Team:NTU-Taida/Project/Thermal-Avenger
From 2012.igem.org
Darlisereno (Talk | contribs) |
(→Modified promoter cI with CIts) |
||
| (19 intermediate revisions not shown) | |||
| Line 3: | Line 3: | ||
{{:Team:NTU-Taida/Templates/BSHero|Title=Thermal Avenger|Content=}} | {{:Team:NTU-Taida/Templates/BSHero|Title=Thermal Avenger|Content=}} | ||
| + | <!-- Thermal Part --> | ||
| - | < | + | ==P<sub>hs</sub> thermal promoter (toxin, anti-toxin)== |
| - | + | ||
| - | < | + | ''E. coli'' is pretty sensitive and responsive to temperature changes. We brought a thermal sensitive promoter P<sub>hs</sub>, located within dnaG, and transient induced to temperature upshift, to our circuit.(1) As described by Wayne E. Talyor et al., P<sub>hs</sub> can forward upregulation of a series of proteins. The increase activity of P<sub>hs</sub> promoter is partially compatible with the increase synthesis of sigma factor. The quick respsonse of P<sub>hs</sub> is suitable since its the sigma factor synthesis peaks 10 minutes after the temperature upshift, and so does P<sub>hs</sub> RNA levels. The ratio of increase before and after the temperature rise can be more than 20 folds. On particular note, P<sub>hs</sub> is lack of consensus region over -10 region, which explains its poor activity in 30℃. Thus, P<sub>hs</sub> is marked as a candidate in our circuit, which can only be functional after ingestion into human body. |
| - | + | ||
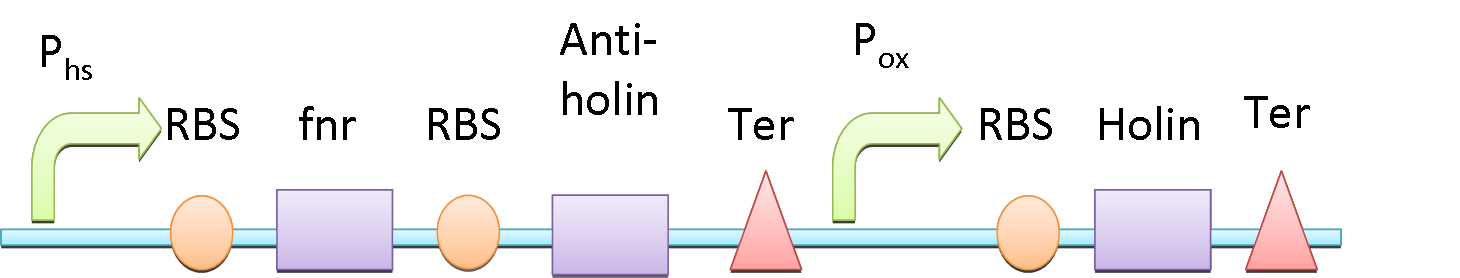
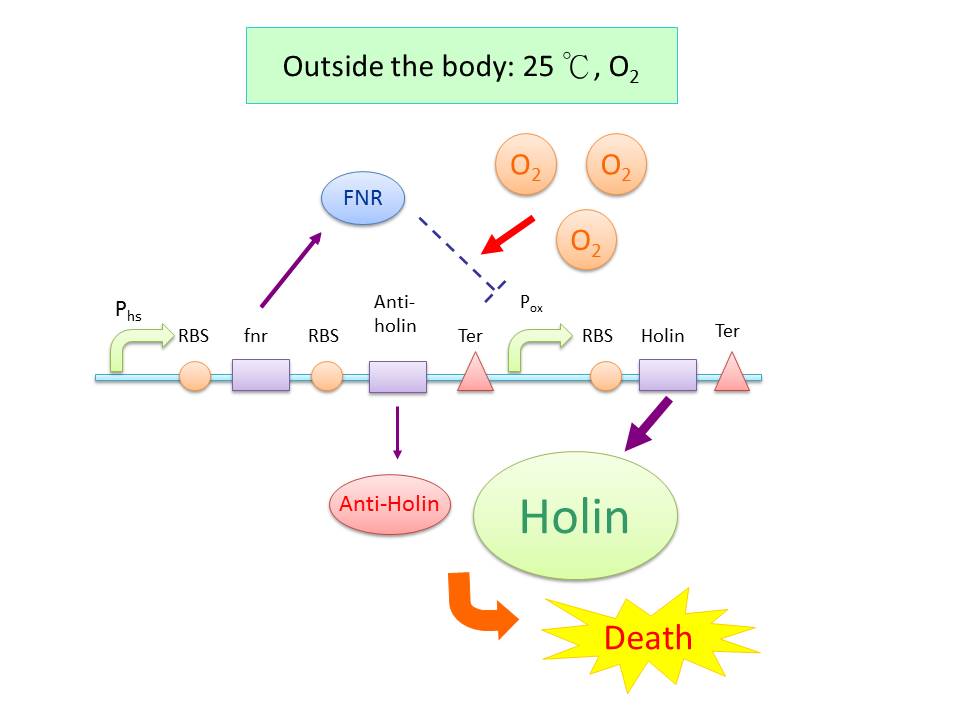
| - | + | We incorporate a toxin and anti-toxin system into our circuit, as a sensitive device to the environment. Holin, a membrane bound protein, can form complex with each other and trigger the disruption of the membrane. Anti-holin, which is bound to the inner membrane, can bind and inactivate Holin. We put Holin under the control of an oxygen sensitive protein, FNR (fumarate nitrate reductase regulator)(2), which detach from its promoter region (P<sub>ox</sub>) in the presence of oxygen and no longer suppress it. When the bacteria is in the anaerobic environment of gut, the temperature raises to 37℃, so the synthesis of Holin is repressed by FNR which is downstream of P<sub>hs</sub>, and the Anti-Holin synthesis also add on to circumvent the lysis of the bacteria.(3) However, when the bacteria are excreted outside the body, the temperature goes down and the bacteria expose to oxygen, the synthesis Holin would not be suppressed, and thus poise for the cellular lysis. This design can make sure that our ''E. coli'' would not spread and grow outside of human body. | |
| - | + | [[File:NTU-Taida-Phs.png|600px|thumb|center|]] | |
| + | {|align="center" | ||
| + | | [[File:NTU-Taida-Project-Thermal-fig1.png|450px|thumb|center|]] | ||
| + | | [[File:NTU-Taida-Project-Thermal-fig2.png|450px|thumb|center|]] | ||
| + | |} | ||
| - | + | ==Modified promoter cI with CIts== | |
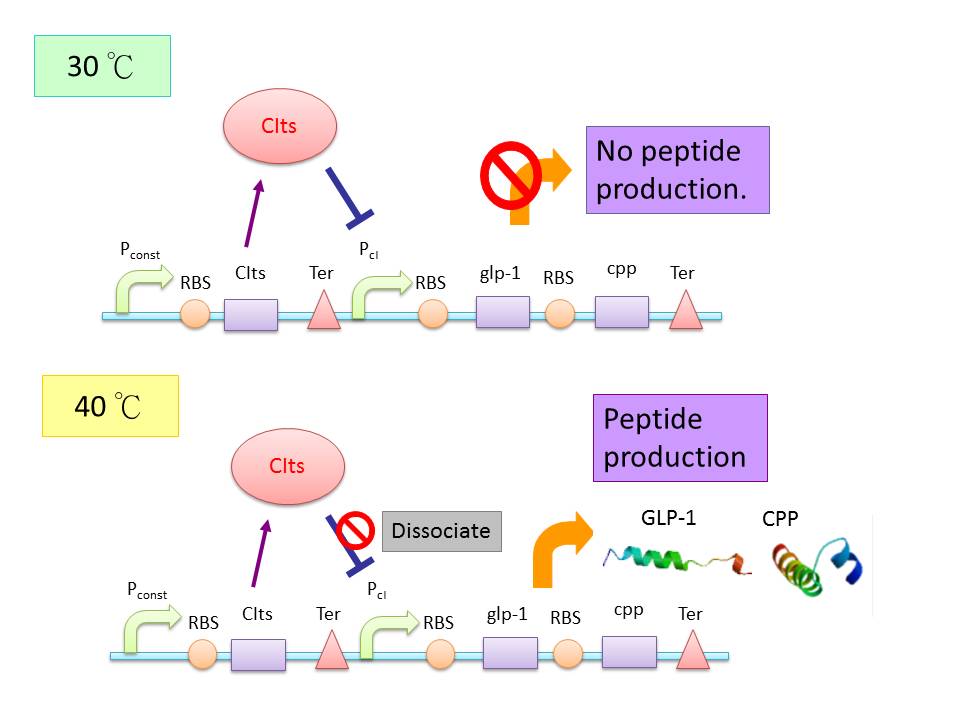
| - | + | This circuit involves a thermosensitive cI promoter under the expression of a strong promoter, J23119, and is followed by a CI promoter (P<sub>cI</sub>) region and reporter of desire. The whole composite circuit is designed by Harvard iGEM team, 2008. Under conditions demonstrated before, its increase of reporter expression after temperature upshift is minimal (1.8 fold). And thus, we design novel cI promoter region with altered binding affinity to CIts repressor in search of better efficiency. (4) | |
| + | The circuit aims to provide stable and sustainable GLP-1 synthesis inside human body, that is, the bacteria showed no ability to secret GLP-1 outside of human body under room temperature, under which the P<sub>cI</sub> is suppressed by CIts. | ||
| - | |||
| - | + | [[File:NTU-Taida-Project-CIts-png|600px|thumb|cneter|]] | |
| + | [[File:NTU-Taida-Project-Thermal-fig3.png|450px|thumb|center|]] | ||
| - | + | ===Reference=== | |
| - | + | 1. Taylor, W. E., D. B. Straus, et al. (1984). "Transcription from a heat-inducible promoter causes heat shock regulation of the sigma subunit of E. coli RNA polymerase." Cell 38(2): 371-381. | |
| - | + | 2. Reinhart, F., S. Achebach, et al. (2008). "Reduced apo-fumarate nitrate reductase regulator (apoFNR) as the major form of FNR in aerobically growing Escherichia coli." J Bacteriol 190(3): 879-886 | |
| - | + | 3. Young, R. (2002). "Bacteriophage holins: deadly diversity." J Mol Microbiol Biotechnol 4(1): 21-36. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | <!-- | + | 4. Wedler, H. and R. Wambutt (1995). "A temperature-sensitive lambda cI repressor functions on a modified operator in yeast cells by masking the TATA element." Mol Gen Genet 248(4): 499-505 |
| + | |||
| + | <!-- EOF --> | ||
| + | {{:Team:NTU-Taida/Templates/ContentEnd}}{{:Team:NTU-Taida/Templates/Footer|ActiveNavbar=Project, #nav-Project-Thermal}} | ||
Latest revision as of 12:45, 22 October 2012
Thermal Avenger
Contents |
Phs thermal promoter (toxin, anti-toxin)
E. coli is pretty sensitive and responsive to temperature changes. We brought a thermal sensitive promoter Phs, located within dnaG, and transient induced to temperature upshift, to our circuit.(1) As described by Wayne E. Talyor et al., Phs can forward upregulation of a series of proteins. The increase activity of Phs promoter is partially compatible with the increase synthesis of sigma factor. The quick respsonse of Phs is suitable since its the sigma factor synthesis peaks 10 minutes after the temperature upshift, and so does Phs RNA levels. The ratio of increase before and after the temperature rise can be more than 20 folds. On particular note, Phs is lack of consensus region over -10 region, which explains its poor activity in 30℃. Thus, Phs is marked as a candidate in our circuit, which can only be functional after ingestion into human body.
We incorporate a toxin and anti-toxin system into our circuit, as a sensitive device to the environment. Holin, a membrane bound protein, can form complex with each other and trigger the disruption of the membrane. Anti-holin, which is bound to the inner membrane, can bind and inactivate Holin. We put Holin under the control of an oxygen sensitive protein, FNR (fumarate nitrate reductase regulator)(2), which detach from its promoter region (Pox) in the presence of oxygen and no longer suppress it. When the bacteria is in the anaerobic environment of gut, the temperature raises to 37℃, so the synthesis of Holin is repressed by FNR which is downstream of Phs, and the Anti-Holin synthesis also add on to circumvent the lysis of the bacteria.(3) However, when the bacteria are excreted outside the body, the temperature goes down and the bacteria expose to oxygen, the synthesis Holin would not be suppressed, and thus poise for the cellular lysis. This design can make sure that our E. coli would not spread and grow outside of human body.
Modified promoter cI with CIts
This circuit involves a thermosensitive cI promoter under the expression of a strong promoter, J23119, and is followed by a CI promoter (PcI) region and reporter of desire. The whole composite circuit is designed by Harvard iGEM team, 2008. Under conditions demonstrated before, its increase of reporter expression after temperature upshift is minimal (1.8 fold). And thus, we design novel cI promoter region with altered binding affinity to CIts repressor in search of better efficiency. (4) The circuit aims to provide stable and sustainable GLP-1 synthesis inside human body, that is, the bacteria showed no ability to secret GLP-1 outside of human body under room temperature, under which the PcI is suppressed by CIts.
Reference
1. Taylor, W. E., D. B. Straus, et al. (1984). "Transcription from a heat-inducible promoter causes heat shock regulation of the sigma subunit of E. coli RNA polymerase." Cell 38(2): 371-381.
2. Reinhart, F., S. Achebach, et al. (2008). "Reduced apo-fumarate nitrate reductase regulator (apoFNR) as the major form of FNR in aerobically growing Escherichia coli." J Bacteriol 190(3): 879-886
3. Young, R. (2002). "Bacteriophage holins: deadly diversity." J Mol Microbiol Biotechnol 4(1): 21-36.
4. Wedler, H. and R. Wambutt (1995). "A temperature-sensitive lambda cI repressor functions on a modified operator in yeast cells by masking the TATA element." Mol Gen Genet 248(4): 499-505
 "
"