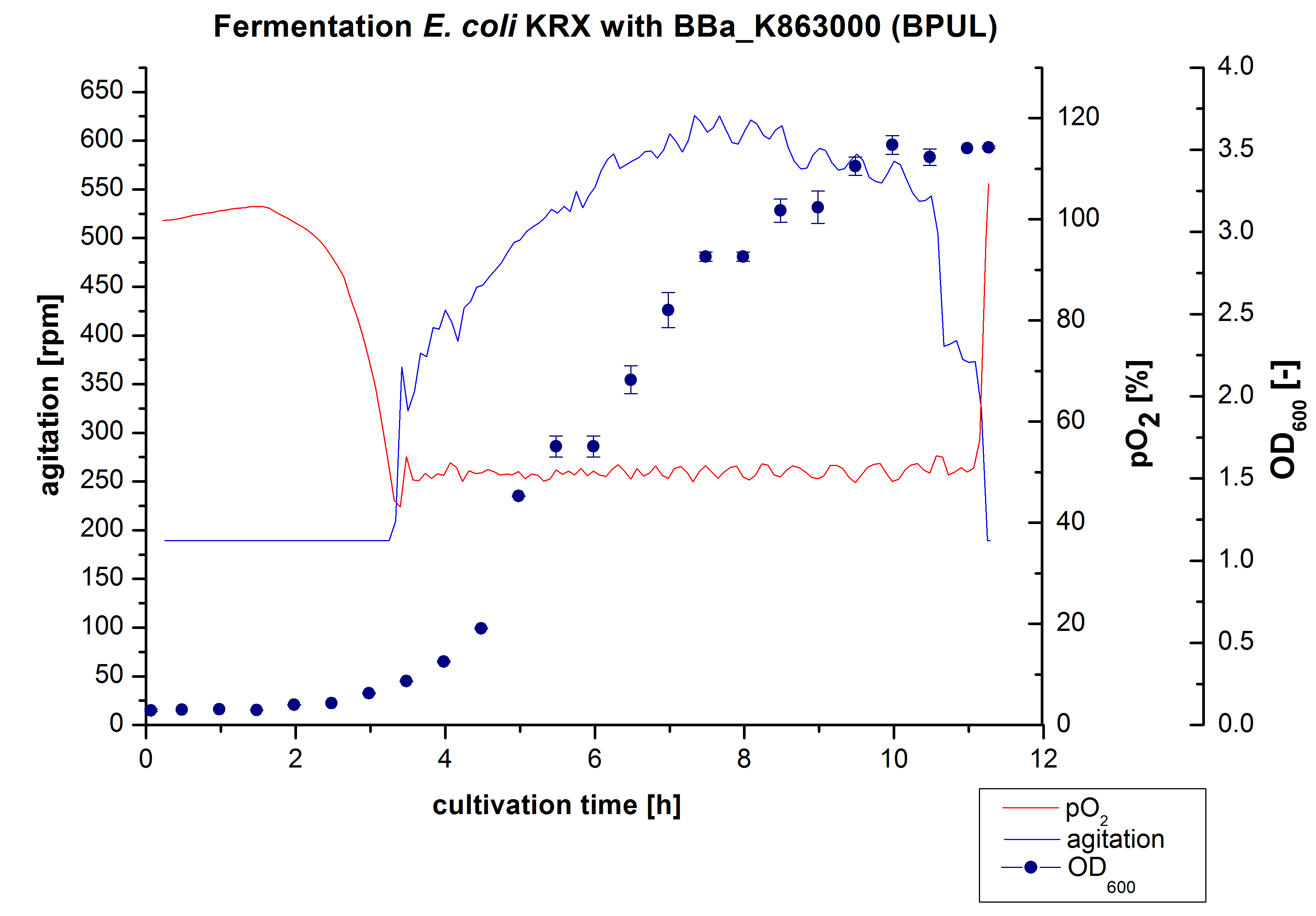
[[File:Bielefeld2012_BPUL3LFermentation.jpg|600px|thumb|center|Figure 1: Fermentation of ''E. coli'' KRX with <partinfo>BBa_K863000</partinfo> (BPUL) in Braun Biostat B, scale: 3 L, [https://2012.igem.org/Team:Bielefeld-Germany/Protocols/Materials#Autoinduction_medium autoinduction medium] + 60 µg/mL chloramphenicol, 37 °C, pH 7, agitation on cascade to hold a pO<sub>2</sub> of 50 %, OD<sub>600</sub> taken every 30 minutes.]] | [[File:Bielefeld2012_BPUL3LFermentation.jpg|600px|thumb|center|Figure 1: Fermentation of ''E. coli'' KRX with <partinfo>BBa_K863000</partinfo> (BPUL) in Braun Biostat B, scale: 3 L, [https://2012.igem.org/Team:Bielefeld-Germany/Protocols/Materials#Autoinduction_medium autoinduction medium] + 60 µg/mL chloramphenicol, 37 °C, pH 7, agitation on cascade to hold a pO<sub>2</sub> of 50 %, OD<sub>600</sub> taken every 30 minutes.]] | ||
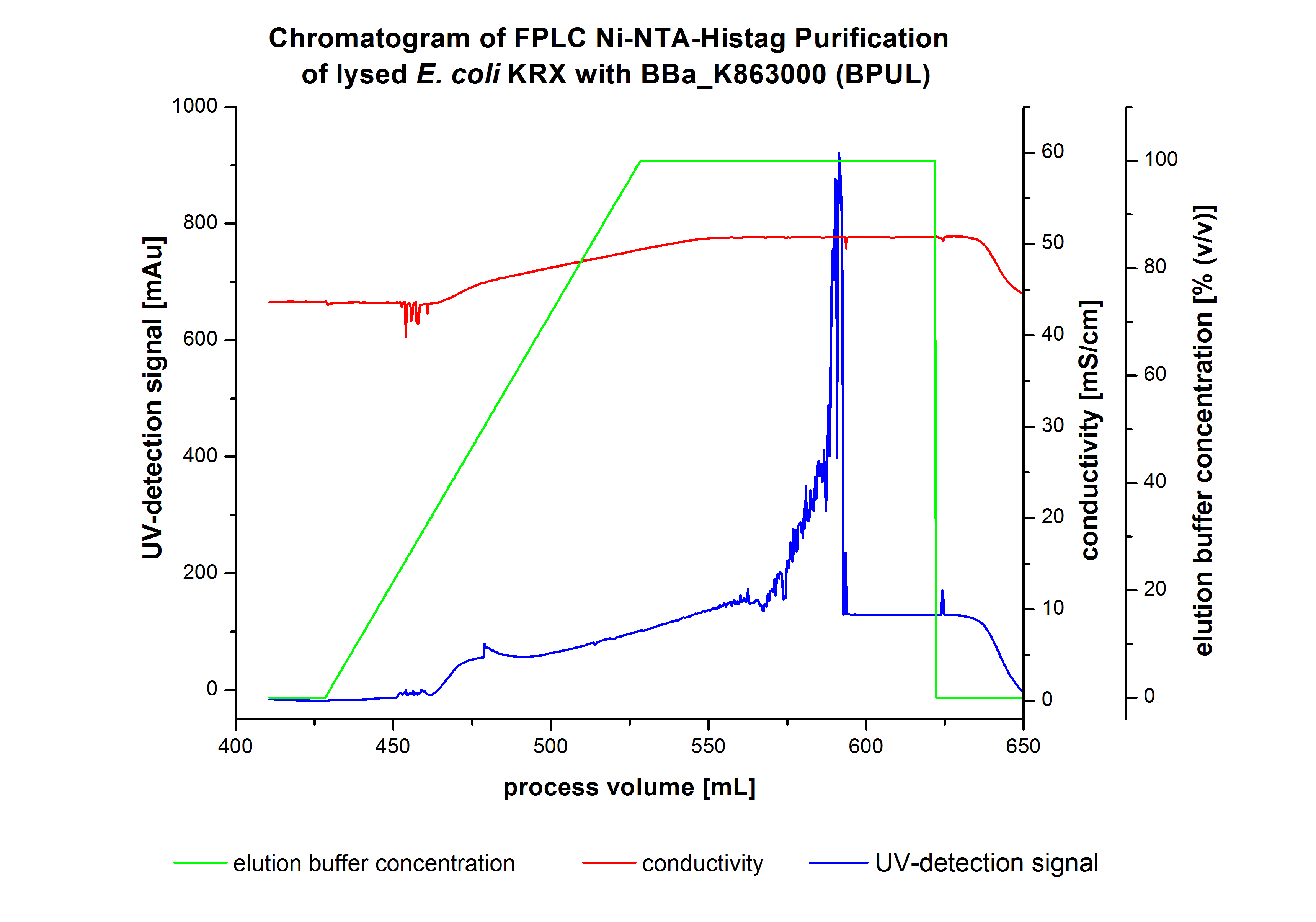
| - | The harvested cell were resuspended in Ni-NTA-euquilibrationbuffer, mechanically lysed by homogenization and centrifuged. The supernatant of the lysed cell paste were loaded on the Ni-NTA | + | The harvested cell were resuspended in Ni-NTA-euquilibrationbuffer, mechanically lysed by homogenization and centrifuged. The supernatant of the lysed cell paste were loaded on the Ni-NTA-column (15ml Ni-NTA resin) with a flowrate of 1 mL min<sup>-1</sup> cm<sup>-2</sup>. After loading the supernatant on the column, the column was washed by 10 column volumes (CV) with equilibrationbuffer.The bounded Protein's were eluted by an increasing elutionbuffer gradient from 0% to 100% with a length of 200mL. The chromatogramm of the BPUL-elution is shown in the following graph: |
| + | |||
<br style="clear: both" /> | <br style="clear: both" /> | ||
[[File:Bielefeld2012_BPUL3LChromatogramm.jpg|600px|thumb|center|Figure 2: Chromatogramm of Wash and Elution from FLPC Ni-NTA-Histag Purification of BPUL produced by 3 L fermentation of ''E. coli'' KRX with <partinfo>BBa_K863000</partinfo>.]] | [[File:Bielefeld2012_BPUL3LChromatogramm.jpg|600px|thumb|center|Figure 2: Chromatogramm of Wash and Elution from FLPC Ni-NTA-Histag Purification of BPUL produced by 3 L fermentation of ''E. coli'' KRX with <partinfo>BBa_K863000</partinfo>.]] | ||
Revision as of 14:37, 23 September 2012
Summary
BPUL - Laccase from Bacillus pumilus DSM 27 (ATCC7061)
After the measurement of activity of BPUL we made a scale-up and fermented E. coli KRX with <partinfo>BBa_K863000</partinfo> in Braun Biostat B with a total volume of 3 L. Agitation speed, pO2 and OD600 were determined and illustrated in Figure 1. We got a long lag phase of 2 hours due to a relativly old preculture. The cell growth caused a decrease in pO2 and after 3 hours the value fell below 50 %, so that the agitation speed was increased automatically. After 8,5 hours the deceleration phase started and therefore the agitation speed was decreased. There is no visible break through induction of protein expression. The maximal OD600 of 3,53 was reached after 10 hours, which means a decrease of 28 % in comparison to the fermentation of E. coli KRX under the same conditions(OD600,max =4,86 after 8,5 hours, time shift due to long lag phase). The cells were harvested after 11 hours.
The harvested cell were resuspended in Ni-NTA-euquilibrationbuffer, mechanically lysed by homogenization and centrifuged. The supernatant of the lysed cell paste were loaded on the Ni-NTA-column (15ml Ni-NTA resin) with a flowrate of 1 mL min-1 cm-2. After loading the supernatant on the column, the column was washed by 10 column volumes (CV) with equilibrationbuffer.The bounded Protein's were eluted by an increasing elutionbuffer gradient from 0% to 100% with a length of 200mL. The chromatogramm of the BPUL-elution is shown in the following graph:
- !!SDS-Page!!
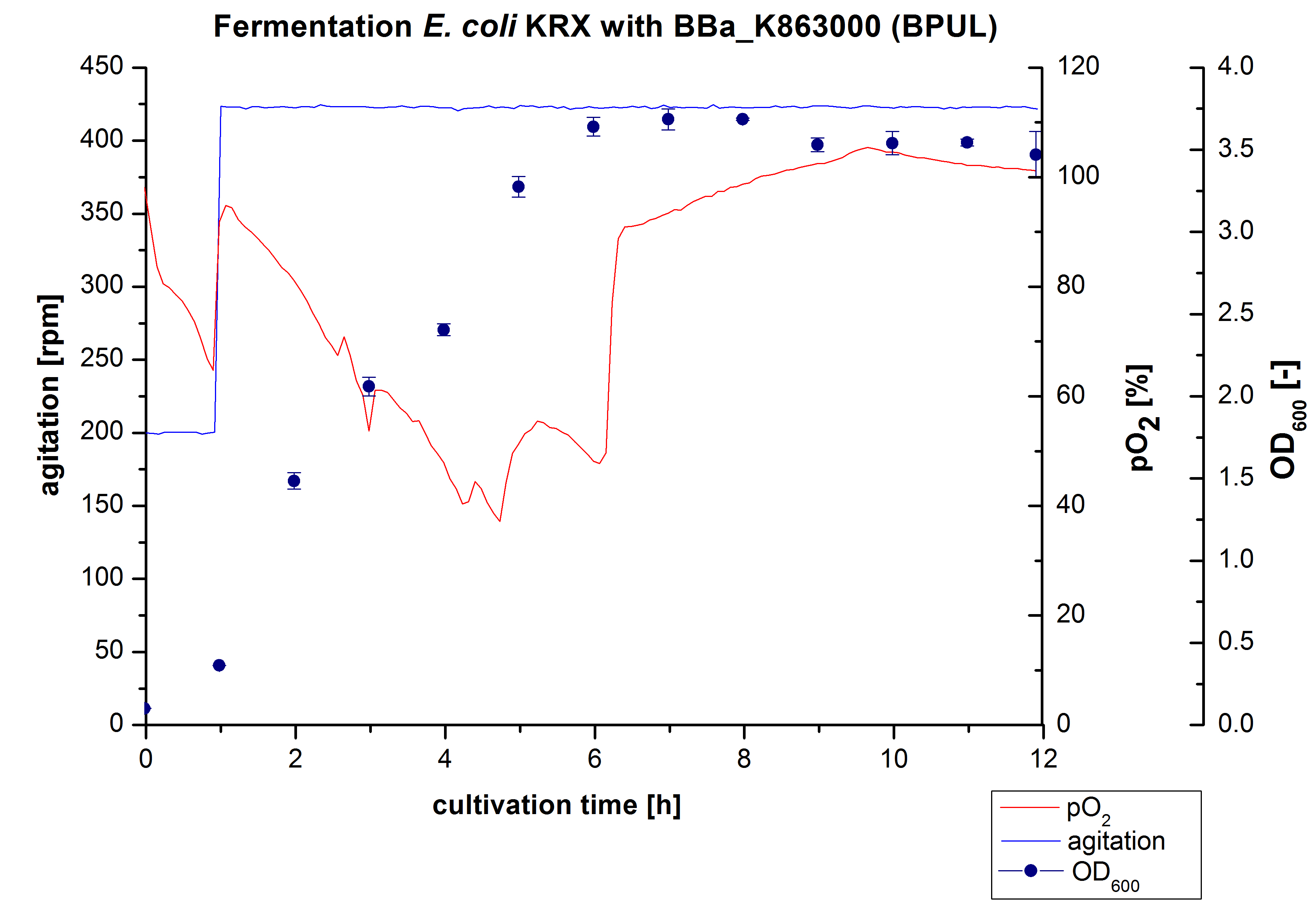
Another scale-up of the fermentation of E. coli KRX with <partinfo>BBa_K863000</partinfo> was made up to a final working volume of 6 L in Bioengineering NFL22. Agitation speed, pO2 and OD600 were determined and illustrated in Figure 3. There was no noticeable lag phase and the cells immediatly began to grow. Agitation speed was increased up to 425 rpm after one hour due to control problems. Then the pO2 sank until a cultivation time of 4,75 hours, when the deceleration phase started. Then it increased again. There is no visible break through induction of protein expression. A maximal OD600 of 3,68 was reached after 7/8 hours of cultivation, which is similar to the 3 L fermentation (OD600 = 3,58 after 10 hours, time shift due to long lag phase). The cells were harvested after 12 hours.
- !!SDS-Page!!
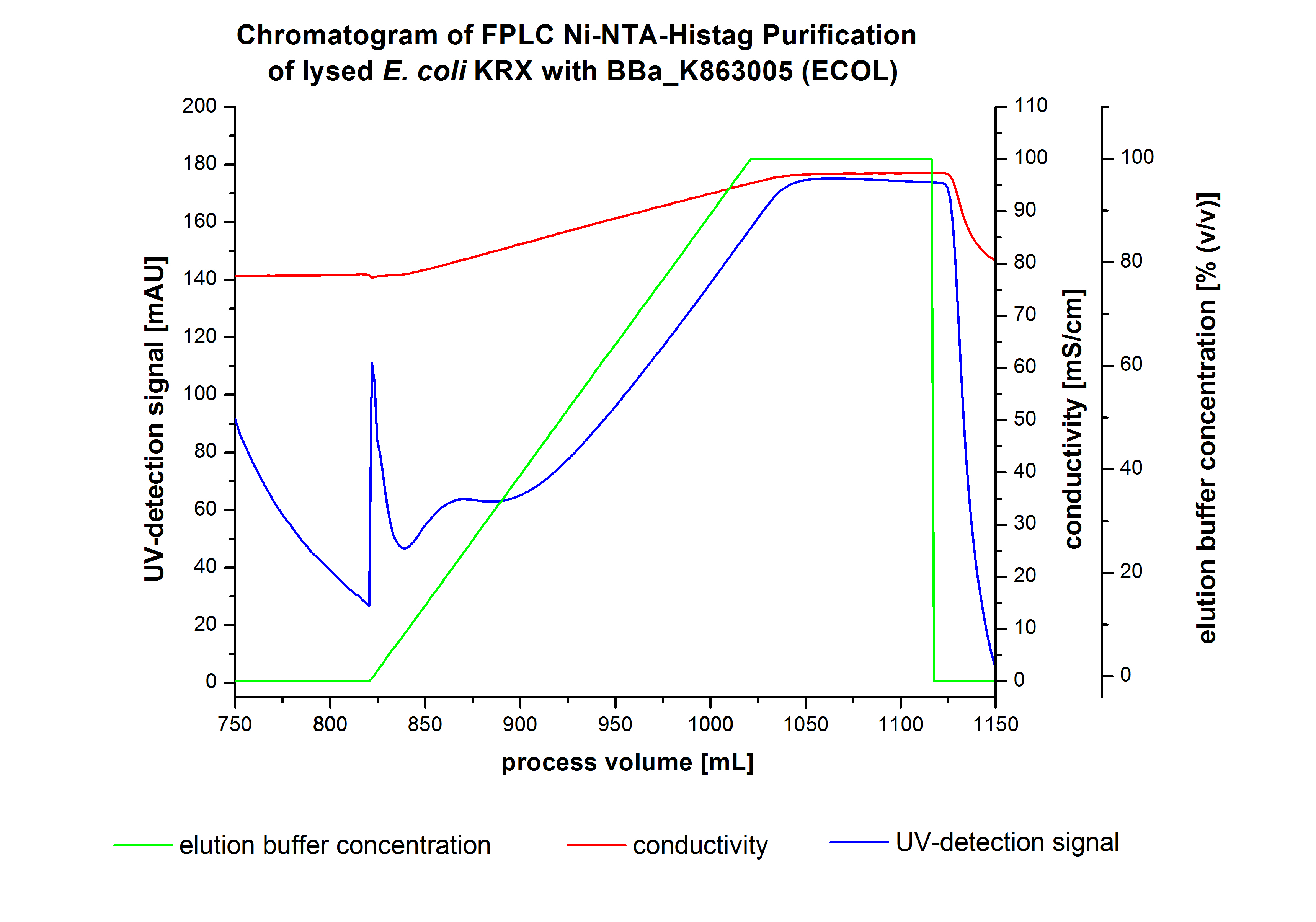
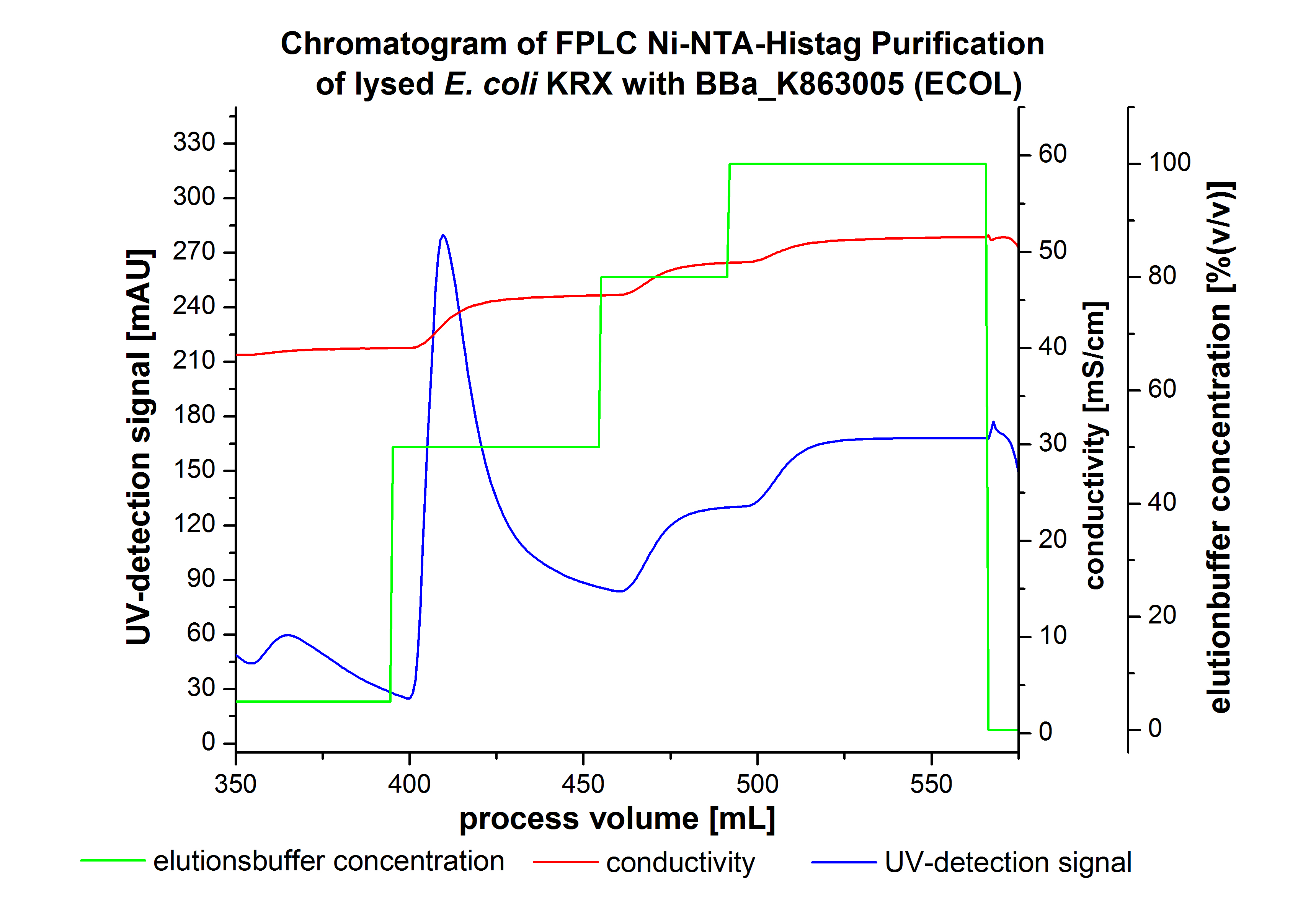
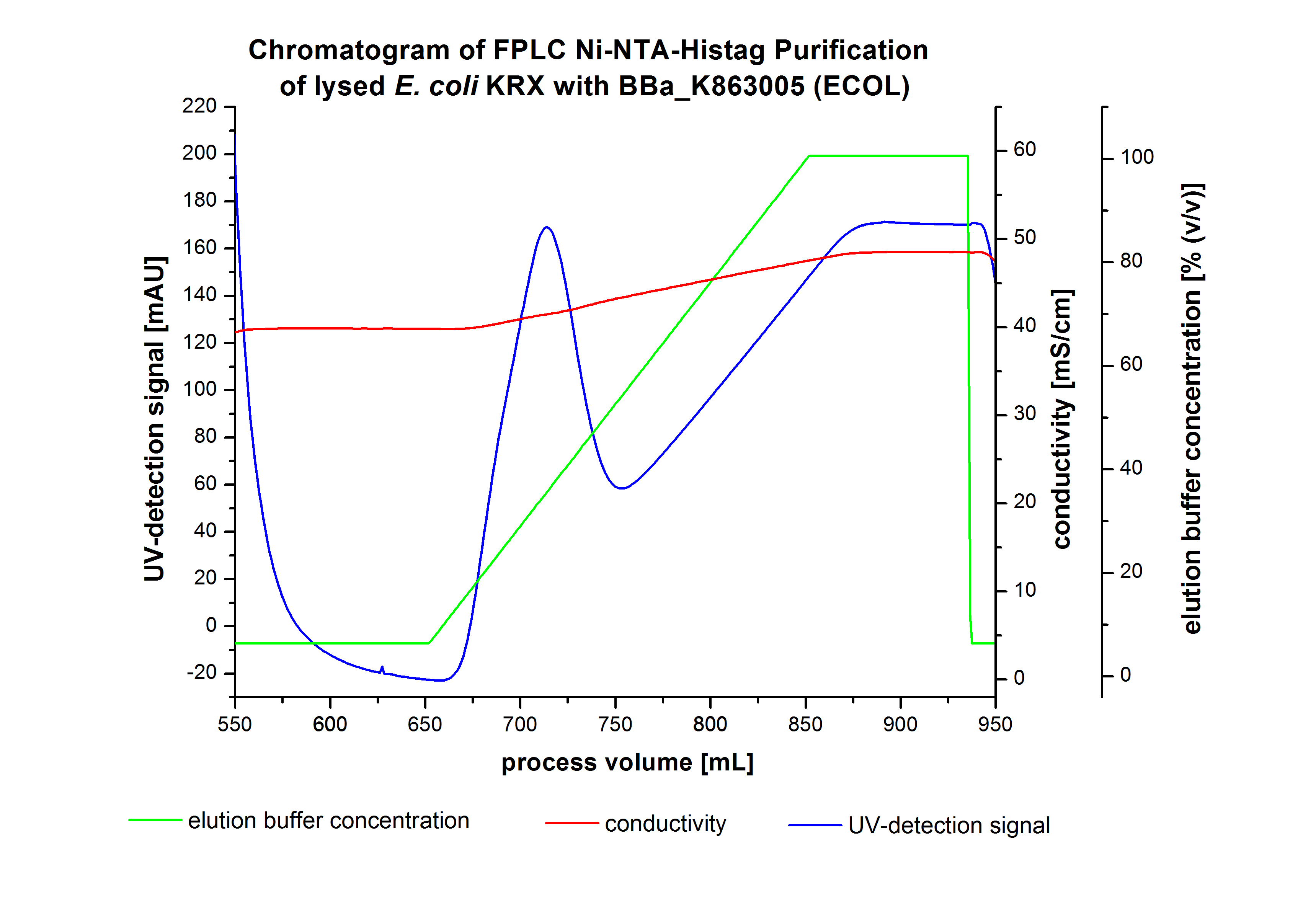
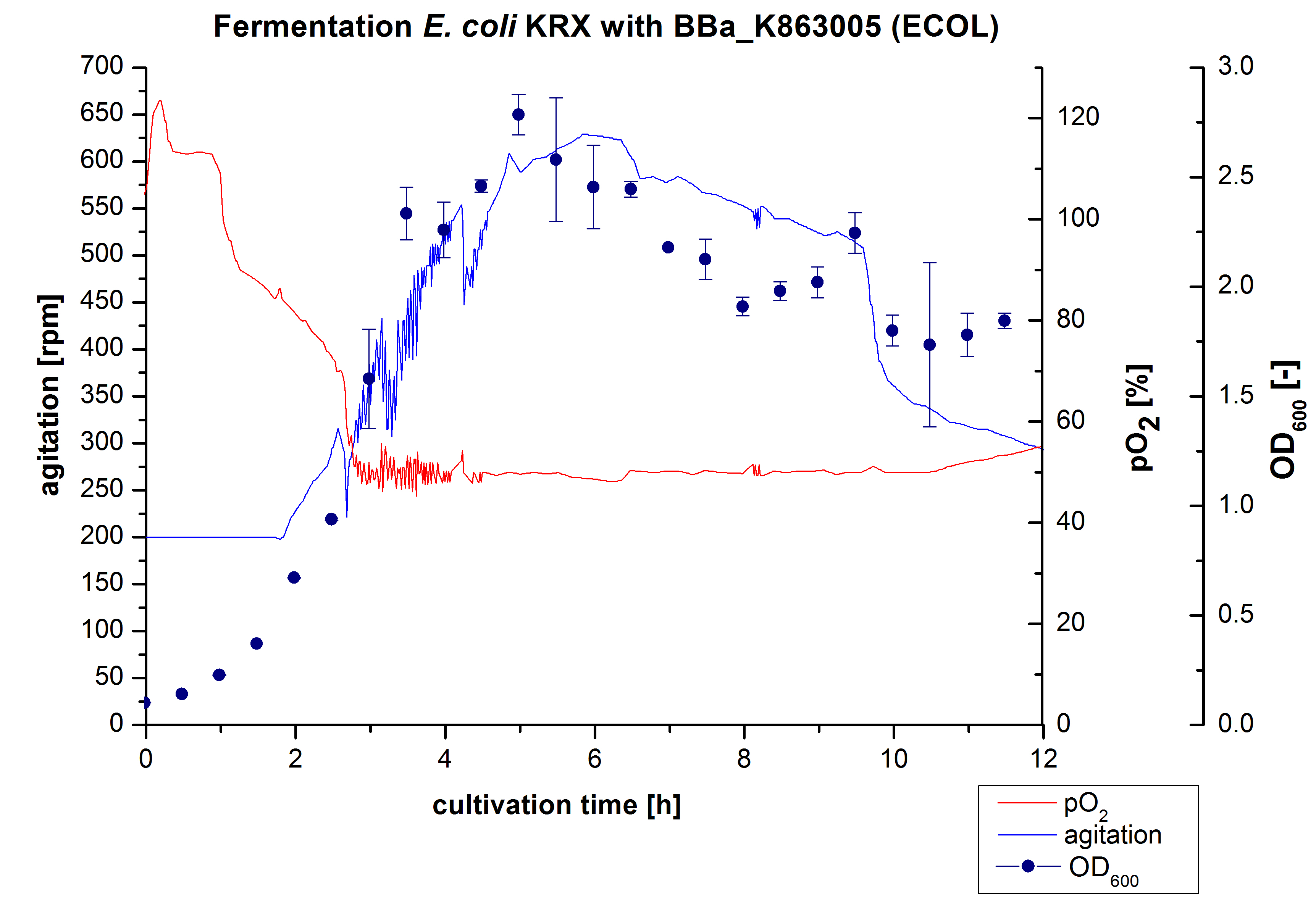
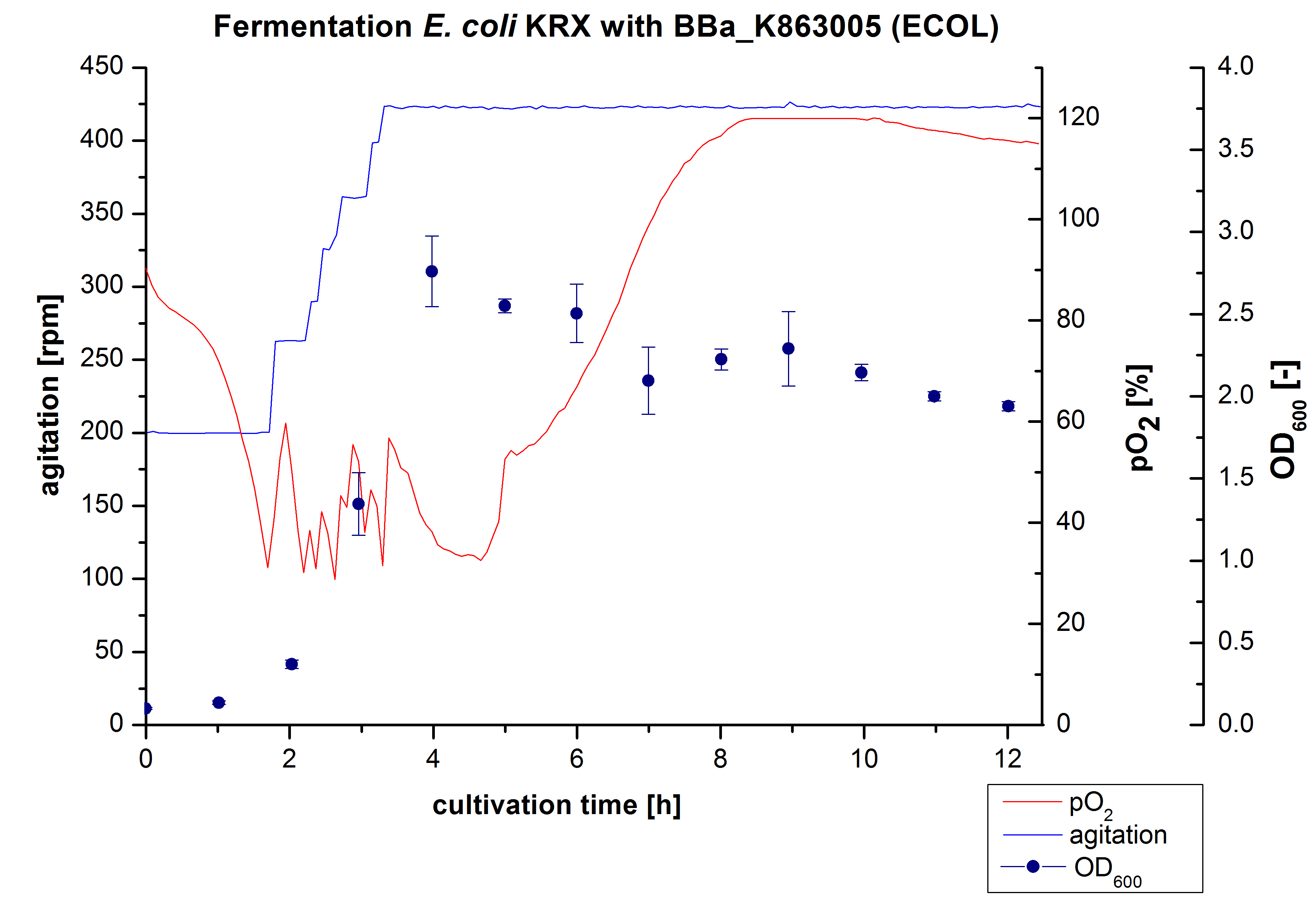
ECOL - Laccase from Escherichia coli BL21 (DE3)
- !!SDS-Page!!
- !!SDS-Page!!
XCCL - Laccase from Xanthomonas campestris pv. campestris B100
BHAL - Laccase from Bacillus halodurans C-125
TTHL - Laccase from Thermus thermophilus HB27
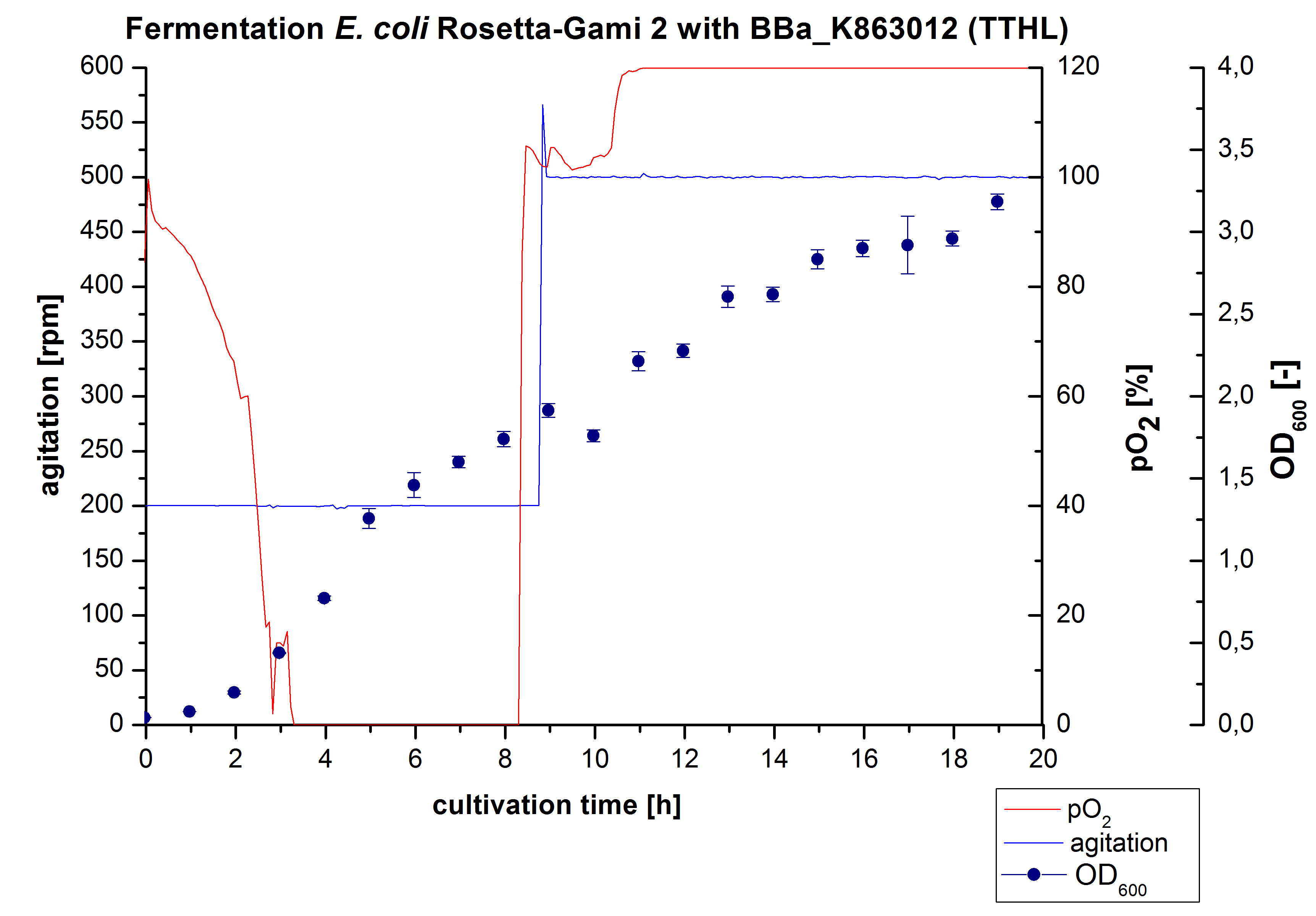
- !!SDS-Page!!
PCIL - Laccase from Pycnoporus cinnabarinus
TVEL5 - Laccase from Trametes versicolor
TVEL10 - Laccase from Trametes versicolor
TVEL13 - Laccase from Trametes versicolor
TVEL20 - Laccase from Trametes versicolor
| 55px | | | | | | | | | | |
 "
"