Team:HokkaidoU Japan/Notebook/aggregation Week 12
From 2012.igem.org
September 17th
Digestion of RBS-phaC-RBs-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2 and ptet-pSB1A2
To make a construct of ptet-RBS-phaC-RBS-phaA-RBS-phaB-RBS-dT-pSB1A2, we digested RBS-phaC-RBs-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2 with XbaI & PstI, ptet-pSB1A2 with SpeI & PstI. Insert (RBS-phaC-RBs-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2)
| DNA solution ( 45=50ng/ul) | 27 ul |
| XbaI | 1 ul |
| PstI | 1 ul |
| 10xM buffer | 4 ul |
| DW | 7 ul |
| Total | 40 ul |
Vector(ptet-pSB1A2)
| DNA solution (about 20 ng/ul) | 4 ul |
| SpeI | 1 ul |
| PstI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 12 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 120 |
| 2 | 60 | 15 |
| 3 | 4 | HOLD |
| Number | Degree | Minute |
| 1 | 37 | 120 |
| 2 | 70 | 20 |
| 3 | 4 | HOLD |
[[image:|thumb|digestion result]]
Ethanol precipitation of RBS-phaB-RBS-eYFP-dT and RBS-phaC-RBS-phaA-pSB1A2
- Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol.
- Centrifuged in 15000 rpm, 15 min at 4C.
- Remove supernatant and added 220 ul of 70% ethanol.
- Centrifuged in 15000 rpm, 10 min at 4C.
- Remove supernatant and air drying in room temperature then added 5 ul of DW.
Ligation of RBS-phaB-RBS-eYFP-dT and RBS-phaC-RBS-phaA-pSB1A2
| Insert DNA (60~70 ng/ul) | 4 ul |
| Vector DNA (20~30 ng/ul) | 2 ul |
| Ligation Mighty Mix | 6 ul |
| Total | 12 ul |
Ligation reaction time was in detail below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
Transformation of RBS-phaC-RBS-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2
Transformation of JM109.
- Mixed 2 ul RBS-phaC-RBS-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2 ligation product to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Mixed 350 ul of LB.
- Prepared and Labeled two plastic plates with LB plate medium which contained appropriate antibiotics (LBA).
- Plated 300 ul of the culture onto first dish and spread.
- Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for 17 hours.
September 18th
Colony PCR of pBAD-RBS-eCFP-RBS-Ag43-dI-pSB1C3
We did colony PCR two times.
| DNA solution | 4 ul |
| Kapa-Taq(Taq polymerase) | 10 ul |
| Forward Primer(200bp down primer) | 0.8 ul |
| Reverse Primer(ag43-f4 primer) | 0.8 ul |
| DW | 4.4 ul |
| Total | 20 ul |
| Number | Degree | Second |
| 1 | 95 | 120 |
| 2 | 95 | 30 |
| 3 | 53.0 | 30 |
| 4 | 72 | 60 |
| 5 | 72 | 60 |
| 6 | 4 | HOLD |
Cycle:2~4 x 35
We used N1 (DW only) and N2(pBAD-RBS-Ag43-dT-pSB1A2)as controls.
Desired product is about 702bp.
Target products didn't exist in all samples. We noticed the reason why such results shown is that we used incorrect pSB1C3 DNA solution which isn't confirmed the sequence. We'll try the synthesis once again in today.
Digestion of pBAD-RBS-eCFP-RBS-Ag43-dT-pSTV28 and pSB1C3
To make a construct of pBAD-RBS-eCFP-RBS-Ag43-dT-pSB1C3, we digested pBAD-RBS-eCFP-RBS-Ag43-dT-pSTV28 with EcoRI & SpeI and pSB1C3 with EcoRI & SpeI. Insert (pBAD-RBS-eCFP-RBS-Ag43-dT-pSTV28)
| DNA solution (about 35ng/ul) | 41 ul |
| EcoRI | 2 ul |
| SpeI | 2 ul |
| 10xH buffer | 5 ul |
| Total | 50 ul |
Vector(pSB1C3)
| DNA solution (about 30~40 ng/ul) | 2 ul |
| EcoRI | 1 ul |
| SpeI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 14 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 120 |
| 2 | 60 | 15 |
| 3 | 4 | HOLD |
[[image:|thumb|digestion result]]
Ethanol precipitation of pBAD-RBS-eCFP-RBS-Ag43-dT--pSB1C3
- Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol.
- Centrifuged in 15000 rpm, 15 min at 4C.
- Remove supernatant and added 220 ul of 70% ethanol.
- Centrifuged in 15000 rpm, 10 min at 4C.
- Remove supernatant and air drying in room temperature then added 5 ul of DW.
Ligation of pBAD-RBS-eCFP-RBS-Ag43-dT and pSB1C3
| Insert DNA | 4 ul |
| Vector DNA | 2 ul |
| Ligation Mighty Mix | 6 ul |
| Total | 12 ul |
Ligation reaction time was in detail below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
Transformation of pBAD-RBS-eCFP-RBS-Ag43-dT-pSB1C3
Transformation of JM109.
- Mixed 2 ul pBAD-RBS-eCFP-RBS-Ag43-dT--pSB1C3 ligation product to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Mixed 350 ul of LB.
- Incubated for 2 hrs to get the resistance to Chloramphenicol.
- Prepared and Labeled two plastic plates with LB plate medium which contained appropriate antibiotics (LBC).
- Plated 300 ul of the culture onto first dish and spread.
- Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for 15 hours.
September 19th
Colony PCR of pBAD-RBS-eCFP-RBS-Ag43-dI-pSTV28
Colony PCR to confirm whether pSTV28 vettor has pBAS-RBs-eCFP-RBs-Ag43-dT as insert or not.
| DNA solution | 4 ul |
| Kapa-Taq(Taq polymerase) | 10 ul |
| Forward Primer(200bp down primer) | 0.8 ul |
| Reverse Primer(ag43-f4 primer) | 0.8 ul |
| DW | 4.4 ul |
| Total | 20 ul |
| Number | Degree | Second |
| 1 | 95 | 120 |
| 2 | 95 | 30 |
| 3 | 53.3 | 30 |
| 4 | 72 | 180 |
| 5 | 72 | 60 |
| 6 | 4 | HOLD |
Cycle:2~4 x 35
We used N1 (DW only) and N2(pBAD-RBS-Ag43-dT-pSB1A2)as controls.
Desired product is about 702bp.
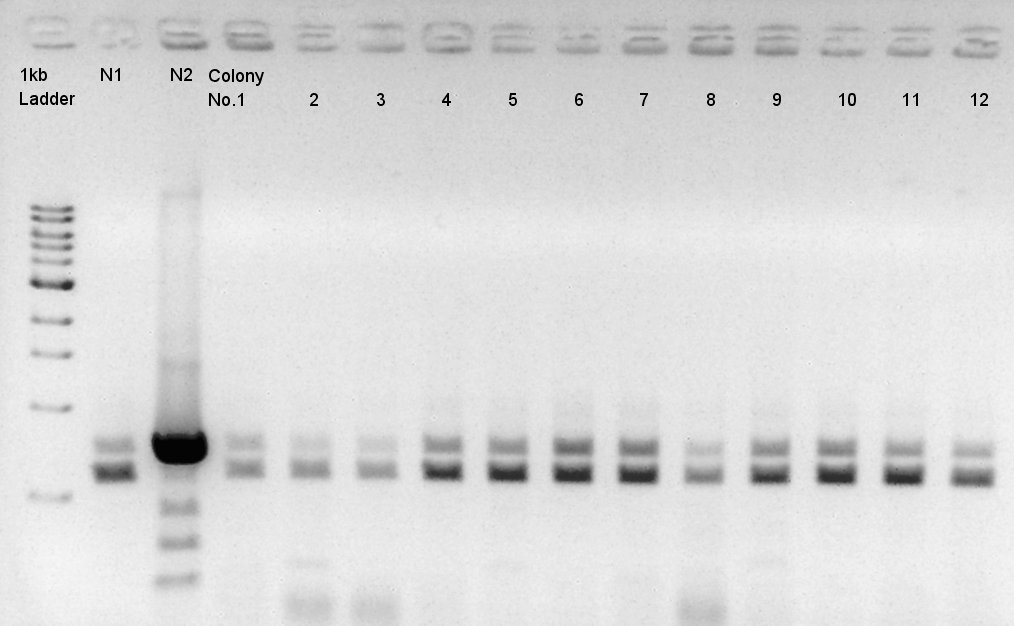
[[image:|thumb|Colony PCR result]]
We noticed the some of colones have pBAD-RBS-eCFP-RBS-Ag43-dT-pSTV28 construct as plasmid DNA.
Colony PCR of pBAD-RBS-eCFP-RBS-Ag43-dI-pSB1C3 and pBAD-RBS-eCFP-RBS-Ag43-dI-pSB1C3
Colony PCR to confirm whether the constructs written above really have eCFP and Ag43 coding sites or not by using primers: one can aneal to a part of eCFP coding site and another can aneal to a part of Ag43 coding site.
| DNA solution | 4 ul |
| Kapa-Taq(Taq polymerase) | 10 ul |
| Forward Primer(FP-F primer) | 0.8 ul |
| Reverse Primer(ag43-R primer) | 0.8 ul |
| DW | 4.4 ul |
| Total | 20 ul |
| Number | Degree | Second |
| 1 | 95 | 120 |
| 2 | 95 | 30 |
| 3 | 53.3 | 30 |
| 4 | 72 | 180 |
| 5 | 72 | 60 |
| 6 | 4 | HOLD |
Cycle:2~4 x 35
We used N1, N2 (DW only) as controls.
Desired product is about 452 bp and 697 bp respectively.
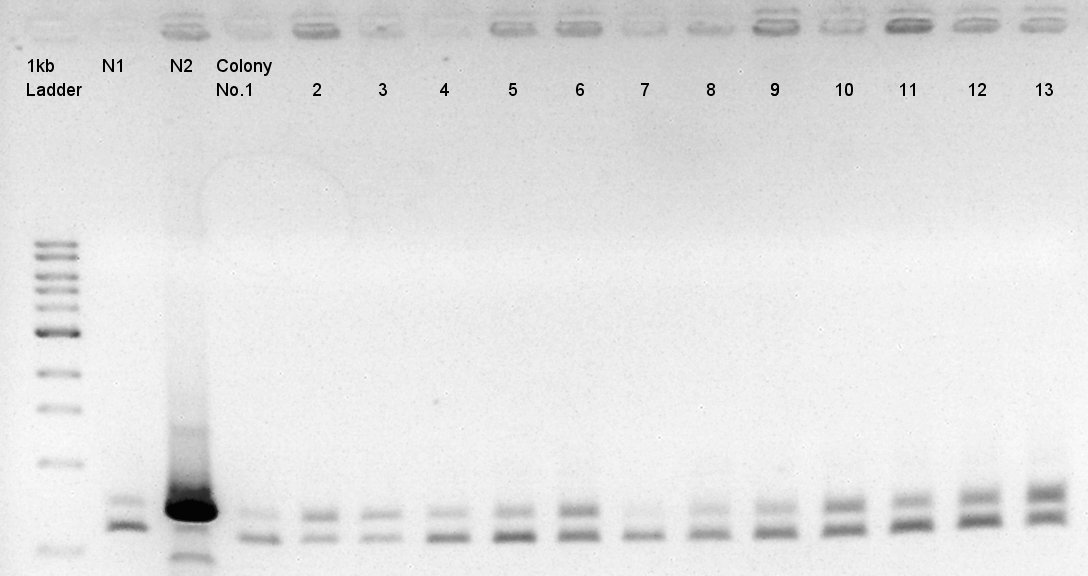
[[image:|thumb|Colony PCR result]]
We noticed the some of colones have pBAD-RBS-eCFP-RBS-Ag43-dT-pSTV28 construct as plasmid DNA.
September 20th
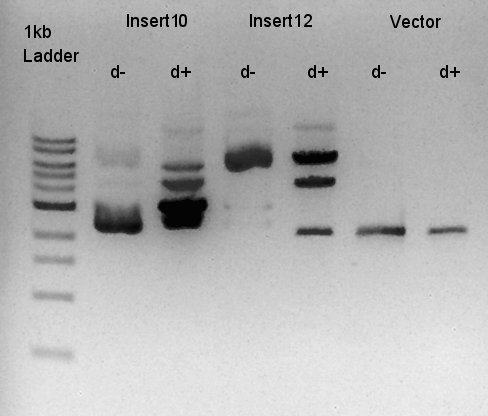
Digestion of ptet-RBS-phaC-RBs-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2 (DNA solution eluted from colony No.10, 12 respectively in ) and ptet-pSB1A2
To make a construct of ptet-RBS-phaC-RBS-phaA-RBS-phaB-RBS-dT-pSB1A2, we digested RBS-phaC-RBs-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2 with XbaI & PstI, ptet-pSB1A2 with SpeI & PstI. We prepared DNa solution derived from No.10 and No.12 colony selected selected by the result of colony PCR in 18th.
Insert No.10 (RBS-phaC-RBs-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2)
| DNA solution ( 40 ng/ul) | 20 ul |
| EcoRI | 1 ul |
| PstI | 1 ul |
| 10xH buffer | 4 ul |
| DW | 4 ul |
| Total | 30 ul |
Insert No.12 (RBS-phaC-RBs-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2)
| DNA solution ( 40 ng/ul) | 20 ul |
| EcoRI | 1 ul |
| PstI | 1 ul |
| 10xH buffer | 4 ul |
| DW | 4 ul |
| Total | 30 ul |
Vector(ptet-pSB1A2)
| DNA solution (about 40 ng/ul) | 2 ul |
| EcoRI | 1 ul |
| PstI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 14 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 120 |
| 2 | 60 | 15 |
| 3 | 4 | HOLD |
Ethanol precipitation of RBS-phaB-RBS-eYFP-dT and RBS-phaC-RBS-phaA-pSB1A2
- Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol.
- Centrifuged in 15000 rpm, 15 min at 4C.
- Remove supernatant and added 220 ul of 70% ethanol.
- Centrifuged in 15000 rpm, 10 min at 4C.
- Remove supernatant and air drying in room temperature then added 5 ul of DW.
Ligation of RBS-phaB-RBS-eYFP-dT and RBS-phaC-RBS-phaA-pSB1A2
| Insert DNA (50 ng/ul) | 4 ul |
| Vector DNA (20~30 ng/ul) | 1.5 ul |
| DW | 0.5 ul |
| Ligation Mighty Mix | 6 ul |
| Total | 12 ul |
Ligation reaction time was in detail below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
Transformation of RBS-phaC-RBS-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2
Transformation of JM109.
- Mixed 2 ul RBS-phaC-RBS-phaA-RBS-phaB-RBS-eYFP-dT-pSB1A2 ligation product to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Mixed 350 ul of LB.
- Incubated for 2 hrs to get the resistance to Chloramphenicol.
- Prepared and Labeled two plastic plates with LB plate medium which contained appropriate antibiotics (LBC).
- Plated 300 ul of the culture onto first dish and spread.
- Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for 15 hours.
 "
"