Team:Bielefeld-Germany/Project/Appoach
From 2012.igem.org
Contents |
The conventional methods of sewage treatment plants in order to take care of waste water are insufficient. This appears to be the case because the most common micro contaminants like synthetic Estrogen, Bisphenol A, Dicolfenac etc. are very difficult to break down.
The goal of Bielefeld’s iGEM team is to develope a biological filter-system using immobilized laccases for the purification of municipal and industrial waste water to get rid of these synthetic estrogens and other aromatic compounds. Laccases are copper-containing oxidase enzymes found in many organisms. One of their properties is the ability to degrade a wide range of aromatic and phenolic compounds. For this purpose, genes of various bacterial and eukaryotic laccases were isolated and expressed in Escherichia coli and Pichia pastoris. The choice of the expression system was meanwhile on the enzyme`s glycosylation status.
Isolation of laccase genes and generation of new BioBricks
The first step of our project was to isolate the specific laccase gene sequences and to generate new BioBricks for the iGEM competition. The laccases of the following organisms have been isolated:
Bacterial laccases:- Escherichia coli
- Bacillus halodurans
- Bacillus pumilus
- Streptomyces griseus
- Streptomyces lavendulae
- Thermus thermophilus
- Xanthomonas campestris
Eukaryotic laccases:
- Arabidopsis thaliana
- Pycnoporus cinnabarinus
- Trametes versicolor
- Trametes villosa
For more information about the organisms [|click here].
The additional and essential BioBricks and characteristics, which were used to design our functional plasmids and a new shuttle-vector-system are shown in the following table:
Parts of the E. coli expression vector and the P. pastoris shuttle vector
| Plasmid characteristics | Shuttle vector characteristics |
|---|---|
| T7 promoter region | AOX1 promoter |
| His-Tag-sequence | Mating factor alpha 1 |
| Chloramphenicol resistance | Histidine auxotrophic complementation |
The choice for an expression system was determined by the folding and glycosylation of the individual laccase. The resulting genetically modified organisms produced the laccases for our approach. For the characterization of the different laccases these enzymes have been isolated and purified.
Determining activity and potential of the different laccases
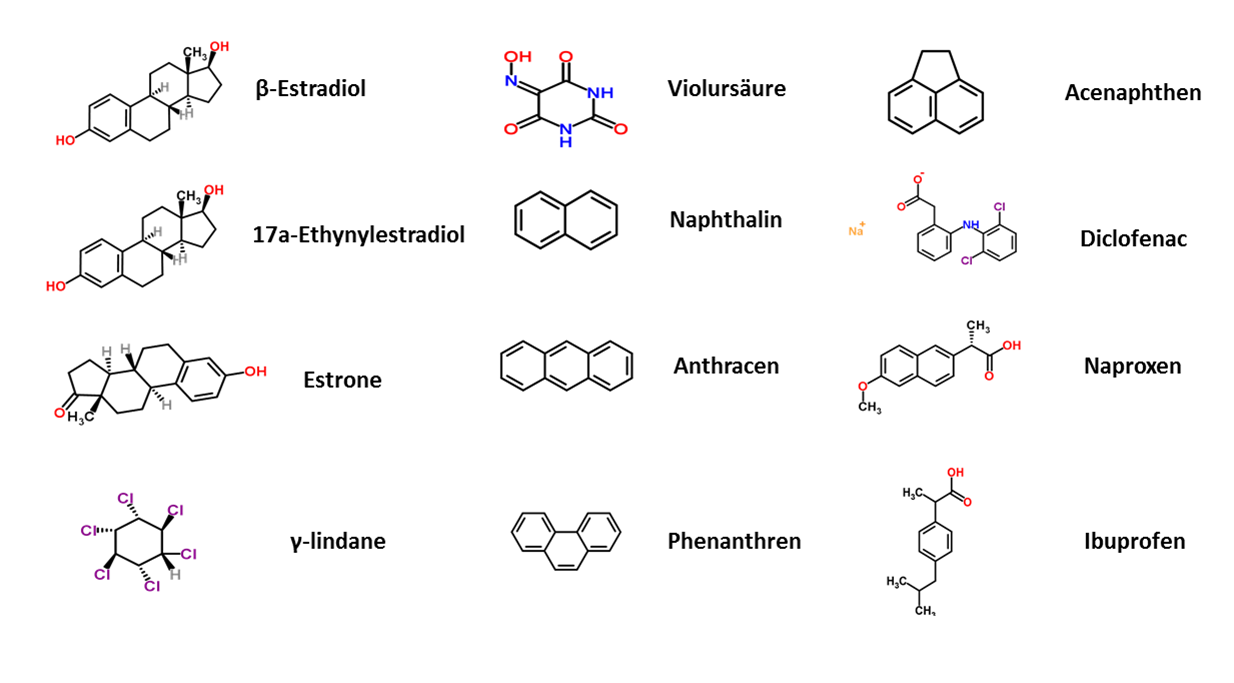
| The next step to generate a efficient filter system was to characterize the purified laccases. To identify the potential of a laccase, the enzyme was examined on its activity and potential to degrade several substances. The degradation potential of the manufactured laccases was determined representatively for substances that originate from different chemical areas, such as analgetics, endocrine substances, pesticides, polycyclic aromatic hydrocarbons and bleaching agents.
In our case we analyzed the specific laccases for their degradation efficiency while varying different parameters like temperature, pH value and buffer system. A great concern of our team is to guarantee https://2012.igem.org/Team:Bielefeld-Germany/Safety the safety] of the generated filter system. Besides the degradation potential, a very important aspect to us is the analysis of the degraded substances, to ensure the harmlessness of the degradation result for the individual laccase. This was investigated by HPLC-masspectroscopy (LC-ESI-qTOF-MS). With the help of this knowledge we wanted to identify the laccase which shows the highest potential for a safe and highly functional filter system. |
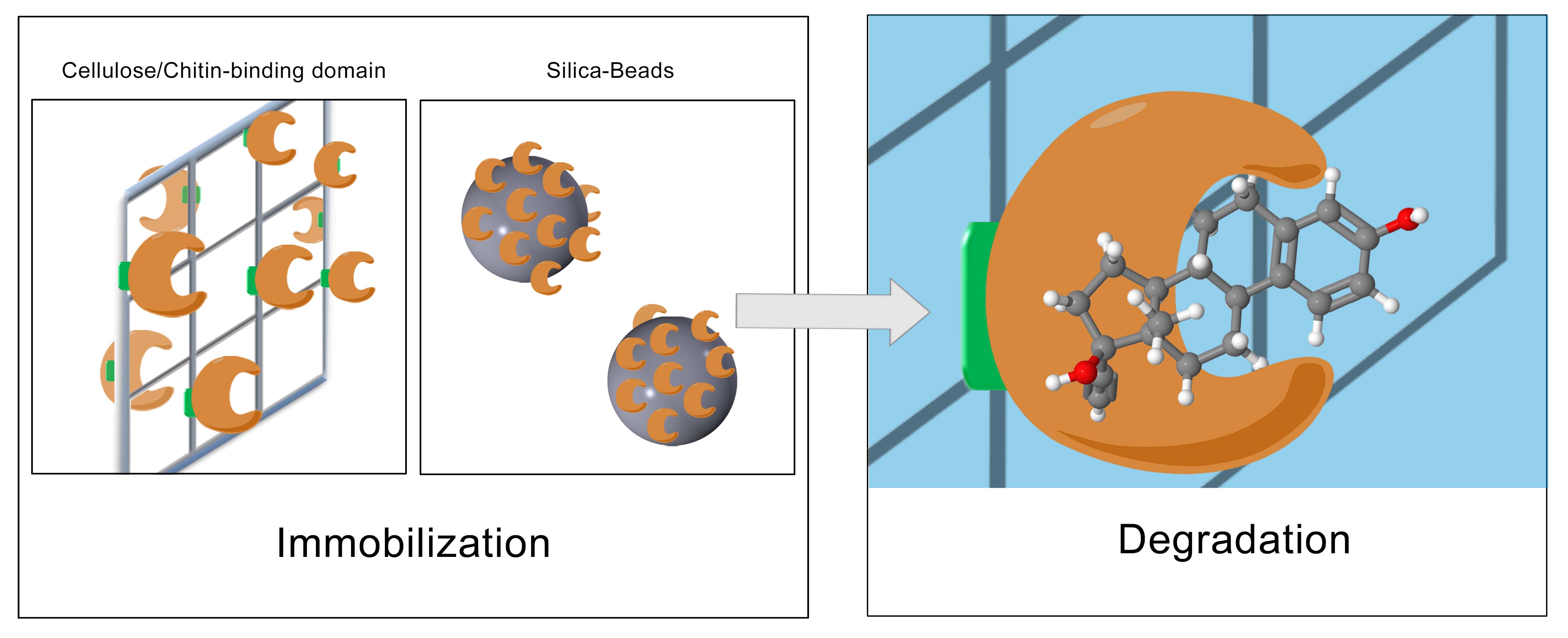
Immobilization and the final development of the filter
The last step to realize our project is to immobilize the produced lacasses with the highest potentials. To generate a filter-system with a high yield of active and immobilized enzymes, different approaches are tested. On the one hand we try to immobilize the enzymes with a chemical immobilization protocol to bind the laccases covalently on special silica-beads (CPC silica beads), and on the other hand we try to generate a natural immobilization protocol by fusing the laccases to natural binding domains, such as the cellulose-binding-domain, the keratin-binding-domain or the chitin-binding-domain. By generating the new natural immobilization protocol three different cellulose-binding-domains of the organisms Cellulomonas fimi, Clostridium josui and Clostridium cellulovorans are investigated. At first the binding-domains are linked to GFP (Green fluorescent Protein) to examine the strength of the binding and the binding-capacity. In the end the domains will be fused to the lacasses.
The goal of the iGEM-Team Bielefeld is to generate a functional filter system with a high efficiency to degrade a high number of different micro contaminants. This filter system will be able to reduce the environmental pollution and improve the quality of the waterbody for the safety of animals and mankind.
| 55px | | | | | | | | | | |
 "
"