Team:NTU-Taida/Project/Safety
From 2012.igem.org
(→Safety of Delivery System) |
(→Horizontal Gene Transfer Blocker) |
||
| (15 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
{{:Team:NTU-Taida/Templates/Header}}{{:Team:NTU-Taida/Templates/Navbar}}{{:Team:NTU-Taida/Templates/Sidebar|Title=Safety}}{{:Team:NTU-Taida/Templates/ContentStart}} | {{:Team:NTU-Taida/Templates/Header}}{{:Team:NTU-Taida/Templates/Navbar}}{{:Team:NTU-Taida/Templates/Sidebar|Title=Safety}}{{:Team:NTU-Taida/Templates/ContentStart}} | ||
| - | {{:Team:NTU-Taida/Templates/BSHero|Title=Safety|Content=}} | + | {{:Team:NTU-Taida/Templates/BSHero|Title=Safety|Content=<p>Several safety issues are considered and answered here. To prevent horizontal gene transfer between our PEPDEX and other intestinal microbes, an RNAi-like system is designed to block this process. Also, to avoid undesirable growth and spread of our PEPDEX in the environment, a suicide device will kill the bacteria once they escape from the human body. Furthermore, if the intestinal bacterial populations have dramatic imbalance caused by PEPDEX, we can adopt traditional antibiotics use and perform the fecal microbiota transplantation to re-establish the normal composition of floral communities of the host.</p>}} |
| - | == | + | == Horizontal Gene Transfer Blocker== |
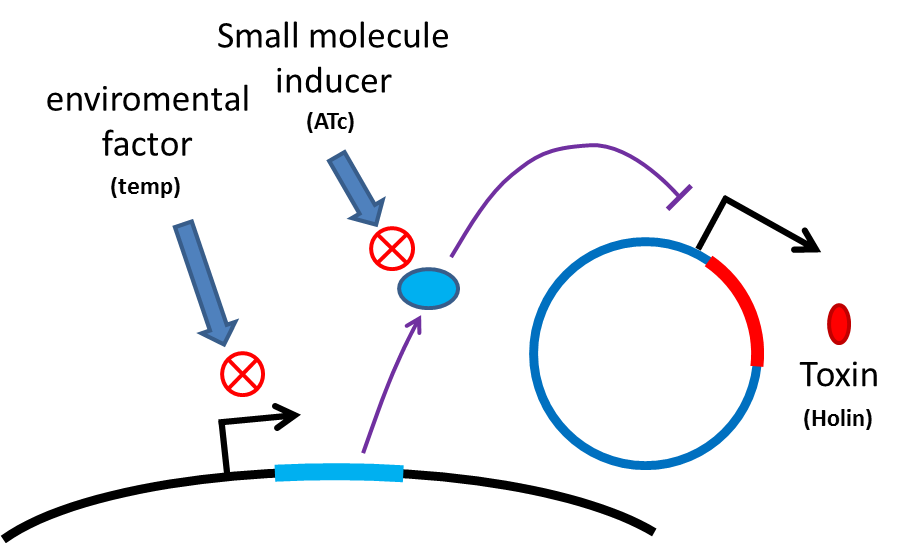
<p style="text-indent: 2em;">Many turn off strategies have been developed, most of them are inducible suicide systems that can be activated at certain conditions. In our project, we plan to use temperature as the activating signal (or alternatively, small molecules). When the course of treatment ends, leaving human body (temperature drop from 37C to room temperature) or administration of small amount of drugs (e.g. tetracycline) will cause suicide gene activation, thereby avoiding recombinant strain/gene pollution. And splitting suicide system to provide repression in trans can prevent plasmid transferring to wild type strains. Many off-the-rack parts can be used.</p> | <p style="text-indent: 2em;">Many turn off strategies have been developed, most of them are inducible suicide systems that can be activated at certain conditions. In our project, we plan to use temperature as the activating signal (or alternatively, small molecules). When the course of treatment ends, leaving human body (temperature drop from 37C to room temperature) or administration of small amount of drugs (e.g. tetracycline) will cause suicide gene activation, thereby avoiding recombinant strain/gene pollution. And splitting suicide system to provide repression in trans can prevent plasmid transferring to wild type strains. Many off-the-rack parts can be used.</p> | ||
[[File:NTU-Taida-Suicide-system.png|300px|thumb|left|alt=new idea about safety|general scheme of suicide system ]] | [[File:NTU-Taida-Suicide-system.png|300px|thumb|left|alt=new idea about safety|general scheme of suicide system ]] | ||
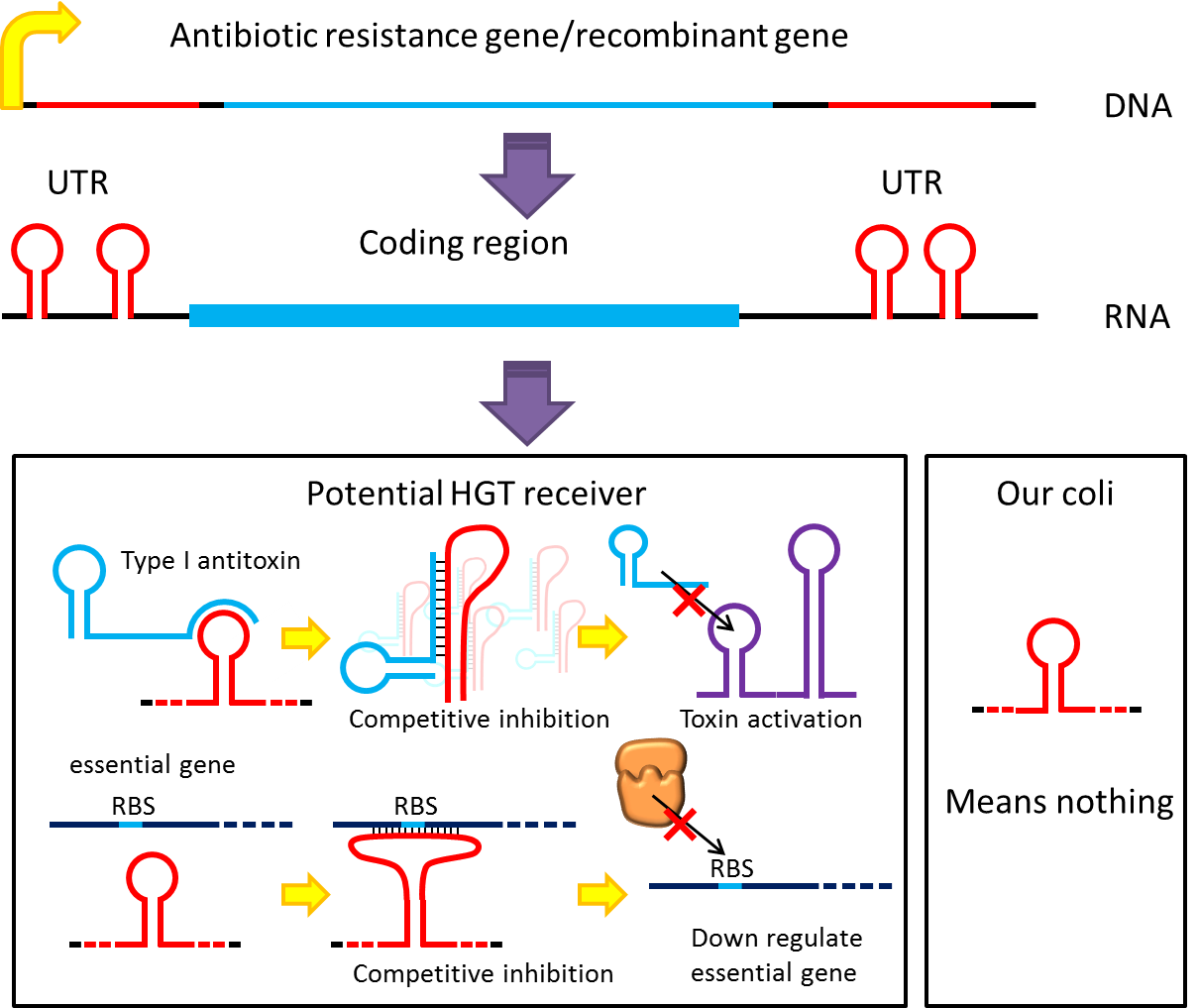
| - | <p style="text-indent: 2em;">However, these designs cannot completely eliminate the risk of horizontal gene transfer (HGT), where recombinant genes can move to other organisms independent of suicide system. Therefore, in addition to suicide system, we propose a new concept to deal with HGT risks by RNA interaction.</p> | + | <p style="text-indent: 2em;">However, these designs cannot completely eliminate the risk of horizontal gene transfer (HGT), where recombinant genes can move to other organisms independent of suicide system. Therefore, in addition to suicide system, we propose a new concept to deal with HGT risks by RNA interaction[[#Ref|[1] ]]. Although bacteria lack RNAi pathway, expressing well designed antisense RNAs have been shown to have inhibitory effect on target RNAs through competitive inhibition.A recent study has demonstrated that expressing peptide nucleic acid (PNA) antisense to antitoxin RNA 5' sequence kills bacteria cells [[#Ref|[2] ]]. We reason that adding rationally designed antisense RNAs on the untranslated region of transcripts may interfere with target RNA’s function or translation, and we can employ this property to prevent HGT. For instance, HGT is more likely to occur between related species, such as lab ''E. coli'' & O157. Laboratory ''E. coli'' have inactivated all its hok/sok toxin-antitoxin system by mutation, but wild bacteria (especially pathogenic bacteria) usually have more active TA locus on its chromosome like ''E. coli'' O157. We plan to put a stem loop from hok mRNA, which can pair with sok RNA, 5’sequence on UTR of antibiotic resistance genes used in our PepdEx system. IF wild bacteria steal our antibiotic resistance genes and express it, its antitoxin will be competitive inhibited, and its toxin will get expressed to kill the thief. This prevents HGT between lab & wild coli. This concept can have wide extension. In addition to targeting antitoxin (functional RNA) of type I TA, it is also possible to design antisense sequences that target RBS to down-regulate targeted protein. Targeting antitoxin of type II TA, essential genes for metabolism, housekeeping genes and any sequences exist in potential HGT receivers but NOT our coli can also be used. Even if the design cannot kill thieves, it can still weaken receivers and reduce the advantages a stolen antibiotic gene may bring in, thus reducing the degree of danger in HGT.</p> |
| - | + | <p style="text-indent: 2em;">A final note: in the past, the repression efficiencies of antisense RNA in bacteria are low. However, after the invention of paired termini antisense RNA (PTasRNA) method, incorporation of U turn/YUNR motif, etc., the concept of competitive inhibition will become more and more feasible for the microbial synthetic biology community[[#Ref|[1 , ]][[#Ref|3 , ]][[#Ref|4] ]].</p> | |
| - | + | ||
| - | <p style="text-indent: 2em;">A final note: in the past, the repression efficiencies of antisense RNA in bacteria are low. However, after the invention of paired termini antisense RNA (PTasRNA) method, incorporation of U turn/YUNR motif, etc., the concept of competitive inhibition will become more and more feasible for the microbial synthetic biology community[[# | + | |
[[File:NTU-Taida-AsRNA.png|700px|center|thumb|alt=new idea about safety|Is it possible to use RNA interaction to avoid HGT? ]] | [[File:NTU-Taida-AsRNA.png|700px|center|thumb|alt=new idea about safety|Is it possible to use RNA interaction to avoid HGT? ]] | ||
| - | === | + | ==Suicide Device== |
| - | <ol> | + | |
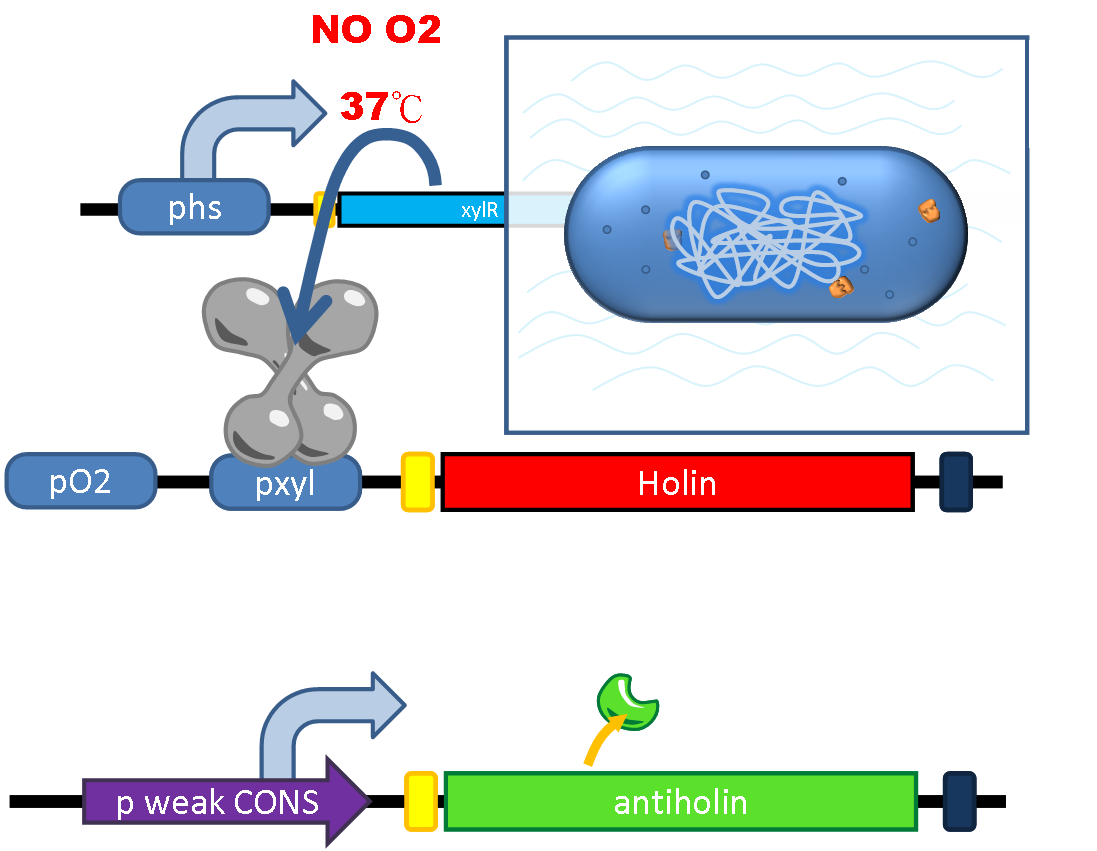
| - | <li | + | In prevention of the occasional escape of PEPDEX from human gut and any undesirable growth and spread in the environment, and for intentional termination of the therapy, we design a suicide device that can be controlled by both '''environmental factors''' ('''temperature''' and '''oxygen level''') and an '''artificial inductive signal (xylose)'''. |
| - | </li><li | + | |
| - | </li><li | + | We incorporate a toxin and anti-toxin system as the killer into our safety device. '''Holin''', a membrane bound protein, can form complex with each other and trigger the disruption of the membrane. '''Anti-holin''', which is bound to the inner membrane, can bind and inactivate Holin. We put Holin under the control of an '''OR gate: double promoter''' formed by an '''oxygen-sensitive promoter P<sub>ox</sub>''' and a '''xylose-sensitive promoter P<sub>xyl</sub>'''. Inside the anaerobic environment of human gut, the oxygen-sensitive promoter P<sub>ox</sub> is repressed by the endogenous '''oxygen sensitive protein FNR (fumarate nitrate reductase regulator)''' of bacteria, while further suppressed is done by '''xylose repressor XlyR''' whose expression is driven by the '''thermal promoter Phs''' (see the [[Team:NTU-Taida/Project/Sensor|Sensor]] section) at the body temperature 37℃. Besides, a weak constitutively expressed Anti-Holin is designed to neutralize the leaky expression of Holin to circumvent the unwanted cellular lysis of the bacteria. |
| - | </li><li | + | |
| + | [[File:NTU-Taida-Project-Suicide 1.png|500px|thumb|center|Bacteria survive under 37℃ in anaerobic environment of gut]] | ||
| + | |||
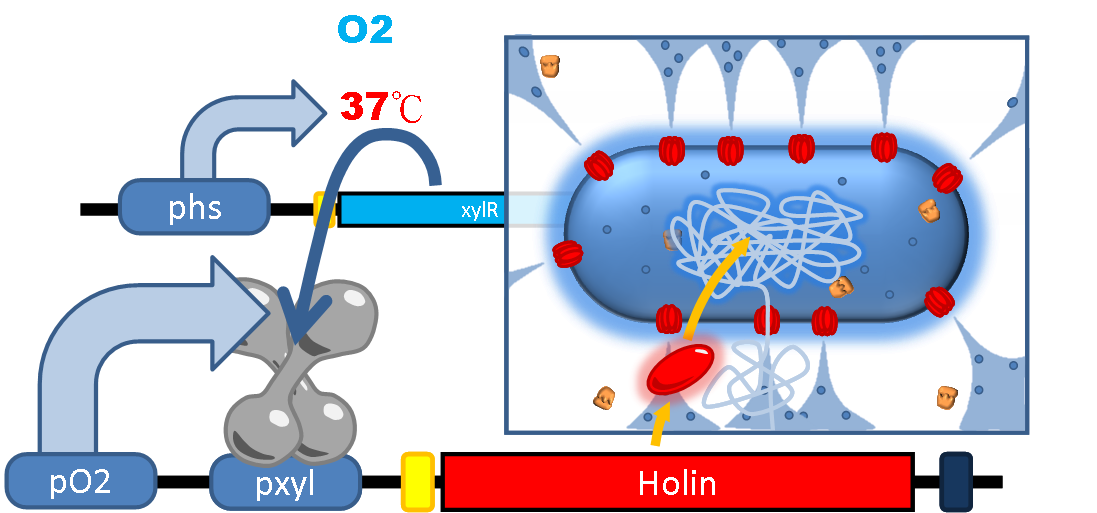
| + | When the bacteria are excreted outside the human gut, the drop in temperature leads to reduced efficiency of Phs and thus decrease the repression level of XlyR. Also if there is any oxygen presented in the environment, FNR will bind to the oxygen molecule and detach from P<sub>ox</sub>. Both of the above situations result in dis-inhibition of Holin expression which kills the bacteria. On the other hand, if we want to terminate the therapy artificially, oral intake of xylose can also induce the detachment of XylR and achieve elimination of the bacteria. Under these security concerns, our PEPDEX can be well controlled and manipulated either automatically by environmental change or intentional induction. | ||
| + | |||
| + | [[File:NTU-Taida-Project-Suicide 2.png|500px|thumb|center|Bacteria die at the presence of oxygen]] | ||
| + | [[File:NTU-Taida-Project-Suicide 3.png|500px|thumb|center|Bacteria die at room temperature]] | ||
| + | [[File:NTU-Taida-Project-Suicide 4.png|500px|thumb|center|Bacteria die under the artificial induction of xylose]] | ||
| + | |||
| + | ==Fecal Microbiota Transplatation== | ||
| + | |||
| + | In addition to the suicide device, '''traditional antibiotics use and fecal microbiota transplantation (FMT)''' can be another option for safety of PEPDEX. Fecal microbiota transplantation has been utilized over decades, originally for treatment of patients with diarrhea and pseudomembanous colitis caused by recalcitrant '''''Clostridium difficile''''' '''infection (CDI)'''. Stool of health donors are preserved and filtrated to produce fecal samples, and those samples can be administrated by different routes: '''enema, transcolonic, nasoduodenal and nasogastic infusion'''. As the roles of gut microbiota in immunity and energy metabolism become more and more clear, this old procedure now has emerging medical application in pathological conditions caused by microbiota-related dysfunction, such as inflammatory bowel diseases, '''inflammatory bowel syndromes, metabolic syndromes, neurodevelopmental disorders, autoimmune diseases, and allergic diseases'''. | ||
| + | If the population of PEPDEX loses balance around the microbial community inside the intestine of the hosts, running out of control and causing some unwanted effects, we can adopt traditional broad-spectrum use of antibiotics for eradication of these bacteria, and then perform fecal microbiota transplantation in order to rebuild the normal microbial communities of guts. | ||
| + | [[File:NTU-Taida-Project-FMT.png|700px|thumb|center|Procedure of fecal microbiota transplantation.]] | ||
| + | |||
| + | ==Reference== | ||
| + | <ol id='Ref'> | ||
| + | <li>Good, L. and J.E. Stach, Synthetic RNA silencing in bacteria - antimicrobial discovery and resistance breaking. Front Microbiol, 2011. 2: p. 185.</li> | ||
| + | <li>Faridani, O.R., et al., Competitive inhibition of natural antisense Sok-RNA interactions activates Hok-mediated cell killing in Escherichia coli. Nucleic Acids Research, 2006. 34(20): p. 5915-22.</li> | ||
| + | <li>Lucks, J.B., et al., Versatile RNA-sensing transcriptional regulators for engineering genetic networks. Proc Natl Acad Sci U S A, 2011. 108(21): p. 8617-22.</li> | ||
| + | <li>Franch, T., et al., Antisense RNA regulation in prokaryotes: rapid RNA/RNA interaction facilitated by a general U-turn loop structure. Journal of Molecular Biology, 1999. 294(5): p. 1115-25.</li> | ||
| + | <li>Taylor, W. E., D. B. Straus, et al. (1984). "Transcription from a heat-inducible promoter causes heat shock regulation of the sigma subunit of E. coli RNA polymerase." Cell 38(2): 371-381. </li> | ||
| + | <li>Reinhart, F., S. Achebach, et al. (2008). "Reduced apo-fumarate nitrate reductase regulator (apoFNR) as the major form of FNR in aerobically growing Escherichia coli." J Bacteriol 190(3): 879-886 </li> | ||
| + | <li>Young, R. (2002). "Bacteriophage holins: deadly diversity." J Mol Microbiol Biotechnol 4(1): 21-36. </li> | ||
| + | <li>Wedler, H. and R. Wambutt (1995). "A temperature-sensitive lambda cI repressor functions on a modified operator in yeast cells by masking the TATA element." Mol Gen Genet 248(4): 499-505 </li> | ||
| + | <li>Borody TJ, et al. (2012) Fecal microbiota transplantation and emerging applications. Nat Rev Gastro & Hepato 9: 88-96.</li> | ||
</ol> | </ol> | ||
<!-- EOF --> | <!-- EOF --> | ||
{{:Team:NTU-Taida/Templates/ContentEnd}}{{:Team:NTU-Taida/Templates/Footer|ActiveNavbar=Project, #nav-Project-Safety}} | {{:Team:NTU-Taida/Templates/ContentEnd}}{{:Team:NTU-Taida/Templates/Footer|ActiveNavbar=Project, #nav-Project-Safety}} | ||
Latest revision as of 02:22, 27 October 2012
Safety
Several safety issues are considered and answered here. To prevent horizontal gene transfer between our PEPDEX and other intestinal microbes, an RNAi-like system is designed to block this process. Also, to avoid undesirable growth and spread of our PEPDEX in the environment, a suicide device will kill the bacteria once they escape from the human body. Furthermore, if the intestinal bacterial populations have dramatic imbalance caused by PEPDEX, we can adopt traditional antibiotics use and perform the fecal microbiota transplantation to re-establish the normal composition of floral communities of the host.
Contents |
Horizontal Gene Transfer Blocker
Many turn off strategies have been developed, most of them are inducible suicide systems that can be activated at certain conditions. In our project, we plan to use temperature as the activating signal (or alternatively, small molecules). When the course of treatment ends, leaving human body (temperature drop from 37C to room temperature) or administration of small amount of drugs (e.g. tetracycline) will cause suicide gene activation, thereby avoiding recombinant strain/gene pollution. And splitting suicide system to provide repression in trans can prevent plasmid transferring to wild type strains. Many off-the-rack parts can be used.
However, these designs cannot completely eliminate the risk of horizontal gene transfer (HGT), where recombinant genes can move to other organisms independent of suicide system. Therefore, in addition to suicide system, we propose a new concept to deal with HGT risks by RNA interaction[1] . Although bacteria lack RNAi pathway, expressing well designed antisense RNAs have been shown to have inhibitory effect on target RNAs through competitive inhibition.A recent study has demonstrated that expressing peptide nucleic acid (PNA) antisense to antitoxin RNA 5' sequence kills bacteria cells [2] . We reason that adding rationally designed antisense RNAs on the untranslated region of transcripts may interfere with target RNA’s function or translation, and we can employ this property to prevent HGT. For instance, HGT is more likely to occur between related species, such as lab E. coli & O157. Laboratory E. coli have inactivated all its hok/sok toxin-antitoxin system by mutation, but wild bacteria (especially pathogenic bacteria) usually have more active TA locus on its chromosome like E. coli O157. We plan to put a stem loop from hok mRNA, which can pair with sok RNA, 5’sequence on UTR of antibiotic resistance genes used in our PepdEx system. IF wild bacteria steal our antibiotic resistance genes and express it, its antitoxin will be competitive inhibited, and its toxin will get expressed to kill the thief. This prevents HGT between lab & wild coli. This concept can have wide extension. In addition to targeting antitoxin (functional RNA) of type I TA, it is also possible to design antisense sequences that target RBS to down-regulate targeted protein. Targeting antitoxin of type II TA, essential genes for metabolism, housekeeping genes and any sequences exist in potential HGT receivers but NOT our coli can also be used. Even if the design cannot kill thieves, it can still weaken receivers and reduce the advantages a stolen antibiotic gene may bring in, thus reducing the degree of danger in HGT.
A final note: in the past, the repression efficiencies of antisense RNA in bacteria are low. However, after the invention of paired termini antisense RNA (PTasRNA) method, incorporation of U turn/YUNR motif, etc., the concept of competitive inhibition will become more and more feasible for the microbial synthetic biology community[1 , 3 , 4] .
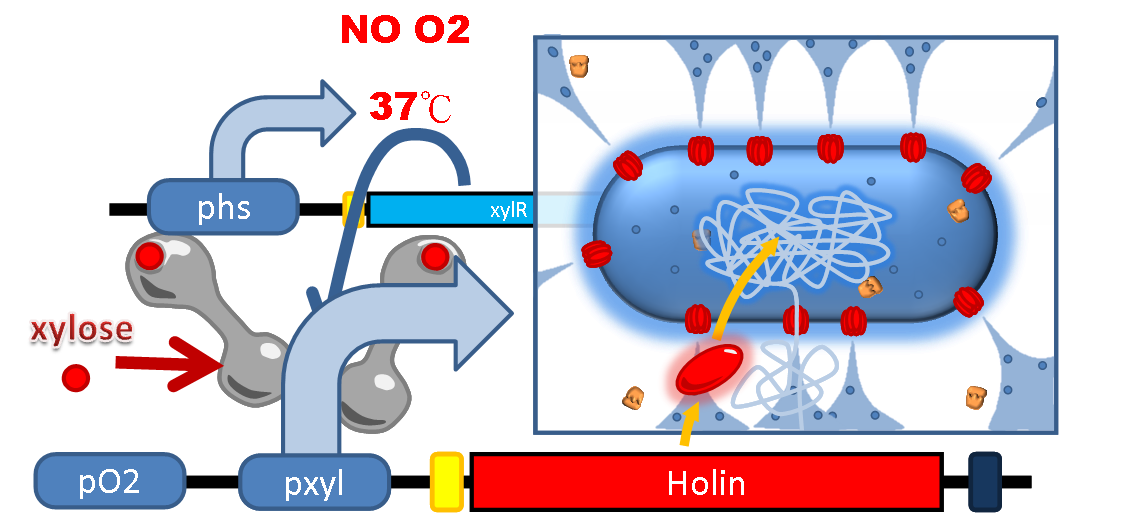
Suicide Device
In prevention of the occasional escape of PEPDEX from human gut and any undesirable growth and spread in the environment, and for intentional termination of the therapy, we design a suicide device that can be controlled by both environmental factors (temperature and oxygen level) and an artificial inductive signal (xylose).
We incorporate a toxin and anti-toxin system as the killer into our safety device. Holin, a membrane bound protein, can form complex with each other and trigger the disruption of the membrane. Anti-holin, which is bound to the inner membrane, can bind and inactivate Holin. We put Holin under the control of an OR gate: double promoter formed by an oxygen-sensitive promoter Pox and a xylose-sensitive promoter Pxyl. Inside the anaerobic environment of human gut, the oxygen-sensitive promoter Pox is repressed by the endogenous oxygen sensitive protein FNR (fumarate nitrate reductase regulator) of bacteria, while further suppressed is done by xylose repressor XlyR whose expression is driven by the thermal promoter Phs (see the Sensor section) at the body temperature 37℃. Besides, a weak constitutively expressed Anti-Holin is designed to neutralize the leaky expression of Holin to circumvent the unwanted cellular lysis of the bacteria.
When the bacteria are excreted outside the human gut, the drop in temperature leads to reduced efficiency of Phs and thus decrease the repression level of XlyR. Also if there is any oxygen presented in the environment, FNR will bind to the oxygen molecule and detach from Pox. Both of the above situations result in dis-inhibition of Holin expression which kills the bacteria. On the other hand, if we want to terminate the therapy artificially, oral intake of xylose can also induce the detachment of XylR and achieve elimination of the bacteria. Under these security concerns, our PEPDEX can be well controlled and manipulated either automatically by environmental change or intentional induction.
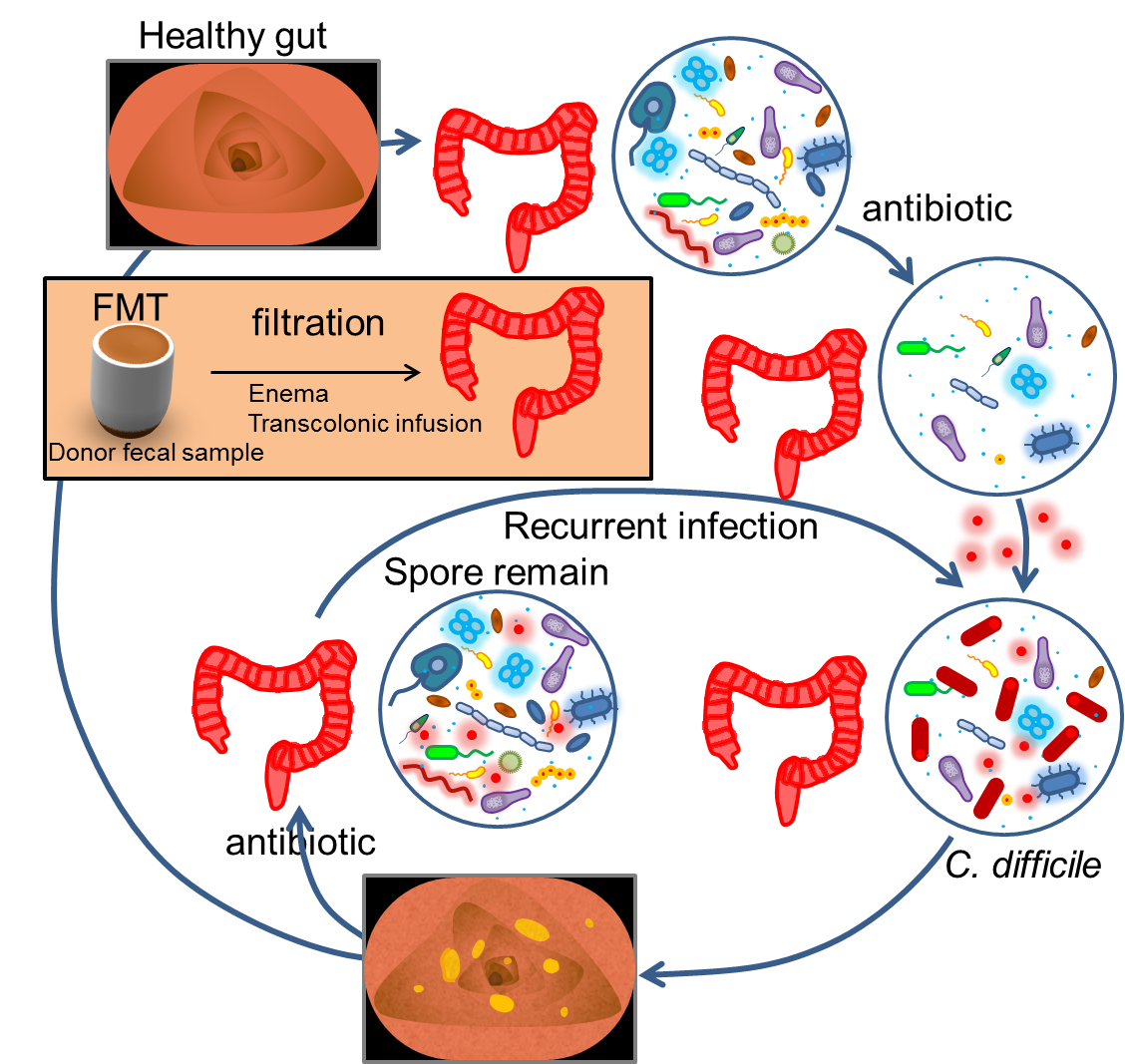
Fecal Microbiota Transplatation
In addition to the suicide device, traditional antibiotics use and fecal microbiota transplantation (FMT) can be another option for safety of PEPDEX. Fecal microbiota transplantation has been utilized over decades, originally for treatment of patients with diarrhea and pseudomembanous colitis caused by recalcitrant Clostridium difficile infection (CDI). Stool of health donors are preserved and filtrated to produce fecal samples, and those samples can be administrated by different routes: enema, transcolonic, nasoduodenal and nasogastic infusion. As the roles of gut microbiota in immunity and energy metabolism become more and more clear, this old procedure now has emerging medical application in pathological conditions caused by microbiota-related dysfunction, such as inflammatory bowel diseases, inflammatory bowel syndromes, metabolic syndromes, neurodevelopmental disorders, autoimmune diseases, and allergic diseases. If the population of PEPDEX loses balance around the microbial community inside the intestine of the hosts, running out of control and causing some unwanted effects, we can adopt traditional broad-spectrum use of antibiotics for eradication of these bacteria, and then perform fecal microbiota transplantation in order to rebuild the normal microbial communities of guts.
Reference
- Good, L. and J.E. Stach, Synthetic RNA silencing in bacteria - antimicrobial discovery and resistance breaking. Front Microbiol, 2011. 2: p. 185.
- Faridani, O.R., et al., Competitive inhibition of natural antisense Sok-RNA interactions activates Hok-mediated cell killing in Escherichia coli. Nucleic Acids Research, 2006. 34(20): p. 5915-22.
- Lucks, J.B., et al., Versatile RNA-sensing transcriptional regulators for engineering genetic networks. Proc Natl Acad Sci U S A, 2011. 108(21): p. 8617-22.
- Franch, T., et al., Antisense RNA regulation in prokaryotes: rapid RNA/RNA interaction facilitated by a general U-turn loop structure. Journal of Molecular Biology, 1999. 294(5): p. 1115-25.
- Taylor, W. E., D. B. Straus, et al. (1984). "Transcription from a heat-inducible promoter causes heat shock regulation of the sigma subunit of E. coli RNA polymerase." Cell 38(2): 371-381.
- Reinhart, F., S. Achebach, et al. (2008). "Reduced apo-fumarate nitrate reductase regulator (apoFNR) as the major form of FNR in aerobically growing Escherichia coli." J Bacteriol 190(3): 879-886
- Young, R. (2002). "Bacteriophage holins: deadly diversity." J Mol Microbiol Biotechnol 4(1): 21-36.
- Wedler, H. and R. Wambutt (1995). "A temperature-sensitive lambda cI repressor functions on a modified operator in yeast cells by masking the TATA element." Mol Gen Genet 248(4): 499-505
- Borody TJ, et al. (2012) Fecal microbiota transplantation and emerging applications. Nat Rev Gastro & Hepato 9: 88-96.
 "
"