Team:HokkaidoU Japan/Notebook/aggregation Week 6
From 2012.igem.org
| (28 intermediate revisions not shown) | |||
| Line 4: | Line 4: | ||
<!-- DO NOT EDIT OVER THIS LINE @iTakeshi --> | <!-- DO NOT EDIT OVER THIS LINE @iTakeshi --> | ||
<div class="hokkaidou-notebook-daily"> | <div class="hokkaidou-notebook-daily"> | ||
| - | ==August 6th== | + | ===August 6th=== |
| - | <div> | + | <div class="hokkaidou-section"> |
| - | ==Ethanol precipitation== | + | ====Ethanol precipitation==== |
| - | + | ||
Ethanol precipitation for digestion and gel extraction product. | Ethanol precipitation for digestion and gel extraction product. | ||
#Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol. | #Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol. | ||
| - | #Centrifuged | + | #Centrifuged at 15000 rpm, 10 min at 4C. |
| - | # | + | #Removed supernatant and added 220 ul of 70% ethanol. |
| - | #Centrifuged | + | #Centrifuged at 15000 rpm, 10 min at 4C. |
| - | # | + | #Removed supernatant and dried out at room temperature, after that added 10 ul of DW. |
| - | + | ||
| - | ==Ligation== | + | ====Ligation==== |
| - | + | ||
We ligated pT7-RBS on pSB1K3 (digested Spe1) as vector and Ag43-dT (digested Spe1 and Xba1 and HindIII) as insert. | We ligated pT7-RBS on pSB1K3 (digested Spe1) as vector and Ag43-dT (digested Spe1 and Xba1 and HindIII) as insert. | ||
| - | |||
{|class="hokkaidou-table-ligation" | {|class="hokkaidou-table-ligation" | ||
| Line 38: | Line 34: | ||
|10 ul | |10 ul | ||
|} | |} | ||
| - | |||
Ligation reaction time was written below. | Ligation reaction time was written below. | ||
| - | |||
{|class="hokkaidou-table-ligation" | {|class="hokkaidou-table-ligation" | ||
|- | |- | ||
| Line 56: | Line 50: | ||
|Hold | |Hold | ||
|} | |} | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | == | + | ====Plasmid extraction==== |
| - | + | Plasmid extraction of pBad-RBS. We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions. | |
| - | Transformation of plasmid DNA (pT7-RBS-Ag43-dT on pSB1K3) | + | [[image:HokkaidoU2012_120806_pBad-rbs.jpg|thumb|Plasmid extraction result]] |
| + | |||
| + | ====Transformation==== | ||
| + | Transformation of plasmid DNA (pT7-RBS-Ag43-dT on pSB1K3) into DH5α. | ||
#Added 2 ul of plasmid DNA to 50 ul of thawed competent cells on ice. | #Added 2 ul of plasmid DNA to 50 ul of thawed competent cells on ice. | ||
#Incubated on ice for 30 min. | #Incubated on ice for 30 min. | ||
| Line 75: | Line 66: | ||
#Incubated them at 37C for 16 hours. | #Incubated them at 37C for 16 hours. | ||
| - | + | ====Ethanol precipitation==== | |
| - | + | Ethanol precipitation for plasmid extraction product (pBad-RBS). Because the refine of plasmid extraction product is not enough. <br /> | |
| - | ==Ethanol precipitation== | + | We used 15 ul of all solution. |
| - | + | ||
| - | Ethanol precipitation for | + | |
| - | We | + | |
#Added 1.5 ul of NaoAc(3 M), 1.5 ul of glycogen and 38 ul of 100% ethanol. | #Added 1.5 ul of NaoAc(3 M), 1.5 ul of glycogen and 38 ul of 100% ethanol. | ||
| - | #Centrifuged | + | #Centrifuged at 15000 rpm, 10 min at 4C. |
| - | # | + | #Removed supernatant and added 220 ul of 70% ethanol. |
| - | #Centrifuged | + | #Centrifuged at 15000 rpm, 5 min at 4C. |
| - | # | + | #Removed supernatant and dried out at room temperature, after that added 10 ul of DW. |
| - | + | ||
| - | == | + | ====Digestion==== |
| - | + | Digestion of ethanol precipitation product(pBad-RBS). | |
| - | + | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| Line 108: | Line 94: | ||
|10 ul | |10 ul | ||
|} | |} | ||
| - | |||
| - | |||
| - | + | [[image:HokkaidoU2012_120807_pbad-rbs-digested.jpg|thumb|digestion result]] | |
| + | From this result, speI digested DNA completely.<br /> | ||
So I think that the pT7-RBS on pSB1K3 DNA solution is not pure. | So I think that the pT7-RBS on pSB1K3 DNA solution is not pure. | ||
</div></div> | </div></div> | ||
| - | |||
<div class="hokkaidou-notebook-daily"> | <div class="hokkaidou-notebook-daily"> | ||
| - | ==August 7th== | + | ===August 7th=== |
| - | <div> | + | <div class="hokkaidou-section"> |
| - | ==Gel extraction== | + | ====Gel extraction==== |
| - | + | ||
Gel extraction for digestion product. We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50 ul of DNA solution. | Gel extraction for digestion product. We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50 ul of DNA solution. | ||
| - | |||
| - | ==Ethanol precipitation== | + | ====Ethanol precipitation==== |
| - | + | ||
Ethanol precipitation for digestion and gel extraction product. | Ethanol precipitation for digestion and gel extraction product. | ||
#Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol. | #Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol. | ||
| - | #Centrifuged | + | #Centrifuged at 15000 rpm, 10 min at 4C. |
| - | # | + | #Removed supernatant and added 220 ul of 70% ethanol. |
| - | #Centrifuged | + | #Centrifuged at 15000 rpm, 10 min at 4C. |
| - | # | + | #Removed supernatant and dried out at room temperature, after that added 10 ul of DW. |
| - | + | ||
| - | ==Colony PCR== | + | ====Colony PCR==== |
| - | + | ||
Colony PCR to confirm that whether the Ag43-dT was successfully ligated with pT7-RBS on pSB1K3 or not. | Colony PCR to confirm that whether the Ag43-dT was successfully ligated with pT7-RBS on pSB1K3 or not. | ||
| + | {|class="hokkaidou-table-pcr-reagent" | ||
| + | |- | ||
| + | |DNA solution | ||
| + | |4 ul (1colony/10 ul DW) | ||
| + | |- | ||
| + | |Kapa-Taq(Taq polymerase) | ||
| + | |5 ul | ||
| + | |- | ||
| + | |Forward Primer(Ag43-f4 primer) | ||
| + | |0.5 ul | ||
| + | |- | ||
| + | |Reverse Primer(PS-R primer) | ||
| + | |0.5 ul | ||
| + | |- | ||
| + | |Total | ||
| + | |10 ul | ||
| + | |} | ||
| + | {|class="hokkaidou-table-pcr-time" | ||
| + | |- | ||
| + | |Number | ||
| + | |Degree | ||
| + | |Second | ||
| + | |- | ||
| + | |1 | ||
| + | |95 | ||
| + | |120 | ||
| + | |- | ||
| + | |2 | ||
| + | |95 | ||
| + | |30 | ||
| + | |- | ||
| + | |3 | ||
| + | |53 | ||
| + | |30 | ||
| + | |- | ||
| + | |4 | ||
| + | |72 | ||
| + | |60 | ||
| + | |- | ||
| + | |5 | ||
| + | |72 | ||
| + | |60 | ||
| + | |- | ||
| + | |6 | ||
| + | |4 | ||
| + | |HOLD | ||
| + | |} | ||
| + | Cycle:2~4 x 35 | ||
| + | |||
| + | We used N1 (DW only) and N2 (Ag43 on pSB1C3 (K346007)) as controls. | ||
| + | Desired product is about 500~600bp. | ||
| + | [[image:HokkaidoU2012_120807_colonyPCR.jpg|thumb|Colony PCR result]] | ||
| + | We thought that colonies of No. 1 and 2 and 5 and 11 and 14 have deep band. | ||
| + | Next step, we resuspended these 5 colonies and cultured (add 1700 ul LB and 2 ul Amp) for 15 hours at 37C. | ||
| + | </div></div> | ||
| + | |||
| + | <div class="hokkaidou-notebook-daily"> | ||
| + | ===August 8th=== | ||
| + | <div class="hokkaidou-section"> | ||
| + | ====Plasmid extraction==== | ||
| + | Plasmid extraction of colony No. 1 and 14. We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions. | ||
| + | [[image:HokkaidoU2012_120808_pt7-rbs-ag43-dt.jpg|thumb|Plasmid extraction result]] | ||
| + | |||
| + | And then, we did plasmid extraction for colony No. 2,5,11. The results of them are the same. | ||
| + | |||
| + | ====Sequencing==== | ||
| + | PCR for sequencing. | ||
| + | {| | ||
| + | |DNA | ||
| + | |primer | ||
| + | |- | ||
| + | |Ag43 plasmid extraction product | ||
| + | |100bp-up forward primer, | ||
| + | |ag43-f1, | ||
| + | |ag43-f2, | ||
| + | |ag43-f3, | ||
| + | |ag43-f4 | ||
| + | |- | ||
| + | |K542009 | ||
| + | |100bp-up forward primer, | ||
| + | |ag43-f1, | ||
| + | |ag43-f2, | ||
| + | |ag43-f3, | ||
| + | |ag43-f4 | ||
| + | |- | ||
| + | |pT7-RBS on pSB1K3 | ||
| + | |100bp-up forward primer | ||
| + | |- | ||
| + | |pT7-RBS on pSB1C3 | ||
| + | |100bp-up forward primer | ||
| + | |- | ||
| + | |Ag43-dT on pSB1AK3 | ||
| + | |100bp-up forward primer, | ||
| + | |ag43-f1, | ||
| + | |ag43-f2, | ||
| + | |ag43-f3, | ||
| + | |ag43-f4 | ||
| + | |- | ||
| + | |Ag43-dT on pSB1T3 | ||
| + | |100bp-up forward primer, | ||
| + | |ag43-f1, | ||
| + | |ag43-f2, | ||
| + | |ag43-f3, | ||
| + | |ag43-f4 | ||
| + | |- | ||
| + | |pT7-RBS on pSB1K3 | ||
| + | |100bp-up forward primer | ||
| + | |} | ||
| + | |||
| + | |||
| + | Sequencing PCR | ||
| + | {|class="hokkaidou-table-sequencing-pcr-reagent" | ||
| + | |- | ||
| + | |template DNA | ||
| + | |1 ul | ||
| + | |- | ||
| + | |Ready Reaction Premix | ||
| + | |1 ul | ||
| + | |- | ||
| + | |5x Sequencing Buffer | ||
| + | |1.5 ul | ||
| + | |- | ||
| + | |H2O | ||
| + | |5 ul | ||
| + | |- | ||
| + | |Primer(1 pmol/ul) | ||
| + | |1.5 ul | ||
| + | |- | ||
| + | |Total | ||
| + | |10 ul | ||
| + | |} | ||
| + | |||
| + | |||
| + | {|class="hokkaidou-table-pcr-time" | ||
| + | |- | ||
| + | |Number | ||
| + | |Degree | ||
| + | |Second | ||
| + | |- | ||
| + | |1 | ||
| + | |96 | ||
| + | |10 | ||
| + | |- | ||
| + | |2 | ||
| + | |50 | ||
| + | |5 | ||
| + | |- | ||
| + | |3 | ||
| + | |60 | ||
| + | |240 | ||
| + | |- | ||
| + | |4 | ||
| + | |4 | ||
| + | |HOLD | ||
| + | |} | ||
| + | Cycle:1~3 x 25 | ||
| + | |||
| + | |||
| + | To purify the PCR product, we did ethanol precipitation. | ||
| + | Ethanol precipitation for sequencing. | ||
| + | #Added 10 ul of H2O, 2 ul of NaoAc, 1 ul of glycogen and 50 ul of 100% ethanol. | ||
| + | #Centrifuged at 15000 rpm, 15 min at 26C. | ||
| + | #Removed supernatant and added 100ul of 70% ethanol. | ||
| + | #Centrifuged at 15000 rpm, 5 min at 26C. | ||
| + | #Removed supernatant and dried out at room temperature, after that added 10 ul of DW. | ||
| + | |||
| + | Then ran a sequencing machine (ByoDye Terminator v3.1 Cycle Sequencing Kit) | ||
| + | </div></div> | ||
| + | |||
| + | <div class="hokkaidou-notebook-daily"> | ||
| + | ===August 9th=== | ||
| + | <div class="hokkaidou-section"> | ||
| + | ====Ligation==== | ||
| + | We ligated pT7-RBS on pSB1K3 (digested Spe1) as vector and Ag43-dT (digested Spe1 and Xba1 and HindIII) as insert.<br /> | ||
| + | We did ligation (pT7-RBS on pSB1K3 and Ag43-dT) and transformation it, at August 6th. | ||
| + | However, we could not get the target plasmid. (pT7-RBS-Ag43-dT on pSB1K3) | ||
| + | So, we did ligation by using more Insert DNA part. | ||
| + | |||
| + | {|class="hokkaidou-table-ligation" | ||
| + | |- | ||
| + | |pT7-RBS | ||
| + | |1 ul | ||
| + | |- | ||
| + | |Ag43-dT | ||
| + | |4 ul | ||
| + | |- | ||
| + | |Ligation Mighty Mix(TAKARA) | ||
| + | |5 ul | ||
| + | |- | ||
| + | |Total | ||
| + | |10 ul | ||
| + | |} | ||
| + | |||
| + | |||
| + | Ligation reaction time was written below. | ||
| + | {|class="hokkaidou-table-ligation" | ||
| + | |- | ||
| + | |Degree | ||
| + | |Minute | ||
| + | |- | ||
| + | |16 | ||
| + | |30 | ||
| + | |- | ||
| + | |65 | ||
| + | |10 | ||
| + | |- | ||
| + | |4 | ||
| + | |Hold | ||
| + | |} | ||
| + | |||
| + | ====Transformation==== | ||
| + | Transformation of plasmid DNA ligation product (pT7-RBS-Ag43-dT on pSB1K3) into DH5α.<br /> | ||
| + | Transformation of August 6th, we found only 14 colonies. So this time, we use 3 ul of DNA solution for transformation. | ||
| + | #Added 3 ul of plasmid DNA to 50 ul of thawed competent cells on ice. | ||
| + | #Incubated on ice for 30 min. | ||
| + | #Added 400 ul of LB then incubated the cells for 2 hours at 37C. | ||
| + | #Prepared and Labeled two LBK plates. | ||
| + | #Plated 300 ul of the culture onto first dish and spread. | ||
| + | #Added 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread. | ||
| + | #Incubated them at 37C for 16 hours. | ||
| + | </div></div> | ||
| + | |||
| + | <div class="hokkaidou-notebook-daily"> | ||
| + | ===August 11st=== | ||
| + | <div class="hokkaidou-section"> | ||
| + | ====Colony PCR==== | ||
| + | Colony PCR to confirm that whether the Ag43-dT was successfully ligated with pT7-RBS on pSB1K3 or not. | ||
{|class="hokkaidou-table-pcr-reagent" | {|class="hokkaidou-table-pcr-reagent" | ||
|- | |- | ||
| Line 190: | Line 396: | ||
Cycle:2~4 x 35 | Cycle:2~4 x 35 | ||
| - | We used N1 (DW only) and N2 (Ag43 on | + | We used N1 (DW only) and N2 (Ag43-dT on pSB1AK3) as controls. |
Desired product is about 500~600bp. | Desired product is about 500~600bp. | ||
| + | [[image:HokkaidoU1120811 coloP.jpg|thumb|Colony PCR result]] | ||
| - | + | We thought that colonies of No. 6 and 8 is like N2 band. | |
| + | Next step, we resuspended these 2 colonies and cultured (add 1700 ul LB and 2 ul Amp) for 16 hours at 37C. | ||
| + | </div></div> | ||
| - | </div><div> | + | <div class="hokkaidou-notebook-daily"> |
| + | ===August 12nd=== | ||
| + | <div class="hokkaidou-section"> | ||
| + | ====Plasmid extraction==== | ||
| + | Plasmid extraction of No. 6 and 8. We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions. | ||
| + | [[image:HokkaidoU2012_120812_pt7-dt.jpg|thumb|plasmid extraction result]] | ||
| + | </div></div> | ||
<!-- DO NOT EDIT UNDER THIS LINE @iTakeshi --> | <!-- DO NOT EDIT UNDER THIS LINE @iTakeshi --> | ||
Latest revision as of 02:26, 27 September 2012
Contents |
August 6th
Ethanol precipitation
Ethanol precipitation for digestion and gel extraction product.
- Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol.
- Centrifuged at 15000 rpm, 10 min at 4C.
- Removed supernatant and added 220 ul of 70% ethanol.
- Centrifuged at 15000 rpm, 10 min at 4C.
- Removed supernatant and dried out at room temperature, after that added 10 ul of DW.
Ligation
We ligated pT7-RBS on pSB1K3 (digested Spe1) as vector and Ag43-dT (digested Spe1 and Xba1 and HindIII) as insert.
| pT7-RBS | 1 ul |
| Ag43-dT | 2 ul |
| DW | 2 ul |
| Ligation Mighty Mix(TAKARA) | 5 ul |
| Total | 10 ul |
Ligation reaction time was written below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
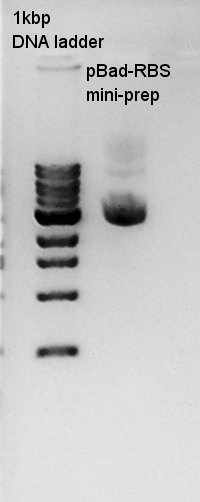
Plasmid extraction
Plasmid extraction of pBad-RBS. We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions.
Transformation
Transformation of plasmid DNA (pT7-RBS-Ag43-dT on pSB1K3) into DH5α.
- Added 2 ul of plasmid DNA to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Added 600 ul of LB then incubated the cells for 2 hours at 37C.
- Prepared and Labeled two LBK plates.
- Plated 300 ul of the culture onto first dish and spread.
- Added 900 ul of LB to 100 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated them at 37C for 16 hours.
Ethanol precipitation
Ethanol precipitation for plasmid extraction product (pBad-RBS). Because the refine of plasmid extraction product is not enough.
We used 15 ul of all solution.
- Added 1.5 ul of NaoAc(3 M), 1.5 ul of glycogen and 38 ul of 100% ethanol.
- Centrifuged at 15000 rpm, 10 min at 4C.
- Removed supernatant and added 220 ul of 70% ethanol.
- Centrifuged at 15000 rpm, 5 min at 4C.
- Removed supernatant and dried out at room temperature, after that added 10 ul of DW.
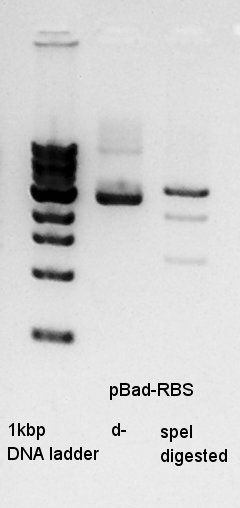
Digestion
Digestion of ethanol precipitation product(pBad-RBS).
| DNA solution | 3 ul |
| SpeI | 1 ul |
| 10xM buffer | 1 ul |
| DW | 5 ul |
| Total | 10 ul |
From this result, speI digested DNA completely.
So I think that the pT7-RBS on pSB1K3 DNA solution is not pure.
August 7th
Gel extraction
Gel extraction for digestion product. We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50 ul of DNA solution.
Ethanol precipitation
Ethanol precipitation for digestion and gel extraction product.
- Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol.
- Centrifuged at 15000 rpm, 10 min at 4C.
- Removed supernatant and added 220 ul of 70% ethanol.
- Centrifuged at 15000 rpm, 10 min at 4C.
- Removed supernatant and dried out at room temperature, after that added 10 ul of DW.
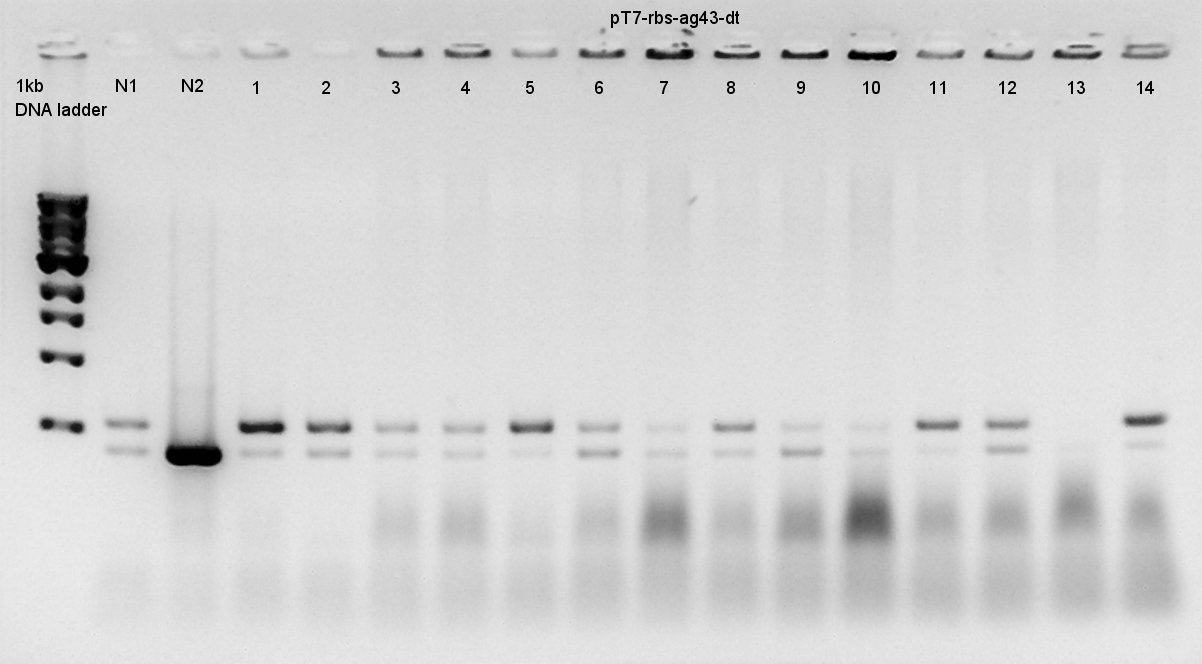
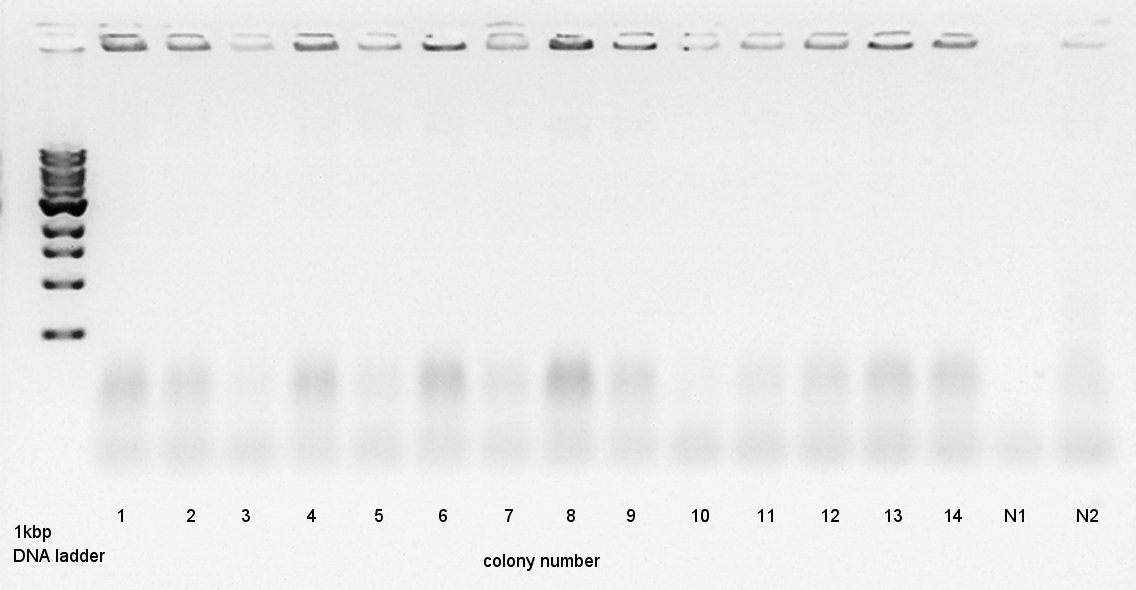
Colony PCR
Colony PCR to confirm that whether the Ag43-dT was successfully ligated with pT7-RBS on pSB1K3 or not.
| DNA solution | 4 ul (1colony/10 ul DW) |
| Kapa-Taq(Taq polymerase) | 5 ul |
| Forward Primer(Ag43-f4 primer) | 0.5 ul |
| Reverse Primer(PS-R primer) | 0.5 ul |
| Total | 10 ul |
| Number | Degree | Second |
| 1 | 95 | 120 |
| 2 | 95 | 30 |
| 3 | 53 | 30 |
| 4 | 72 | 60 |
| 5 | 72 | 60 |
| 6 | 4 | HOLD |
Cycle:2~4 x 35
We used N1 (DW only) and N2 (Ag43 on pSB1C3 (K346007)) as controls. Desired product is about 500~600bp.
We thought that colonies of No. 1 and 2 and 5 and 11 and 14 have deep band. Next step, we resuspended these 5 colonies and cultured (add 1700 ul LB and 2 ul Amp) for 15 hours at 37C.
August 8th
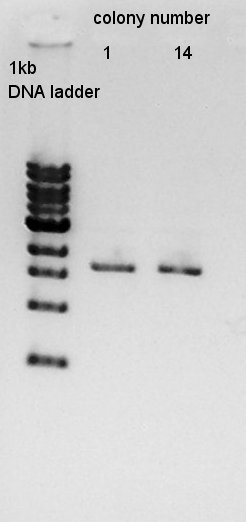
Plasmid extraction
Plasmid extraction of colony No. 1 and 14. We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions.
And then, we did plasmid extraction for colony No. 2,5,11. The results of them are the same.
Sequencing
PCR for sequencing.
| DNA | primer | ||||
| Ag43 plasmid extraction product | 100bp-up forward primer, | ag43-f1, | ag43-f2, | ag43-f3, | ag43-f4 |
| K542009 | 100bp-up forward primer, | ag43-f1, | ag43-f2, | ag43-f3, | ag43-f4 |
| pT7-RBS on pSB1K3 | 100bp-up forward primer | ||||
| pT7-RBS on pSB1C3 | 100bp-up forward primer | ||||
| Ag43-dT on pSB1AK3 | 100bp-up forward primer, | ag43-f1, | ag43-f2, | ag43-f3, | ag43-f4 |
| Ag43-dT on pSB1T3 | 100bp-up forward primer, | ag43-f1, | ag43-f2, | ag43-f3, | ag43-f4 |
| pT7-RBS on pSB1K3 | 100bp-up forward primer |
Sequencing PCR
| template DNA | 1 ul |
| Ready Reaction Premix | 1 ul |
| 5x Sequencing Buffer | 1.5 ul |
| H2O | 5 ul |
| Primer(1 pmol/ul) | 1.5 ul |
| Total | 10 ul |
| Number | Degree | Second |
| 1 | 96 | 10 |
| 2 | 50 | 5 |
| 3 | 60 | 240 |
| 4 | 4 | HOLD |
Cycle:1~3 x 25
To purify the PCR product, we did ethanol precipitation.
Ethanol precipitation for sequencing.
- Added 10 ul of H2O, 2 ul of NaoAc, 1 ul of glycogen and 50 ul of 100% ethanol.
- Centrifuged at 15000 rpm, 15 min at 26C.
- Removed supernatant and added 100ul of 70% ethanol.
- Centrifuged at 15000 rpm, 5 min at 26C.
- Removed supernatant and dried out at room temperature, after that added 10 ul of DW.
Then ran a sequencing machine (ByoDye Terminator v3.1 Cycle Sequencing Kit)
August 9th
Ligation
We ligated pT7-RBS on pSB1K3 (digested Spe1) as vector and Ag43-dT (digested Spe1 and Xba1 and HindIII) as insert.
We did ligation (pT7-RBS on pSB1K3 and Ag43-dT) and transformation it, at August 6th.
However, we could not get the target plasmid. (pT7-RBS-Ag43-dT on pSB1K3)
So, we did ligation by using more Insert DNA part.
| pT7-RBS | 1 ul |
| Ag43-dT | 4 ul |
| Ligation Mighty Mix(TAKARA) | 5 ul |
| Total | 10 ul |
Ligation reaction time was written below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
Transformation
Transformation of plasmid DNA ligation product (pT7-RBS-Ag43-dT on pSB1K3) into DH5α.
Transformation of August 6th, we found only 14 colonies. So this time, we use 3 ul of DNA solution for transformation.
- Added 3 ul of plasmid DNA to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Added 400 ul of LB then incubated the cells for 2 hours at 37C.
- Prepared and Labeled two LBK plates.
- Plated 300 ul of the culture onto first dish and spread.
- Added 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated them at 37C for 16 hours.
August 11st
Colony PCR
Colony PCR to confirm that whether the Ag43-dT was successfully ligated with pT7-RBS on pSB1K3 or not.
| DNA solution | 4 ul (1colony/10 ul DW) |
| Kapa-Taq(Taq polymerase) | 5 ul |
| Forward Primer(Ag43-f4 primer) | 0.5 ul |
| Reverse Primer(PS-R primer) | 0.5 ul |
| Total | 10 ul |
| Number | Degree | Second |
| 1 | 95 | 120 |
| 2 | 95 | 30 |
| 3 | 53 | 30 |
| 4 | 72 | 60 |
| 5 | 72 | 60 |
| 6 | 4 | HOLD |
Cycle:2~4 x 35
We used N1 (DW only) and N2 (Ag43-dT on pSB1AK3) as controls. Desired product is about 500~600bp.
We thought that colonies of No. 6 and 8 is like N2 band. Next step, we resuspended these 2 colonies and cultured (add 1700 ul LB and 2 ul Amp) for 16 hours at 37C.
 "
"