Team:HokkaidoU Japan/Notebook/Week 2
From 2012.igem.org
| Line 1: | Line 1: | ||
| + | {{Team:HokkaidoU_Japan/header}} | ||
| + | {{Team:HokkaidoU_Japan/nav.notebook}} | ||
| + | <div id="hokkaidou-column-main"> | ||
| + | <!-- DO NOT EDIT OVER THIS LINE @iTakeshi --> | ||
==July 9th== | ==July 9th== | ||
<div class="hokkaidou-notebook-daily"> | <div class="hokkaidou-notebook-daily"> | ||
Revision as of 05:35, 16 July 2012
Contents |
July 9th
pT7 + RBS (3A Assembly) and Ag43 + dT (standard assembly) ligation products were transformed and cultivated then some colonies were formed (12~16 colonies) so we confirmed really insert DNA (3A:pT7 and RBS, standard:Ag43 and dT) were inserted to vector or not by colony PCR.
- Colony PCR
Colony PCR for assembly products.Each product reacted recipes written below.
- picked up each 12(16 is best but there were only 12 colonies) colonies from LB plates by Autoclaved toothpicks.
- Dipped into 10ul DW in 1.5ml eppendorf tubes.
- from 10ul DW, 4ul was added in PCR reaction solution (below), 6ul was mixed with 200ul LB.
- Ran PCR machine in recipe below.
- Electrophoresis for confirmation of PCR results.
PCR reaction solution
| DNA solution | 4ul |
| KapaTaq ready mix | 5ul |
| BioBrick prefix forward primer | 0.5ul |
| BioBrick suffix reverse primer | 0.5ul |
| Total | 10ul |
PCR recipe
(pT7 + RBS)
| Number | Degree | Second |
| 1 | 94 | 120 |
| 2 | 94 | 30 |
| 3 | 68 | 60 |
| 4 | 4 | HOLD |
Cycle:2~3 x 40
(Ag43 + dT)
Ag43 + dT (+ Biobrick prefis & suffix) is about 3290bp. Extension step needed >180seconds.
| Number | Degree | Second |
| 1 | 94 | 120 |
| 2 | 94 | 30 |
| 3 | 68 | 180 |
| 4 | 4 | HOLD |
Cycle:2~3 x 35
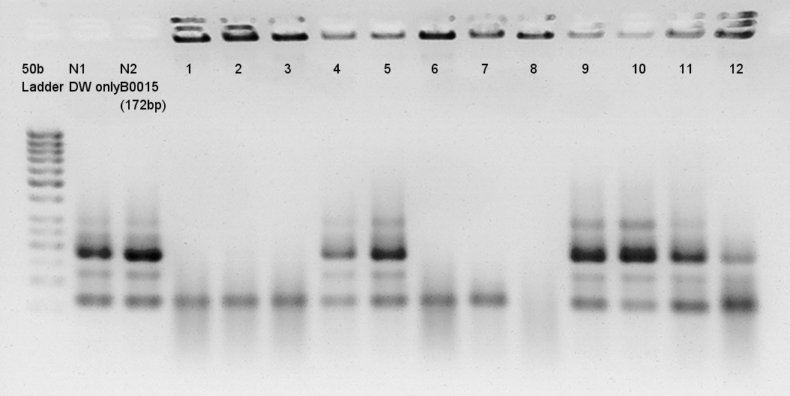
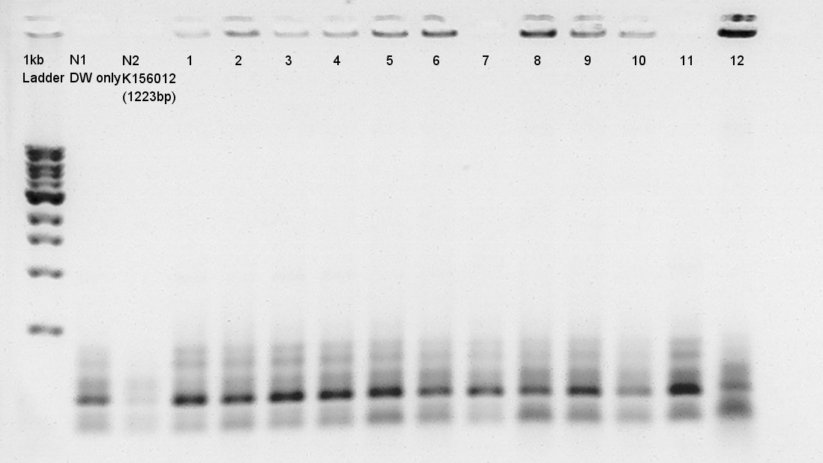
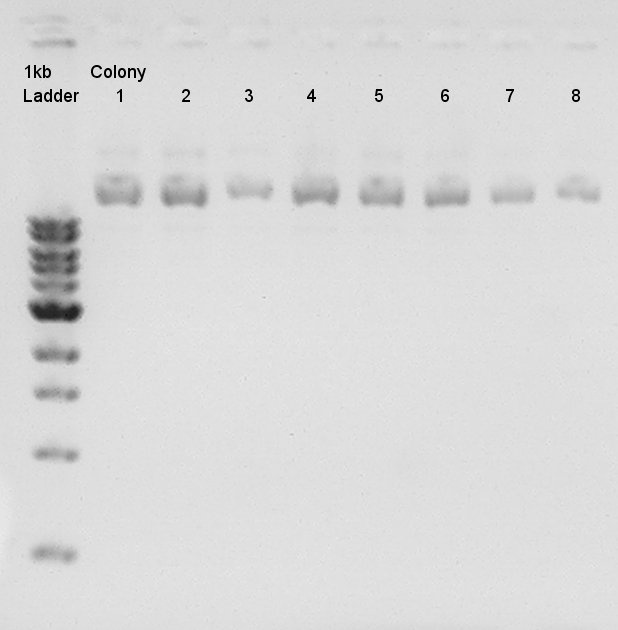
- Electrophoresis results
Electrophoresis for (pT7 + RBS) in 2% agarose gel and (Ag43 + dT) in 1% agarose gel.
pT7 + RBS on pSB1K3
bbp-Insert-bbs:86bp
Ag43 + dT on pSB1AK3
bbp-Insert-bbs:3290bp
We couldn't confirm insert DNA were really ligated with Vector or not.
Next we tried confirmation of insert DNA by Electrophoresis of mini-prep products.
For mini-prep, we needed do liquid culture.
- Liquid culturing
Liquid culture for mini-prep((pT7 + RBS) on pSB1K3 and (Ag43 + dT) on pSB1AK3).
- Prepared 1800ul LB solutions.
- To these LB solutions, added 200ul of LB solutions (colony PCR solutions were pre-cultivated in about 2hrs) and each antibiotics(Km (for (pT7 + RBS) on pSB1K3) and Amp(for (Ag43 + dT) on pSB1AK3)).
- Cultivated 15hrs30min.
July 10th
- Mini-prep
Mini-prep for some colonies (we selected colonies which showed more correct bands than other colonies, pT7+RBS were No.4, 5, 9, 10 colonies and Ag43 + dT were No.1, 2, 3, 4 colonies)cultivated in LBA(Ag43 + dT) and LBK(pT7 + RBS) in 15hrs 30min.
- Mini-prep for LB culturing products along the protocol of FastGene Plasmid Mini kit(Nippon Genetics).
- Electrophoresis (Used pre-migrated 1% agarose gels with 5ul EtBr)in 30min.
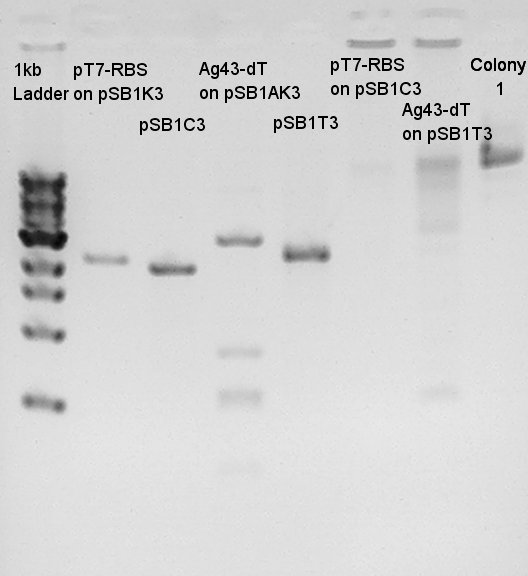
Electrophoresis resulsts
pT7 + RBS on pSB1K3(Total 2247bp)
Ag43 + dT on pSB1AK3(Total 6444bp)
To confirm more about insert DNA, we tried to digest these DNA with EcoRI and PstI.
- Digestion
Digestion for confirmation of insert DNA. Each DNA mini-prep products were digested with E & P.
Digestion recipe
pT7-RBS
| pT7-RBS | 1,5ul |
| EcoRI | 1ul |
| PstI | 1ul |
| 10xH buffer | 2ul |
| DW | 14.5ul |
| Total | 20ul |
Digestion recipe
Ag43-dT
| Ag43-dT | 4ul |
| EcoRI | 1ul |
| PstI | 1ul |
| 10xH buffer | 2ul |
| DW | 12ul |
| Total | 20ul |
Digestioned at 37c in 2hrs.
Digestion results
pT7+RBS
Ag43+dT
Insert DNA were correct we thought. We tried digestion for 3A Assembly[pT7-RBS + Ag43-dT + pSB1C3]
- Digestion
Digestion for 3A Assembly.
pT7-RBS
| DNA | 17ul |
| EcoRI | 1ul |
| SpeI | 1ul |
| 10xH buffer | 3ul |
| DW | 8ul |
| Total | 30ul |
Ag43-dT
| DNA | 12.5ul |
| XbaI | 1ul |
| PstI | 1ul |
| 10xH buffer | 2ul |
| DW | 3.5ul |
| Total | 20ul |
pSB1C3
| DNA | 20ul |
| EcoRI | 1ul |
| PstI | 1ul |
| 10xH buffer | 3ul |
| DW | 5ul |
| Total | 30ul |
Reacted in 2hrs at 37c.
July 11th
- Ligation
Ligation of pT7-RBS + Ag43-dT + pSB1C3(3A Assembly)
Ligation recipe
| pT7-RBS | 2ul |
| Ag43-dT | 2ul |
| pSB1C3 | 3ul |
| Ligation Mighty Mix(TAKARA) | 8ul |
| Total | 16ul |
Ligation reaction time was written below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
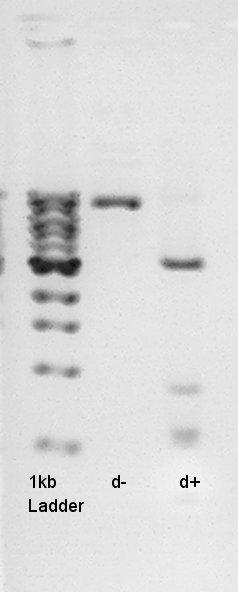
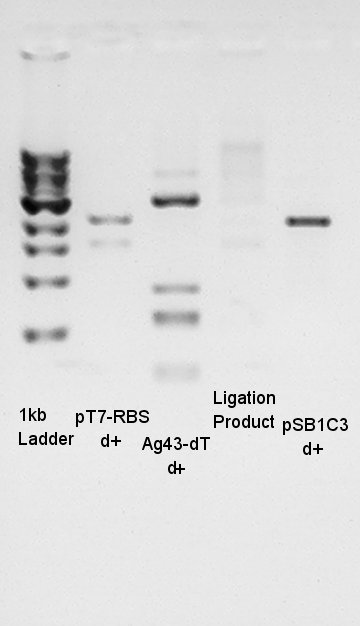
Electrophoresis result
- Transformation
Transformation for pT7-RBS + Ag43-dT + pSB1C3 into BL21(DE3)(E.coli which have T7 polymerase coadin site in their genomic DNA).
- Added DNA soltions (Ligation products) 1ul to BL21(DE3) compitent cell.
- Stood on ice in 30min.
- Heatshock for 1min at 42c.
- Added 200ul of LB to transformed BL21(DE3) solution.
- Pre-cultivate in 2hrs
- Splead 200ul of LB&BL21(DE3) solution supernant to LBC.
- 50ul of LB&BL21(DE3) solution were added to 200ul LB solution then spread 200ul to LBC plate.
- Cultivated.
July 12th
- Colony PCR
Colony PCR for ligation product.Each products were reacted in recipes written below.
- picked up each 16 colonies from LB plates by Autoclaved toothpicks.
- Dipped into 10ul DW in 1.5ml eppendorf tubes.
- from 10ul DW, 4ul was added in PCR reaction solution (below), 6ul was mixed with 200ul LB.
- Ran PCR machine in recipe below.
- Electrophoresis for confirmation of PCR results.
PCR reaction solution
| DNA solution | 4ul |
| KapaTaq ready mix | 5ul |
| BioBrick prefix forward primer | 0.5ul |
| BioBrick suffix reverse primer | 0.5ul |
| Total | 10ul |
PCR recipe
(pT7 + RBS)
| Number | Degree | Second |
| 1 | 94 | 120 |
| 2 | 94 | 30 |
| 3 | 68 | 90 |
| 4 | 4 | HOLD |
Cycle:2~3 x 40
- Liquid culturing
Liquid culture for some colonies used in colony PCR.
- Prepared 200ul LB solutions.
- To these LB solutions, added 6ul of LB solutions (colony PCR solutions were pre-cultivated in about 3hrs) and added 2ml LB and antibiotic(Cp).
- Cultivated 18hrs30min.
July 13th
- Mini-prep
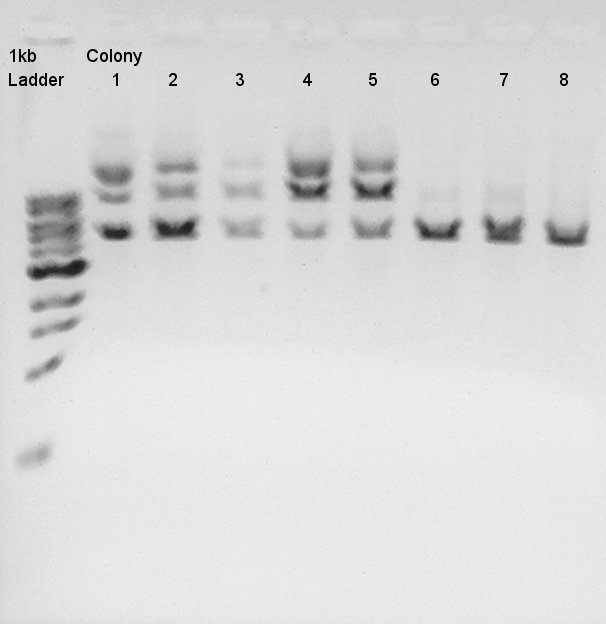
Mini-prep for colony No.1~8 We used mini-prep kit FastGene Plasmid Mini Kit (Nippon Genetics).
Mini-prep result
- Digestion
Digestion to confirm how DNA fragments ligated.
Digestion Recipe
| DNA | 4ul |
| EcoRI | 0.5ul |
| PstI | 0.5ul |
| 10xH buffer | 2ul |
| DW | 13ul |
| Total | 20ul |
Digestion result
July 14th
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
Blue Screen!!!!
- Ligation
Ligation for digestion fragments written above.
Ligation recipe Each DNA fragments (pT7-RBS + pSB1C3 and Ag43-dT + pSB1T3) were reacted in recipe written below.
| Insert | 5ul |
| Vector | 1ul |
| Ligation Mighty Mix(TAKARA) | 6ul |
| Total | 12ul |
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
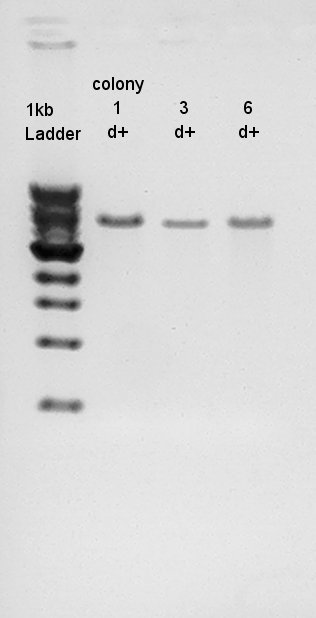
Ligation result with colony no.1 (see colony pcr result in 9th)
- Transformation
Transformation for ligation products written above.
- Added DNA soltions (Ligation products) 1ul to DH5α compitent cell.
- Stood on ice in 30min.
- Added 600ul of LB to transformed DH5α solution.
- Pre-cultivate in 2hrs
- Splead 300ul of LB&DH5α solution to LBC and LBT , 100ul added into 900ul of LB.
- Splead 300ul of LB&DH5α solution from 1000ul LB (100ul added into 900ul) to LBC and LBT.
- Cultivated in 21hrs.
and to get more conformation about pT7-RBS-Ag43-dT on pSB1C3 was really ligated, we tried digest this DNA with EcoRI and PstI once more time.
result
- Liquid culture
Liquid culture for Ag43(BBa_K346007)
- Picked up one colony from single colony isolated plate.
- Dipped into LBC.
- cultivated.
July 15th
- Gel extraction
We used Gel Extraction Kit (FastGene Gel/PCR extraction kit:NipponGenetics) to purify digestion products (see 14th).
- Digestion
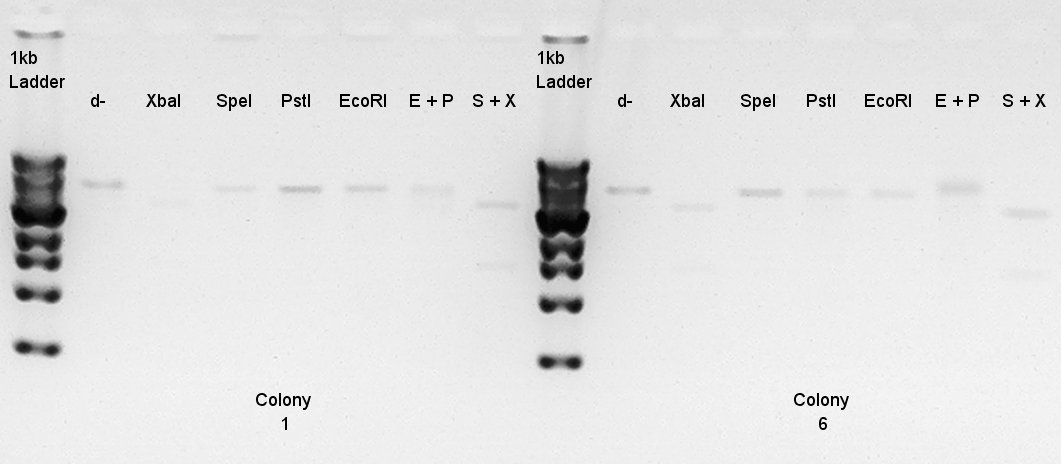
Digestion to confirm what kind of restriction enzyme cutting sites these DNA (colony 1 and 6) have.
EcoRI
| DNA solution | 6ul |
| EcoRI | 1ul |
| 10xH buffer | 1ul |
| DW | 2ul |
| Total | 10ul |
XbaI
| DNA solution | 6ul |
| XbaI | 1ul |
| 10xM buffer | 1ul |
| BSA | 1ul |
| DW | 1ul |
| Total | 10ul |
PstI
| DNA solution | 6ul |
| PstI | 1ul |
| 10xH buffer | 1ul |
| DW | 2ul |
| Total | 10ul |
SpeI
| DNA solution | 6ul |
| SpeI | 1ul |
| 10xM buffer | 1ul |
| DW | 2ul |
| Total | 10ul |
EcoRI + PstI
| DNA solution | 6ul |
| EcoRI | 1ul |
| PstI | 1ul |
| 10xH buffer | 1ul |
| DW | 11ul |
| Total | 20ul |
XbaI + SpeI
| DNA solution | 6ul |
| XbaI | 1ul |
| SpeI | 1ul |
| 10xM buffer | 1ul |
| DW | 11ul |
| Total | 20ul |
- Ethanol precipitation
Ethanol precipitation for digestion products.
- Added 2ul of NaoAc, 1.5ul of glycogen and 50ul of 100% ethanol.
- Centrifuged in 15000rpm, 10min at 4C.
- Remove supernatant and added 220ul of 70% ethanol.
- Centrifuged in 15000rpm, 5min at 4C.
- Remove supernatant and air drying in room temperature then added 5ul of DW.
- Electrophoresis
Electrophoresis for digest-ethanol precipitation products.
- added 5ul of EtBr.
- Migrated in 30min.
Digestion result
Digestion results for pT7-RBS-RFP-dT on pSB1C3 (once digested with EcoRI and PstI)
- Single colony isolation
Single colony isolation for Transformation products synthesized yesterday.
- one colony picked up from cultivated LBC and LBT plate.
- Spread on LBC and LBT.
- Cultivated.
- Digestion
Digestion of Ag43(with S,P), dT(With X,P) and pT7-RBS(With E).
Ag43
| DNA solution | 9ul |
| SpeI | 1ul |
| PstI | 1ul |
| 10xH buffer | 2ul |
| DW | 7ul |
| Total | 20ul |
dT
| DNA solution | 1.1ul |
| XbaI | 1ul |
| PstI | 1ul |
| 10xM buffer | 2ul |
| DW | 14.9ul |
| Total | 20ul |
pT7-RBS
| DNA solution | 6ul |
| EcoRI | 1ul |
| 10xH buffer | 2ul |
| DW | 11ul |
| Total | 20ul |
 "
"