Team:HokkaidoU Japan/Notebook/aggregation Week 9
From 2012.igem.org
| Line 16: | Line 16: | ||
==Plasmid extraction== | ==Plasmid extraction== | ||
<p> | <p> | ||
| - | + | We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions. | |
</p> | </p> | ||
[[image:HokkaidoU2012_120827_take-iroiro.jpg|thumb|Mini-prep resulsts]] | [[image:HokkaidoU2012_120827_take-iroiro.jpg|thumb|Mini-prep resulsts]] | ||
| Line 113: | Line 113: | ||
{|class="hokkaidou-table-pcr-reagent" | {|class="hokkaidou-table-pcr-reagent" | ||
|Solution | |Solution | ||
| - | |Volume(ul) | + | |Volume (ul) |
|- | |- | ||
|DNA | |DNA | ||
| Line 236: | Line 236: | ||
| - | == | + | ==Plasmid extraction of pT7-RBS-Ag43-dT on pSB1C3== |
<p> | <p> | ||
Mini-prep of pT7-RBS-Ag43-dT on pSB1C3. We re-suspended No.1,2 colonies and incubated. | Mini-prep of pT7-RBS-Ag43-dT on pSB1C3. We re-suspended No.1,2 colonies and incubated. | ||
| Line 248: | Line 248: | ||
{| | {| | ||
|Solution | |Solution | ||
| - | |Value(ul) | + | |Value (ul) |
|- | |- | ||
|1kb ladder | |1kb ladder | ||
| Line 265: | Line 265: | ||
| - | From this result, We estimated that the concentration of Insert DNA solution is about | + | From this result, We estimated that the concentration of Insert DNA solution is about 40 ng/ul, and Vector DNA is also about 40 ng/ul. |
</p> | </p> | ||
| Line 278: | Line 278: | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| - | |DNA solution ( | + | |DNA solution (40 ng/ul) |
|5 ul | |5 ul | ||
|- | |- | ||
| Line 303: | Line 303: | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| - | |DNA solution ( | + | |DNA solution (40 ng/ul) |
|14 ul | |14 ul | ||
|- | |- | ||
| Line 403: | Line 403: | ||
==Transformation== | ==Transformation== | ||
<p> | <p> | ||
| - | Transformation for ligation product (ptet-RBs-eYFP-dT on pSB1A2) | + | Transformation for ligation product (ptet-RBs-eYFP-dT on pSB1A2) in DH5α. |
#Mixed 1 ul of DNA to 50 ul of thawed competent cells on ice. | #Mixed 1 ul of DNA to 50 ul of thawed competent cells on ice. | ||
#Incubated on ice for 30 min. | #Incubated on ice for 30 min. | ||
| Line 410: | Line 410: | ||
#Plated 300 ul of the culture onto first dish and spread. | #Plated 300 ul of the culture onto first dish and spread. | ||
#Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread. | #Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread. | ||
| - | #Incubated the plates at 37C for 15 | + | #Incubated the plates at 37C for 15 hrs. |
</p> | </p> | ||
| Line 427: | Line 427: | ||
| - | + | There were no colony on the LBC plate that was spread solution mixed digestion product. | |
</p> | </p> | ||
</div></div> | </div></div> | ||
| Line 439: | Line 439: | ||
We incubated 9 colonies transformed at August 25th in LBAK solution including 1% L-arabinose for 16 hrs. | We incubated 9 colonies transformed at August 25th in LBAK solution including 1% L-arabinose for 16 hrs. | ||
Then we find that 1 culture expressing Ag43. | Then we find that 1 culture expressing Ag43. | ||
| - | Observed the E. coli cluster by a microscope of 100 magnifications | + | Observed the E. coli cluster by a microscope of 100 magnifications. |
| Line 445: | Line 445: | ||
#1 colony ( could expressing Ag43 ) incubated in 2 ml LB at 37C for 2 hrs. | #1 colony ( could expressing Ag43 ) incubated in 2 ml LB at 37C for 2 hrs. | ||
#Prepared 5 kinds of LB medium differing the concentration of L-arabinose. | #Prepared 5 kinds of LB medium differing the concentration of L-arabinose. | ||
| - | No. 1 :0.001% | + | <br />No. 1 :0.001% |
| - | No. 2 :0.1% | + | <br />No. 2 :0.1% |
| - | No. 3 :0.4% | + | <br />No. 3 :0.4% |
| - | No. 4 :1.0% | + | <br />No. 4 :1.0% |
| - | No. 5 :0% | + | <br />No. 5 :0%<br /> |
#Added 400 ul of culture in several LB medium. | #Added 400 ul of culture in several LB medium. | ||
#Incubated for 17 hrs and 30 min at 37C. | #Incubated for 17 hrs and 30 min at 37C. | ||
| Line 462: | Line 462: | ||
{|class="hokkaidou-table-pcr-reagent" | {|class="hokkaidou-table-pcr-reagent" | ||
|Solution | |Solution | ||
| - | |Volume(ul) | + | |Volume (ul) |
|- | |- | ||
|DNA | |DNA | ||
| Line 523: | Line 523: | ||
==Colony PCR== | ==Colony PCR== | ||
<p> | <p> | ||
| - | Colony PCR of pT7-RBS-Ag43-dT on pSB1C3 transformed at 28th. | + | Colony PCR of pT7-RBS-Ag43-dT on pSB1C3 transformed at August 28th. |
| Line 592: | Line 592: | ||
#Prepared 2 ml LBC (LBA) into culture tubes. | #Prepared 2 ml LBC (LBA) into culture tubes. | ||
#Re-suspended 2 colonies No.7 and No.8 respectively. Ag43-dT on pSB1AK3 was re-suspended the N2 colony. | #Re-suspended 2 colonies No.7 and No.8 respectively. Ag43-dT on pSB1AK3 was re-suspended the N2 colony. | ||
| - | #Incubated at 37C for | + | #Incubated at 37C for 17 hrs. |
</p> | </p> | ||
| Line 600: | Line 600: | ||
{| | {| | ||
|Solution | |Solution | ||
| - | |Value(ul) | + | |Value (ul) |
|- | |- | ||
|1kb ladder | |1kb ladder | ||
| Line 617: | Line 617: | ||
| - | From this result, We estimated that the concentration of Insert DNA solution is about | + | From this result, We estimated that the concentration of Insert DNA solution is about 36 ng/ul, and Vector DNA is also about 29 ng/ul. |
</p> | </p> | ||
| Line 630: | Line 630: | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| - | |DNA solution ( | + | |DNA solution (36 ng/ul) |
|5 ul | |5 ul | ||
|- | |- | ||
| Line 655: | Line 655: | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| - | |DNA solution ( | + | |DNA solution (29 ng/ul) |
|6 ul | |6 ul | ||
|- | |- | ||
| Line 688: | Line 688: | ||
|- | |- | ||
|100x BSA | |100x BSA | ||
| - | |0.2 | + | |0.2 ul |
|- | |- | ||
|DW | |DW | ||
| Line 721: | Line 721: | ||
[[image:HokkaidoU2012 120829 Digestion eCFP RBSonpSB1A2 XbaIcontrol.jpg|thumb|digestion result]] | [[image:HokkaidoU2012 120829 Digestion eCFP RBSonpSB1A2 XbaIcontrol.jpg|thumb|digestion result]] | ||
| - | From this result, Vector DNA were digested | + | From this result, Vector DNA were digested by XbaI, but remains some amount of non-digested DNA. |
| - | And | + | And insert DNA solution were not existed in both d- and d+ lines.It means we failed to extract from gel after electrophoresis. We decided to retry PCR. |
</p> | </p> | ||
| Line 732: | Line 732: | ||
{|class="hokkaidou-table-pcr-reagent" | {|class="hokkaidou-table-pcr-reagent" | ||
|Solution | |Solution | ||
| - | |Volume(ul) | + | |Volume (ul) |
|- | |- | ||
|DNA | |DNA | ||
| Line 799: | Line 799: | ||
==Estimation of concentration of eCFP, pT7-RBS-Ag43-dT-pSB1C3 and Ag43-dT-pSB1AK3== | ==Estimation of concentration of eCFP, pT7-RBS-Ag43-dT-pSB1C3 and Ag43-dT-pSB1AK3== | ||
<p> | <p> | ||
| - | No.7,8 means the colony number of colony PCR of pT7-RBS-Ag43-dT-pSB1C3. | + | No. 7,8 means the colony number of colony PCR of pT7-RBS-Ag43-dT-pSB1C3. |
| Line 806: | Line 806: | ||
We failed the PCR for E0020. | We failed the PCR for E0020. | ||
| - | We estimated the concentration of pT7-RBS-Ag43-dT-pSB1C3 is 31 ng/ul and Ag43-dT-pSB1AK3 is | + | We estimated the concentration of pT7-RBS-Ag43-dT-pSB1C3 is 31 ng/ul and Ag43-dT-pSB1AK3 is 35 ng/ul. |
</p> | </p> | ||
| Line 816: | Line 816: | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| - | |DNA solution ( | + | |DNA solution (31 ng/ul) |
|1 ul | |1 ul | ||
|- | |- | ||
| Line 838: | Line 838: | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| - | |DNA solution ( | + | |DNA solution (35 ng/ul) |
|14 ul | |14 ul | ||
|- | |- | ||
| Line 885: | Line 885: | ||
| - | We did not | + | We did not do digestion of vector DNA sokution, result, because it has only too low concentration to be digested product solution. |
We failed PCR again. | We failed PCR again. | ||
| - | The digestion result was confirmed that the Insert DNA was successfully digested | + | The digestion result was confirmed that the Insert DNA was successfully digested by XbaI and SpeI. |
And we extracted the insert DNA digestion product from agarose gel by gel-extraction kit and got 20 ul of Insert DNA solution. | And we extracted the insert DNA digestion product from agarose gel by gel-extraction kit and got 20 ul of Insert DNA solution. | ||
</p> | </p> | ||
| Line 900: | Line 900: | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| - | |DNA solution ( | + | |DNA solution (35 ng/ul) |
|20 ul | |20 ul | ||
|- | |- | ||
| Line 951: | Line 951: | ||
==Ethanol precipitation of pT7-RBS-Ag43-dT-pSB1C3 and Ag43-dT== | ==Ethanol precipitation of pT7-RBS-Ag43-dT-pSB1C3 and Ag43-dT== | ||
<p> | <p> | ||
| - | We did ethanol precipitation to get more high concentration | + | We did ethanol precipitation to get more high concentration DNA solution of each part. And we got 5ul of each DNA solution. |
[[image:HokkaidoU2012 120831 Insert-d--d+ Vector-d+.jpg|thumb|ethanol precipitation result]] | [[image:HokkaidoU2012 120831 Insert-d--d+ Vector-d+.jpg|thumb|ethanol precipitation result]] | ||
| - | The electrophoresis result showed that the pT7-RBS-pSB1C3 construct was successfully digested | + | The electrophoresis result showed that the pT7-RBS-pSB1C3 construct was successfully digested by SpeI and get enough concentration of DNA solution to ligate. |
</p> | </p> | ||
| Line 1,013: | Line 1,013: | ||
<div class="hokkaidou-notebook-daily"> | <div class="hokkaidou-notebook-daily"> | ||
| - | ==August | + | ==August 31st== |
<div> | <div> | ||
| - | == | + | ==Plasmid extraction for pBAD-RBS-Ag43-dT on pSB1AK3== |
<p> | <p> | ||
We used promega SV miniprep kit. | We used promega SV miniprep kit. | ||
| Line 1,157: | Line 1,157: | ||
#Prepared 2 ml LBA into culture tubes. | #Prepared 2 ml LBA into culture tubes. | ||
#Re-suspended 1 colony. | #Re-suspended 1 colony. | ||
| - | #Incubated at 37C for | + | #Incubated at 37C for 16 hrs. |
</p> | </p> | ||
| Line 1,234: | Line 1,234: | ||
==Ethanol precipitation== | ==Ethanol precipitation== | ||
<p> | <p> | ||
| - | Ethanol precipitation to get more high concentration of pBAD-RBS-Ag43-dT solution and pSB1C3 solution digested | + | Ethanol precipitation to get more high concentration of pBAD-RBS-Ag43-dT solution and pSB1C3 solution digested by EcoRI & SpeI. |
#Added 2 ul of NaoAc, 1.5 ul of glycogen and 50 ul of 100% ethanol. | #Added 2 ul of NaoAc, 1.5 ul of glycogen and 50 ul of 100% ethanol. | ||
#Centrifuged at 15000 rpm, 10 min at 4C. | #Centrifuged at 15000 rpm, 10 min at 4C. | ||
| Line 1,295: | Line 1,295: | ||
</p> | </p> | ||
| - | == | + | ==Plasmid extraction of Ag43-dT-pSB1AK3 and RBS-pSB1A2== |
<p> | <p> | ||
We used mini-prep kit of Nippon genetics and got 50 ul Ag43-dT-pSB1AK3 plasmid DNA solution. | We used mini-prep kit of Nippon genetics and got 50 ul Ag43-dT-pSB1AK3 plasmid DNA solution. | ||
| Line 1,302: | Line 1,302: | ||
==Digestion of eCFP-pSB1A2 and RBS-pSB1A2== | ==Digestion of eCFP-pSB1A2 and RBS-pSB1A2== | ||
<p> | <p> | ||
| - | To make a construct of eCFP-RBS-pSB1A2, we digested eCFP-pSB1A2 | + | To make a construct of eCFP-RBS-pSB1A2, we digested eCFP-pSB1A2 by EcoRI and SpeI, and RBS-pSB1A2 with EcoRI and XbaI. And we digested pT7-RBS-pSB1C3 by XbaI as a control for confirmation of the ability to digest. |
Insert (eCFP-pSB1A2) | Insert (eCFP-pSB1A2) | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| - | |DNA solution ( | + | |DNA solution (35 ng/ul) |
|11 ul | |11 ul | ||
|- | |- | ||
| Line 1,332: | Line 1,332: | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| - | |DNA solution ( | + | |DNA solution (29 ng/ul) |
|6 ul | |6 ul | ||
|- | |- | ||
| Line 1,365: | Line 1,365: | ||
|- | |- | ||
|100x BSA | |100x BSA | ||
| - | |0.2 | + | |0.2 ul |
|- | |- | ||
|DW | |DW | ||
| Line 1,436: | Line 1,436: | ||
|1 ul | |1 ul | ||
|- | |- | ||
| - | |Insert DNA (20 ng/ul | + | |Insert DNA (20 ng/ul) |
|4 ul | |4 ul | ||
|- | |- | ||
Revision as of 14:48, 26 September 2012
August 27th
Aggregation check
We found a lot of colonies on LBA plate, spreaded at August 26th. We incubated 3 colonies in LBAK solution including 1% L-arabinose for 18 hrs. However, we could not find expression of Ag43 protein.
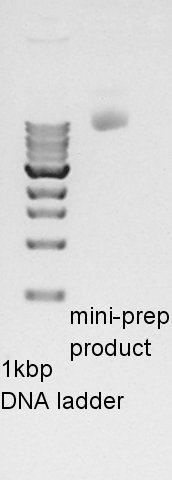
Plasmid extraction
We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions.
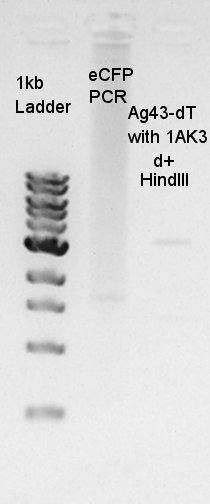
Colony PCR
Colony PCR to confirm that whether the pT7-RBS-Ag43-dT on pSB1C3 was successfully ligated or not.
| DNA solution | 4 ul |
| Kapa-Taq(Taq polymerase) | 5 ul |
| Forward Primer(ag43-f4 primer) | 0.5 ul |
| Reverse Primer(200bp down primer) | 0.5 ul |
| Total | 10 ul |
| Number | Degree | Second |
| 1 | 95 | 120 |
| 2 | 95 | 30 |
| 3 | 53.0 | 30 |
| 4 | 72 | 60 |
| 5 | 72 | 60 |
| 6 | 4 | HOLD |
Cycle:2~4 x 35
We used N1 (DW only) and N2 (Ag43-dT on pSB1AK3) as controls. Desired product is about 695bp.
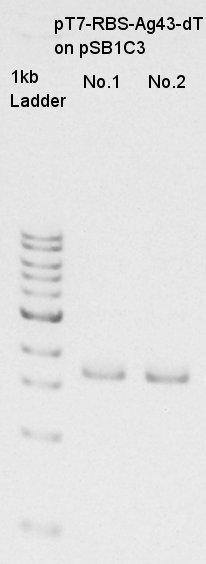
The result did not show the band clearly. We selected No.1 and 2 colony for incubation.
Incubation for mini-prep of pT7-RBS-Ag43-dT on pSB1C3
Incubation of pT7-RBS-Ag43-dT on pSB1C3 in LBC liquid medium.
- Prepared 2 ml LBC into culture tubes.
- Re-suspended 2 colonies (No.1 and No.2 respectively).
- Incubated at 37C for 15 hrs.
Transformation of ptet-RBS(B0034)-CFP-dT on pSB1A2
- Mixed 1 ul DNA to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Mixed 350 ul of LB.
- Prepared and Labeled two plastic plates with LBC.
- Plated 300 ul of the culture onto first dish and spread.
- Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for 15 hours.
PCR of RBS-YFP-dT
Amplified the construct with 100bp-up-EX primer and 200bp-down-PS primer. Mixed PCR solutions.
| Solution | Volume (ul) |
| DNA | 1 |
| 100bp-up-EX | 1 |
| 200bp-down-PS | 1 |
| MgSO4 | 3 |
| dNTP | 5 |
| 10x KOD-Plus-Neo Buffer | 5 |
| KOD-Plus-Neo | 1 |
| DW | 33 |
| Total | 50 |
| Number | Degree | Second |
| 1 | 94 | 120 |
| 2 | 98 | 10 |
| 3 | 58.2 | 30 |
| 4 | 68 | 60 |
| 5 | 4 | HOLD |
Cycle:2~4 x 35
August 28th
Digestion
We digested mini-prep products (pBAD-RBS-Ag43-dT on pSB1AK3) by EcoRI and SpeI. Because I'd like to check the size of insert and vector of getting plasmid.
| DNA solution | 5 ul |
| EcoRI | 1 ul |
| SpeI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 11 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 180 |
| 2 | 60 | 15 |
| 3 | 4 | HOLD |
Gel extraction
Gel extraction for digestion product. We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50 ul of DNA solution.
Plasmid extraction of pT7-RBS-Ag43-dT on pSB1C3
Mini-prep of pT7-RBS-Ag43-dT on pSB1C3. We re-suspended No.1,2 colonies and incubated.
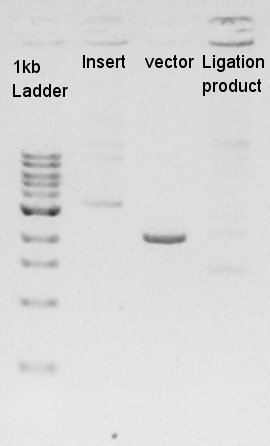
Estimation of Concentration of RBS-eYFP-dT and ptetR-pSB1A2
| Solution | Value (ul) |
| 1kb ladder | 1 |
| Insert | 1 |
| Vector | 1 |
From this result, We estimated that the concentration of Insert DNA solution is about 40 ng/ul, and Vector DNA is also about 40 ng/ul.
Digestion of ptet-pSB1A2 and RBS-Ag43-dT
ptet-pSB1A2(Vector) was digested with SpeI and PstI, and RBS-Ag43-dT(Insert) was cut with XbaI and PstI. Vector
| DNA solution (40 ng/ul) | 5 ul |
| SpeI | 0.5 ul |
| PstI | 0.5 ul |
| 10xH buffer | 2 ul |
| DW | 12 ul |
| Total | 20 ul |
Inset
| DNA solution (40 ng/ul) | 14 ul |
| XbaI | 1 ul |
| PstI | 1 ul |
| 10xM buffer | 2 ul |
| DW | 2 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 180 |
| 2 | 60 | 15 |
| 3 | 4 | HOLD |
From this result, we confirmed that Insert and Vector DNA were digested.
Ethanol Precipitation
Ethanol precipitation for ptet-pSB1A2 and RBS-eYFP-dT digestion products.
- Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol.
- Centrifuged at 15000 rpm, 15 min at 4C.
- Removed supernatant and added 220 ul of 70% ethanol.
- Centrifuged at 15000 rpm, 10 min at 4C.
- Removed supernatant and air drying at room temperature then added 10 ul of DW.
Ligation
Ligation for ptet-pSB1A2 and RBS-eYFP-dT. We used Ligation Mighty Mix (TAKARA BIO INC.) which contains ligase and buffer.
| Vector DNA | 3 ul |
| Insert DNA | 3 ul |
| Ligation Mighty Mix | 6 ul |
| Total | 12 ul |
Ligation reaction time was in detail below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
Transformation
Transformation for ligation product (ptet-RBs-eYFP-dT on pSB1A2) in DH5α.
- Mixed 1 ul of DNA to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Mixed 350 ul of LB.
- Prepared and Labeled two plastic plates with LBA.
- Plated 300 ul of the culture onto first dish and spread.
- Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for 15 hrs.
Gel extraction product check of pT7-RBS-Ag43-dT on pSB1C3
We couldn't get desired plasmid DNA by transformation. We doubted contamination of non-digested vector DNA and decided to test the gel extraction product of pT7-RBS on pSB1C3 was successfully separated from non-digested product or not by transformation.
- Mixed 1 ul of each DNA of ligation product and digestion product to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Mixed 350 ul of LB.
- Incubated for 2 hrs to get the resistance to Chloramphenicol.
- Prepared and Labeled two plastic plates with LBC.
- Plated 300 ul of the culture onto first dish and spread.
- Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for 16 hours.
August 29th
Aggregation check
We incubated 9 colonies transformed at August 25th in LBAK solution including 1% L-arabinose for 16 hrs. Then we find that 1 culture expressing Ag43. Observed the E. coli cluster by a microscope of 100 magnifications. Then we checked whether pBAD promoter is behaving exactly or not.
- 1 colony ( could expressing Ag43 ) incubated in 2 ml LB at 37C for 2 hrs.
- Prepared 5 kinds of LB medium differing the concentration of L-arabinose.
No. 1 :0.001%
No. 2 :0.1%
No. 3 :0.4%
No. 4 :1.0%
No. 5 :0%
- Added 400 ul of culture in several LB medium.
- Incubated for 17 hrs and 30 min at 37C.
PCR of eCFP(E0020)
Amplified the part with 100bp-up-EX primer and 200bp-down-PS primer. Desired product is about 800~900bp. Mixed PCR solutions.
| Solution | Volume (ul) |
| DNA | 1 |
| 100bp-up-EX | 1 |
| 200bp-down-PS | 1 |
| MgSO4 | 3 |
| dNTP | 5 |
| 10x KOD-Plus-Neo Buffer | 5 |
| KOD-Plus-Neo | 1 |
| DW | 33 |
| Total | 50 |
| Number | Degree | Second |
| 1 | 94 | 120 |
| 2 | 98 | 10 |
| 3 | 58.2 | 30 |
| 4 | 68 | 60 |
| 5 | 4 | HOLD |
Cycle:2~4 x 35
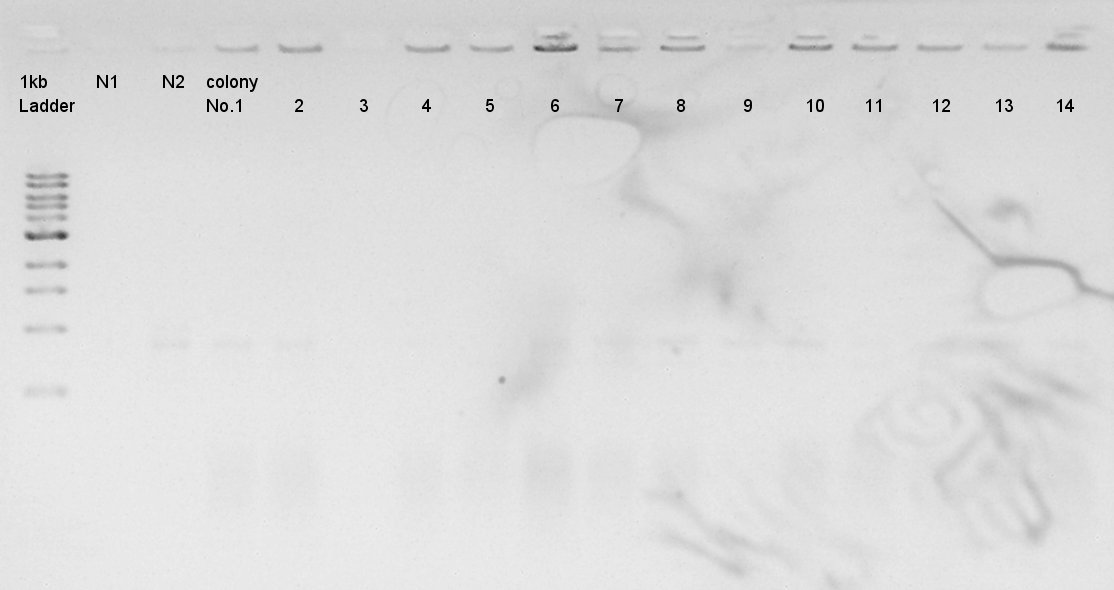
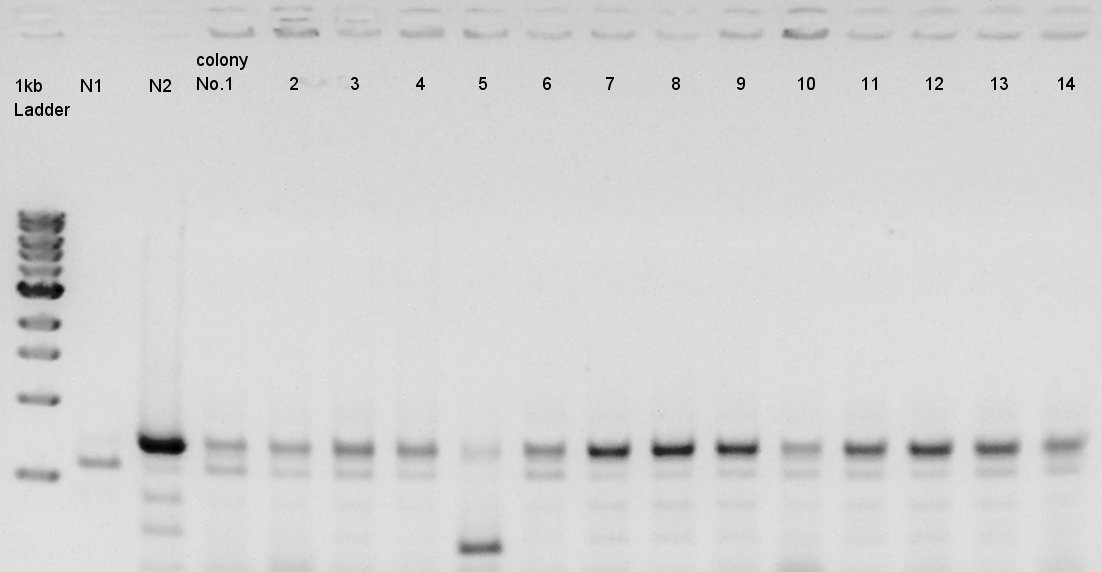
Colony PCR
Colony PCR of pT7-RBS-Ag43-dT on pSB1C3 transformed at August 28th.
| DNA solution | 4 ul |
| Kapa-Taq(Taq polymerase) | 5 ul |
| Forward Primer(ag43-f4 primer) | 0.5 ul |
| Reverse Primer(200bp down primer) | 0.5 ul |
| Total | 10 ul |
| Number | Degree | Second |
| 1 | 95 | 120 |
| 2 | 95 | 30 |
| 3 | 53.0 | 30 |
| 4 | 72 | 60 |
| 5 | 72 | 60 |
| 6 | 4 | HOLD |
Cycle:2~4 x 35
We used N1 (DW only) and N2 (Ag43-dT on pSB1AK3) as controls. Desired product is about 695bp. This length is almost same as N2.
We decided to incubate the No.7 and 8 colony.
Incubation for mini-prep of pT7-RBS-Ag43-dTonpSB1C3 and Ag43-dTonpSB1AK3
Incubation of pT7-RBS-Ag43-dTonpSB1C3 (and Ag43-dT on pSB1AK3) in LBC (LBA) liquid medium.
- Prepared 2 ml LBC (LBA) into culture tubes.
- Re-suspended 2 colonies No.7 and No.8 respectively. Ag43-dT on pSB1AK3 was re-suspended the N2 colony.
- Incubated at 37C for 17 hrs.
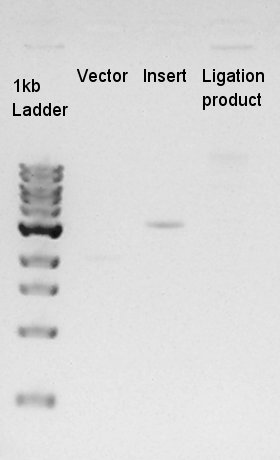
Estimation of Concentration of RBS-eYFP-dT (PCR product) and ptetR-pSB1A2
| Solution | Value (ul) |
| 1kb ladder | 1 |
| Insert | 1 |
| Vector | 1 |
From this result, We estimated that the concentration of Insert DNA solution is about 36 ng/ul, and Vector DNA is also about 29 ng/ul.
Digestion of ptet-pSB1A2 and RBS-Ag43-dT
ptet-pSB1A2(Vector) was digested with EcoRI and SpeI, and RBS-Ag43-dT(Insert) was cut with EcoRI and XbaI. And we used construct of pT7-RBS on pSB1C3 as control for the function of XbaI. Insert (eCFP)
| DNA solution (36 ng/ul) | 5 ul |
| EcoRI | 1 ul |
| SpeI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 5 ul |
| Total | 20 ul |
Vector(RBS on pSB1A2)
| DNA solution (29 ng/ul) | 6 ul |
| EcoRI | 1 ul |
| XbaI | 1 ul |
| 10xM buffer | 2 ul |
| DW | 10 ul |
| Total | 20 ul |
control (pT7-RBs on pSB1C3)
| DNA solution (30 ng/ul) | 6 ul |
| XbaI | 1 ul |
| 10xM buffer | 2 ul |
| 100x BSA | 0.2 ul |
| DW | 11 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 120 |
| 2 | 60 | 15 |
| 3 | 4 | HOLD |
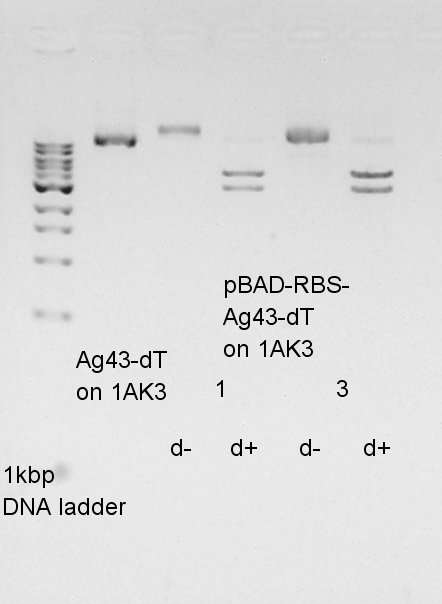
From this result, Vector DNA were digested by XbaI, but remains some amount of non-digested DNA. And insert DNA solution were not existed in both d- and d+ lines.It means we failed to extract from gel after electrophoresis. We decided to retry PCR.
PCR of eCFP(E0020)
Amplified the part with 100bp-up-EX primer and 200bp-down-PS primer. Desired product is about 800~900bp. Mixed PCR solutions.
| Solution | Volume (ul) |
| DNA | 1 |
| 100bp-up-EX | 1 |
| 200bp-down-PS | 1 |
| MgSO4 | 3 |
| dNTP | 5 |
| 10x KOD-Plus-Neo Buffer | 5 |
| KOD-Plus-Neo | 1 |
| DW | 33 |
| Total | 50 |
| Number | Degree | Second |
| 1 | 94 | 120 |
| 2 | 98 | 10 |
| 3 | 58.0 | 30 |
| 4 | 68 | 60 |
| 5 | 4 | HOLD |
Cycle:2~4 x 35
August 30th
Estimation of concentration of eCFP, pT7-RBS-Ag43-dT-pSB1C3 and Ag43-dT-pSB1AK3
No. 7,8 means the colony number of colony PCR of pT7-RBS-Ag43-dT-pSB1C3.
We failed the PCR for E0020.
We estimated the concentration of pT7-RBS-Ag43-dT-pSB1C3 is 31 ng/ul and Ag43-dT-pSB1AK3 is 35 ng/ul.
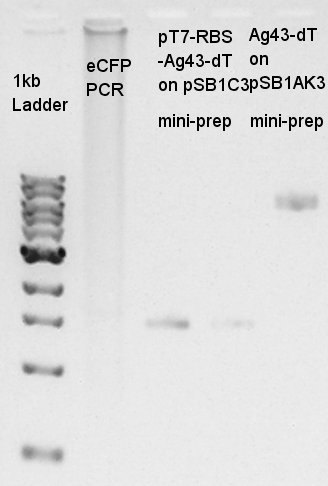
Digestion of pT7-RBS-pSB1C3 and Ag43-dT-pSB1AK3
Digestion of pT7-RBS-pSB1C3 (as Vector) and Ag43-dT-pSB1AK3 (as Insert) to make pT7-RBs-Ag43-dT-pSB1C3 construct. Vector
| DNA solution (31 ng/ul) | 1 ul |
| SpeI | 1 ul |
| 10xm buffer | 2 ul |
| DW | 16 ul |
| Total | 20 ul |
Inset
| DNA solution (35 ng/ul) | 14 ul |
| XbaI | 1 ul |
| SpeI | 1 ul |
| 10xM buffer | 2 ul |
| DW | 2 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 120 |
| 2 | 60 | 15 |
| 3 | 4 | HOLD |
As a background, we did PCR for eCFP again.
We did not do digestion of vector DNA sokution, result, because it has only too low concentration to be digested product solution.
We failed PCR again.
The digestion result was confirmed that the Insert DNA was successfully digested by XbaI and SpeI.
And we extracted the insert DNA digestion product from agarose gel by gel-extraction kit and got 20 ul of Insert DNA solution.
Digestion of pSB1AK3 with hindIII
The gel-extracted DNA solution contains Ag43-dT DNA and pSB1AK3 DNA because these DNA have same size of base pair and exist as same band in agarose gel. Thus we digest pSB1AK3 with HindIII to separate it from Ag43-dT.
| DNA solution (35 ng/ul) | 20 ul |
| HindIII | 1 ul |
| 10xM buffer | 3 ul |
| DW | 7 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 180 |
| 2 | 70 | 10 |
| 3 | 4 | HOLD |
As a background, we did PCR for eCFP again.
We failed the PCR for E0020.
The digestion result was confirmed that the Insert DNA was successfully digested with HindIII.
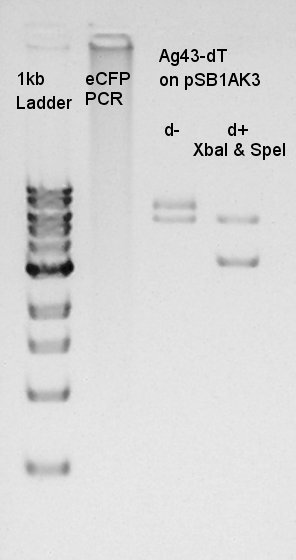
Ethanol precipitation of pT7-RBS-Ag43-dT-pSB1C3 and Ag43-dT
We did ethanol precipitation to get more high concentration DNA solution of each part. And we got 5ul of each DNA solution.
The electrophoresis result showed that the pT7-RBS-pSB1C3 construct was successfully digested by SpeI and get enough concentration of DNA solution to ligate.
Ligation of pT7-RBS-pSB1C3 and Ag43-dT
Ligation and pT7-RBS-pSB1C3 (as Vector) and Ag43-dT (as Insert). We used Ligation Mighty Mix (TAKARA BIO INC.) which contains ligase and buffer.
| Vector DNA | 2.5 ul |
| Insert DNA | 2.5 ul |
| Ligation Mighty Mix | 5 ul |
| Total | 10 ul |
Ligation reaction time was in detail below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
Incubation for mini-prep of Ag43-dT-pSB1AK3 and ptet-RBS-YFP-dT-pSB1A2
Incubation of Ag43-dT-pSB1AK3 and ptet-RBS-YFP-dT-pSB1A2 in LBA liquid medium.
- Prepared 2 ml LBC into culture tubes.
- Re-suspended 1 colony from each plate that each construct plated.
- Incubated at 37C for 22 hrs (Ag43-dT-pSB1AK3 was Incubated for 41 hrs)
August 31st
Plasmid extraction for pBAD-RBS-Ag43-dT on pSB1AK3
We used promega SV miniprep kit. We got 100 ul DNA solution.
Digestion
We digested mini-prep products (pBAD-RBS-Ag43-dT on pSB1AK3) by EcoRI and SpeI. Because I'd like to check the size of insert and vector of getting plasmid.
| DNA solution | 6 ul |
| EcoRI | 1 ul |
| SpeI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 10 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 120 |
| 2 | 60 | 15 |
| 3 | 4 | HOLD |
Transformation of pT7-RBS-Ag43-dT-pSB1C3 and eCFP (E0020)
- Mixed 2 ul ligation product and 1ul E0020 DNA solution to each 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Mixed 350 ul of LB.
- Prepared and Labeled two plastic plates with LB plate medium which contained appropriate antibiotics (LBA and LBC).
- Incubated for 2 hrs to get the resistance to Chloramphenicol (E0020 mixture skipped this step).
- Plated 300 ul of the culture onto first dish and spread.
- Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for 15 hours.
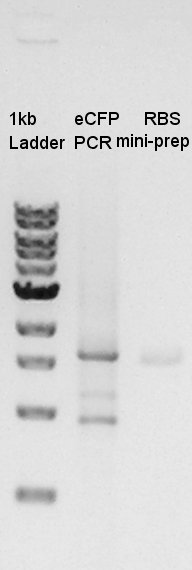
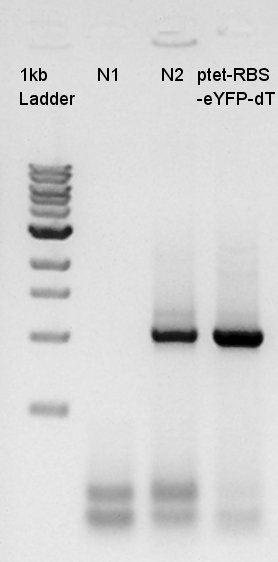
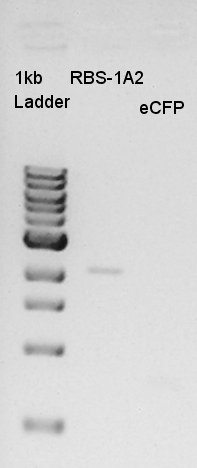
Confirmation of amplification of ptet-RBS-eYFP-dT-pSB1A2
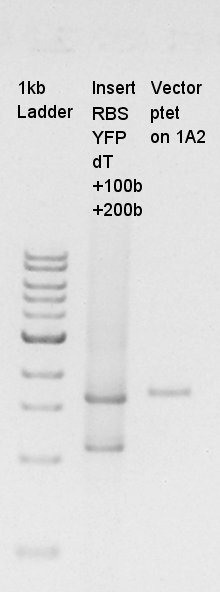
After mini-prep, we confirmed whether the desired construct ptet-RBS-eYFP-dT was really amplified or not by PCR. We chose EX-F primer as forward primer and PS-R primer as reverse primer then amplified about 1000bp of insert DNA. As controls, we chose DW as N1 and ptet-RBS-eCFP-dT construct as N2.
| DNA solution | 4 ul |
| Kapa-Taq(Taq polymerase) | 5 ul |
| Forward Primer(EX-F primer) | 0.5 ul |
| Reverse Primer(PS-R primer) | 0.5 ul |
| Total | 10 ul |
| Number | Degree | Second |
| 1 | 95 | 120 |
| 2 | 95 | 30 |
| 3 | 68.9 | 30 |
| 4 | 72 | 60 |
| 5 | 72 | 60 |
| 6 | 4 | HOLD |
Cycle:2~4 x 35
This image showed that the desired construct (about 1000 bp) amplified by incubation.
Incubation for mini-prep of eCFP-pSB1A2
Incubation of eCFP-pSB1A2 in LBA liquid medium.
- Prepared 2 ml LBA into culture tubes.
- Re-suspended 1 colony.
- Incubated at 37C for 16 hrs.
Transformation of ptet-RBS-eYFP-dT-pSB1A2 and pT7-RBS-Ag43-dT-pSB1C3
- Mixed 2 ul pT7-RBS-Ag43-dT-pSB1C3 ligation product and 1ul ptet-RBS-eYFP-dT-pSB1A2 DNA solution to each 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Mixed 350 ul of LB.
- Prepared and Labeled two plastic plates with LB plate medium which contained appropriate antibiotics (LBA and LBC).
- Incubated for 2 hrs to get the resistance to Chloramphenicol (ptet-RBS-eYFP-dT-pSB1A2 mixture skipped this step).
- Plated 300 ul of the culture onto first dish and spread.
- Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for hours.
September 1st
Digestion for pSB1C3
We have to change the plasmid backbone to pSB1C3. We got the insert DNA at August 31th. So we need to get the pSB1C3 solution digested by EcoRI and SpeI.
| pSB1C3 (60 ng/ul) | 3 ul |
| Eco RI | 1 ul |
| SpeI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 13 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 180 |
| 2 | 60 | 15 |
| 3 | 4 | HOLD |
Gel extraction
Gel extraction for digestion product. We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50 ul of DNA solution.
Ethanol precipitation
Ethanol precipitation to get more high concentration of pBAD-RBS-Ag43-dT solution and pSB1C3 solution digested by EcoRI & SpeI.
- Added 2 ul of NaoAc, 1.5 ul of glycogen and 50 ul of 100% ethanol.
- Centrifuged at 15000 rpm, 10 min at 4C.
- Removed supernatant and added 220 ul of 70% ethanol.
- Centrifuged at 15000 rpm, 10 min at 4C.
- Removed supernatant and air drying at room temperature then added 10 ul of DW.
Ligation
Ligation for insert DNA and pSB1C3.
| Vector DNA | 1 ul |
| Insert DNA | 4 ul |
| Ligation Mighty Mix | 5 ul |
| Total | 10 ul |
Ligation reaction time was in detail below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
Transformation
Transformation for ligation product in DH5α.
- Added 1 ul of DNA to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Added 350 ul of LB.
- Incubated the cells for 2 hours at 37C.
- Plated 300 ul of the culture onto first LBC dish and spread.
- Added 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second LBC dish and spread.
- Incubated the plates at 37C for 18 hrs.
Plasmid extraction of Ag43-dT-pSB1AK3 and RBS-pSB1A2
We used mini-prep kit of Nippon genetics and got 50 ul Ag43-dT-pSB1AK3 plasmid DNA solution.
Digestion of eCFP-pSB1A2 and RBS-pSB1A2
To make a construct of eCFP-RBS-pSB1A2, we digested eCFP-pSB1A2 by EcoRI and SpeI, and RBS-pSB1A2 with EcoRI and XbaI. And we digested pT7-RBS-pSB1C3 by XbaI as a control for confirmation of the ability to digest. Insert (eCFP-pSB1A2)
| DNA solution (35 ng/ul) | 11 ul |
| EcoRI | 1 ul |
| SpeI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 5 ul |
| Total | 20 ul |
Vector(RBS-pSB1A2)
| DNA solution (29 ng/ul) | 6 ul |
| EcoRI | 1 ul |
| XbaI | 1 ul |
| 10xM buffer | 2 ul |
| DW | 10 ul |
| Total | 20 ul |
control (pT7-RBS-pSB1C3)
| DNA solution (30 ng/ul) | 6 ul |
| XbaI | 1 ul |
| 10xM buffer | 2 ul |
| 100x BSA | 0.2 ul |
| DW | 11 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 120 |
| 2 | 60 | 15 |
| 3 | 4 | HOLD |
From this image, we confirmed that DNA were digested into fragments and all of restriction enzyme worked.
September 2nd
Ethanol precipitation of digestion products (eCFP and RBS-pSB1A2) and estimation of concentration
- Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol.
- Centrifuged at 14000 rpm, 30 min at 4C.
- Removed supernatant and added 220 ul of 70% ethanol.
- Centrifuged at 15000 rpm, 15 min at 4C.
- Removed supernatant and air drying at room temperature then added 5 ul of DW.
We estimated the concentration of ethanol presipitation products.The concentration of Insert DNA solution is about 20 ng/ul and Vector DNA solution is about 37 ng/ul.
Incubation of pT7-RBS-Ag43-dT-pSB1C3 transformation product
We failed to cultivation of above construct two times so we tried to incubation in LBC liquid medium.
- Prepared 1.8 ml LBC into culture tubes.
- Re-suspended 200 ul transformation product solution (which was mixed with LB and incubated for 3 hrs).
- Incubated at 37C.
Ligation of eCFP and RBS-pSB1A2
| Vector DNA (37 ng/ul) | 1 ul |
| Insert DNA (20 ng/ul) | 4 ul |
| Ligation Mighty Mix | 6 ul |
| Total | 11 ul |
Ligation reaction time was in detail below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
Transformation of eCFP-RBS-pSB1A2
- Mixed 2 ul eCFP-RBS-pSB1A2 ligation product to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Mixed 350 ul of LB.
- Prepared and Labeled two plastic plates with LB plate medium which contained appropriate antibiotics (LBA).
- Plated 300 ul of the culture onto first dish and spread.
- Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for 19 hours.
 "
"