Team:HokkaidoU Japan/Notebook/aggregation Week 4
From 2012.igem.org
| Line 157: | Line 157: | ||
|} | |} | ||
| - | [[image:|thumb|Digestion result(Ag43-dT and pT7-RBS)]] | + | [[image:HokkaidoU2012 120725 Digestion Ag43-dt on pSB1AK3(HindIII) & pT7-RBS on pSB1K3(SpeI).jpg|thumb|Digestion result(Ag43-dT and pT7-RBS)]] |
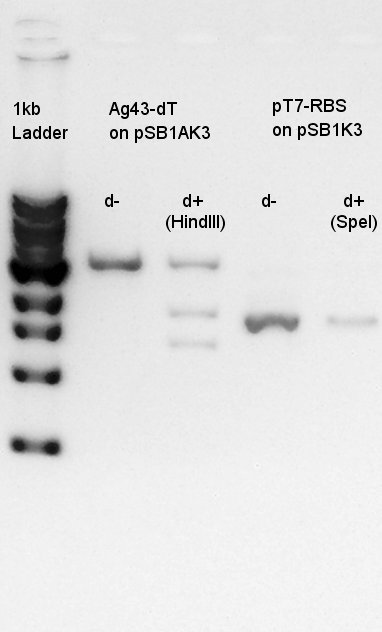
| + | We were confirmed that pAB1AK3 was digested into 1.3k and 1.8k bp fragments by HindIII. But there are a little doubt SpeI wasn't work because the band of pT7-RBS on pSB1K3 of d- and d+ existed same bp area. | ||
==Gel extraction== | ==Gel extraction== | ||
<p> | <p> | ||
| Line 175: | Line 176: | ||
</p> | </p> | ||
| - | + | ||
<div></div> | <div></div> | ||
<!-- DO NOT EDIT UNDER THIS LINE @iTakeshi --> | <!-- DO NOT EDIT UNDER THIS LINE @iTakeshi --> | ||
Revision as of 10:51, 25 July 2012
Contents |
July 23th
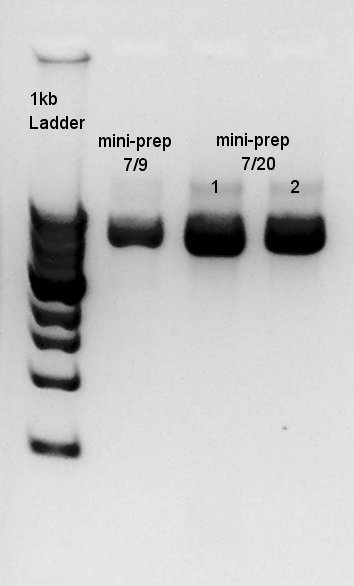
Mini-prep
mini-prep for Ag43(7/17 cultivated colony resuspended and 7/20 cultivated colony resuspended). We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50ul of DNA solutions.
July 24th
Electrophoresis
Electrophoresis for Ag43(mini-preped above) and Ag43 digestion results(digested with EcoRI and SpeI)
In this digestion result, we knew that one or two enzymes didn't work successfully but there are enough concentration of DNA in 3000bp band to use in digestion.
Gel extraction
Gel extraction for digestion production. We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50ul of DNA solution.
Ethanol precipitation
Ethanol precipitation for digestion and gel extraction product.
- Added 5ul of NaoAc, 1.5ul of glycogen and 125ul of 100% ethanol.
- Centrifuged in 15000rpm, 10min at 4C.
- Remove supernatant and added 220ul of 70% ethanol.
- Centrifuged in 15000rpm, 10min at 4C.
- Remove supernatant and air drying in room temperature then added 10ul of DW.
In this result, we estimated that the concentration of ethanol precipitation product is about 40ng/ul.
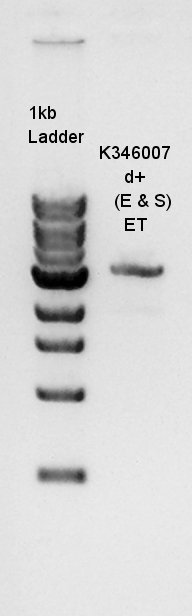
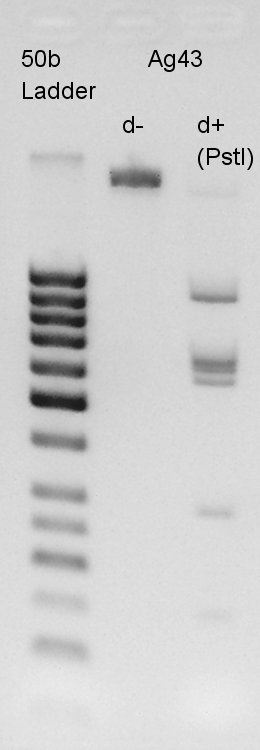
Digestion
Digestion to confirm how many PstI cutting sites are in K346007(Ag43 coading) and Ag43-dT complex digestion with SpeI and XbaI. Ag43 PstI
| DNA solution | 5ul |
| PstI | 1ul |
| 10xH buffer | 2ul |
| DW | 12ul |
| Total | 20ul |
Ag43-dT
SpeI and XbaI
| DNA solution | 12ul |
| SpeI | 1ul |
| XbaI | 1ul |
| 10xH buffer | 2ul |
| DW | 4ul |
| Total | 20ul |
Electrophoresis
Electrophoresis for digestion results.
In this result, we found that there are 6 PstI cutting sites in K346007(Ag43).
Gel extraction
Gel ectraction of Ag43-dt digestio result.We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50ul of DNA solution.
Digestion
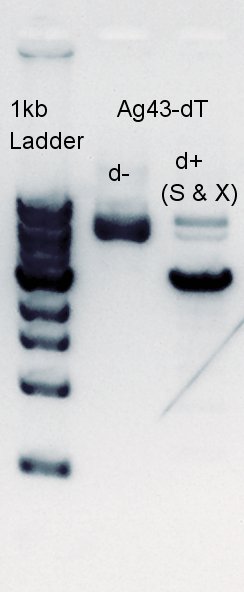
Digestion for Ag43-dT and pSB1AK3 mixture(each DNA fragment is about 3kbp) with HindIII to digest pSB1AK3 into about two 1.5kbp fragments.
| DNA solution | 8ul |
| HindIII | 1ul |
| 10xM buffer | 1ul |
| Total | 10ul |
July 25th
Digestion
Digestion of pT7-RBS on pSB1K3 cutting with SpeI.
| DNA solution | 3ul |
| SpeI | 1ul |
| 10xH buffer | 1ul |
| DW | 5ul |
| Total | 10ul |
We were confirmed that pAB1AK3 was digested into 1.3k and 1.8k bp fragments by HindIII. But there are a little doubt SpeI wasn't work because the band of pT7-RBS on pSB1K3 of d- and d+ existed same bp area.
Gel extraction
<p> Gel extraction of Ag43-dT on pSB1AK3(HindIII) and pT7-RBS on pSB1K3(SpeI). We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50ul of DNA solution.
Ethanol precipitation
Ethanol precipitation of digestion and gel extraction products.
- Added 5ul of NaoAc, 1.5ul of glycogen and 125ul of 100% ethanol.
- Centrifuged in 15000rpm, 10min at 4C.
- Remove supernatant and added 220ul of 70% ethanol.
- Centrifuged in 15000rpm, 5min at 4C.
- Remove supernatant and air drying in room temperature then added 5ul of DW.
 "
"