Team:HokkaidoU Japan/Notebook/aggregation Week 5
From 2012.igem.org
| Line 16: | Line 16: | ||
<p> | <p> | ||
I think that the E. coli which we use transformation is BL21(DE3)pLysS. | I think that the E. coli which we use transformation is BL21(DE3)pLysS. | ||
| - | So, the E. coli could | + | So, the E. coli could grow on LBC plate.<br /> |
I'm going to do transformation , use DH5α. | I'm going to do transformation , use DH5α. | ||
| Line 40: | Line 40: | ||
#Added 2 ml of LBK into culture tubes. | #Added 2 ml of LBK into culture tubes. | ||
#Resuspended colonies. | #Resuspended colonies. | ||
| - | #Incubated the tubes at 37C for | + | #Incubated the tubes at 37C for 18 hrs. |
</p> | </p> | ||
| Line 51: | Line 51: | ||
==Plasmid extraction== | ==Plasmid extraction== | ||
<p> | <p> | ||
| - | + | Mini-prep of pT7-RBS-Ag43-dT which had been incubated from yesterday. We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions. | |
[[image:HokkaidoU2012 120801 pt7-rbs-ag43-dt.jpg|thumb|mini-prep result]] | [[image:HokkaidoU2012 120801 pt7-rbs-ag43-dt.jpg|thumb|mini-prep result]] | ||
</p> | </p> | ||
| Line 57: | Line 57: | ||
==Digestion== | ==Digestion== | ||
<p> | <p> | ||
| - | '''pT7-RBS(20 ng/ul) '''=9 | + | '''pT7-RBS(20 ng/ul) '''=No. 9 |
| - | SpeI 10xH | + | SpeI using 10xH |
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| Line 78: | Line 78: | ||
|} | |} | ||
| - | SpeI 10xM | + | SpeI using 10xM |
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| Line 98: | Line 98: | ||
| - | '''pT7-RBS(30 ng/ul) '''=10 | + | '''pT7-RBS(30 ng/ul) '''=No. 10 |
| - | SpeI 10xH | + | SpeI using 10xH |
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| Line 119: | Line 119: | ||
|} | |} | ||
| - | SpeI 10xM | + | SpeI using 10xM |
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| Line 140: | Line 140: | ||
[[image:HokkaidoU2012 120801 pt7-rbs-digS.jpg|thumb|digestion result]] | [[image:HokkaidoU2012 120801 pt7-rbs-digS.jpg|thumb|digestion result]] | ||
| - | We couldn't | + | We couldn't digested them exactly, so we tried to digest once more time. |
| - | '''pT7-RBS(20 ng/ul) '''=9 | + | '''pT7-RBS(20 ng/ul) '''=No. 9 |
SpeI 10xH | SpeI 10xH | ||
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
| Line 163: | Line 163: | ||
</p> | </p> | ||
| + | |||
==Liquid culture== | ==Liquid culture== | ||
<p> | <p> | ||
| Line 183: | Line 184: | ||
Our competent cell Protocol | Our competent cell Protocol | ||
| - | #Single colony isolation on LB plate | + | #Single colony isolation on LB plate. |
| - | # | + | #Incubated the plate for 15-19 hours at 37C. |
| - | # | + | #Lift colony of E. coli into 2 ml LB |
| - | # | + | #Incubate at 37C for 12-16 hours at 180-200 rpm |
| - | # | + | #Transfer 30 ul, 100 ul, 300 ul of the culture into 100 ml of LB. |
| - | # | + | #Incubate cells at 20C (for 24 hours over) at 140 rpm |
| - | # | + | #Select culture by measuring OD 600.(OD 600=0.5) |
| - | # | + | #Incubate the 300 ml flask for 10 min on ice |
| - | # | + | #Transfer the culture into two 50 ml Falcon tube |
| - | # | + | #Centrifuge at 3000 rpm, 4C for 20 min (TOMY LX-120 rotor), and discard sup. |
| - | # | + | #Suspend the pellet in ice-cold 15 ml of TB (Transformation Buffer).(7.5 ml/tube) |
| - | # | + | #Collect them to one tube. |
| - | # | + | #Centrifuge at 3000 rpm, 4C for 20 min (TOMY LX-120 rotor), and discard sup. |
| - | # | + | #Suspend the pellet in ice-cold 3.2 ml of TB. |
| - | # | + | #Instill 0.24 ml of DMSO in precipitant. |
| - | # | + | #Incubate the 50 ml Falcon tube for 10 min on ice. |
| - | # | + | #Divide 50 ul of solutions in each 0.5 ml tubes. |
| - | #Freezed the suspension | + | #Freezed the suspension by liquid nitrogen. |
| - | # | + | #Stored at –80C. |
| Line 208: | Line 209: | ||
==Electrophoresis== | ==Electrophoresis== | ||
<p> | <p> | ||
| - | Electrophoresis of digestion result of | + | Electrophoresis of digestion result of August 1st.(pT7-RBS on pSB1K3 digested by SpeI) |
[[image:HokkaidoU2012 120802 pT7-RBS on pSB1K3 SpeI.jpg|thumb|Electrophoresis result]] | [[image:HokkaidoU2012 120802 pT7-RBS on pSB1K3 SpeI.jpg|thumb|Electrophoresis result]] | ||
| Line 216: | Line 217: | ||
==Digestion== | ==Digestion== | ||
<p> | <p> | ||
| - | Digestion of pT7-RBS on pSB1K3(more fresh one) | + | Digestion of pT7-RBS on pSB1K3 (more fresh one) |
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
|- | |- | ||
| Line 244: | Line 245: | ||
==Electrophoresis== | ==Electrophoresis== | ||
<p> | <p> | ||
| - | Electrophoresis for the result of digestion | + | Electrophoresis for the result of digestion. |
[[image:HokkaidoU2012 120803 pt7-rbs-dspe1.jpg|thumb|Electrophoresis result]] | [[image:HokkaidoU2012 120803 pt7-rbs-dspe1.jpg|thumb|Electrophoresis result]] | ||
</p> | </p> | ||
| Line 311: | Line 312: | ||
#Plate 300 ul of the transformation onto first dish and spread. | #Plate 300 ul of the transformation onto first dish and spread. | ||
#Added 900 ul of LB to 100 ul of the transformation and plated 300 ul of it onto second dish and spread. | #Added 900 ul of LB to 100 ul of the transformation and plated 300 ul of it onto second dish and spread. | ||
| - | #Incubated the plates at 37C for 15 hrs 45 min | + | #Incubated the plates at 37C for 15 hrs 45 min. |
</p> | </p> | ||
| Line 335: | Line 336: | ||
|5 ul | |5 ul | ||
|- | |- | ||
| - | | | + | |Ag43-f4 primer |
|1 ul | |1 ul | ||
|- | |- | ||
| - | | | + | |PS-R primer |
|1 ul | |1 ul | ||
|- | |- | ||
| Line 381: | Line 382: | ||
[[image:HokkaidoU2012 120803 Ag43 PCR(Forward=ag43-f4 Reverse=PS-R).jpg|thumb|PCR result]] | [[image:HokkaidoU2012 120803 Ag43 PCR(Forward=ag43-f4 Reverse=PS-R).jpg|thumb|PCR result]] | ||
| - | From this result, we could see that the ag43-f4 primer had worked and DNA of last 500~600 bp of ag43 to BioBrick suffix were | + | From this result, we could see that the ag43-f4 primer had worked and DNA of last 500~600 bp of ag43 to BioBrick suffix were increased. |
</p> | </p> | ||
| Line 472: | Line 473: | ||
==Plasmid extraction== | ==Plasmid extraction== | ||
<p> | <p> | ||
| - | + | Mini-prep of incubated medium from yesterday. We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions. | |
[[image:HokkaidoU2012 120805 pt7-rbs-ag43-dt.jpg|thumb|mini-prep results]] | [[image:HokkaidoU2012 120805 pt7-rbs-ag43-dt.jpg|thumb|mini-prep results]] | ||
| Line 479: | Line 480: | ||
==Digestion== | ==Digestion== | ||
<p> | <p> | ||
| - | + | Digested pT7-RBS on pSB1K3 and pT7-RBS (once digestioned with SpeI) with speI. | |
| Line 541: | Line 542: | ||
[[image:HokkaidoU2012 120805 pT7-RBS digestion SpeI (no.jpg|thumb|digestion result]] | [[image:HokkaidoU2012 120805 pT7-RBS digestion SpeI (no.jpg|thumb|digestion result]] | ||
| - | From this result the bottom band was misdigested band and middle band was digested band we thought. Was the above band something contaminated in the DNA solution? | + | From this result, the bottom band was misdigested band and middle band was digested band we thought. Was the above band something contaminated in the DNA solution? |
</p> | </p> | ||
==Gel extraction== | ==Gel extraction== | ||
<p> | <p> | ||
| - | Gel extraction of middle band | + | Gel extraction of middle band. We thought this band would be the exactly digested DNA fragment. We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50 ul of DNA solution. |
</p> | </p> | ||
==Sequencing== | ==Sequencing== | ||
<p> | <p> | ||
| - | Sequencing to confirm what kind of DNA we | + | Sequencing to confirm what kind of DNA we made. |
| Line 611: | Line 612: | ||
|5 ul | |5 ul | ||
|- | |- | ||
| - | |Primer( | + | |Primer(1 pmol/ul) |
|1.5 ul | |1.5 ul | ||
|- | |- | ||
| Line 645: | Line 646: | ||
To purify the PCR product, we did ethanol precipitation. | To purify the PCR product, we did ethanol precipitation. | ||
| - | |||
Ethanol precipitation for sequencing. | Ethanol precipitation for sequencing. | ||
| Line 655: | Line 655: | ||
| - | Then run a sequencing machine (ByoDye Terminator v3.1 Cycle Sequencing Kit) | + | Then run a sequencing machine.(ByoDye Terminator v3.1 Cycle Sequencing Kit) |
| - | + | We could not get the sequencing data. | |
</p> | </p> | ||
==Digestion== | ==Digestion== | ||
<p> | <p> | ||
| - | + | Digesting of Ag43-dT (digested by SpeI and XbaI) by HindIII. | |
{|class="hokkaidou-table-digestion" | {|class="hokkaidou-table-digestion" | ||
| Line 684: | Line 684: | ||
[[image:HokkaidoU2012 120805 Ag43-dT on pSB1AK3(d+ with XbaI & SpeI) digestion with HindIII.jpg|thumb|digestion result]] | [[image:HokkaidoU2012 120805 Ag43-dT on pSB1AK3(d+ with XbaI & SpeI) digestion with HindIII.jpg|thumb|digestion result]] | ||
| - | From this result we confirmed that the pSB1AK3 was successfully digested | + | From this result, we confirmed that the pSB1AK3 was successfully digested by HindIII. |
</p> | </p> | ||
Revision as of 13:15, 26 September 2012
July 30th
digestion
I tried to digest the mini-prep products again, and after Ethanol precipitation, we did electrophoresis.
transformation
I think that the E. coli which we use transformation is BL21(DE3)pLysS.
So, the E. coli could grow on LBC plate.
I'm going to do transformation , use DH5α.
Transformation of plasmid DNA ligated at 25th (pT7-RBS-Ag43-dT on pSB1K3) in DH5α.
- Added 2 ul of plasmid DNA to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Added 200 ul of LB then incubated the cells for 2 hours at 37C.
- Prepared and Labeled two petri dishes with LBK.
- Plate 200 ul of the transformation onto first dish and spread.
- Added 450 ul of LB to 50 ul of the transformation and plated 200 ul of it onto second dish and spread.
- Incubated the plates at 37C for 14 hours.
July 31th
liquid culture
Liquid culture for pT7-RBS-Ag43-dT on pSB1K3.
- Added 2 ml of LBK into culture tubes.
- Resuspended colonies.
- Incubated the tubes at 37C for 18 hrs.
August 1st
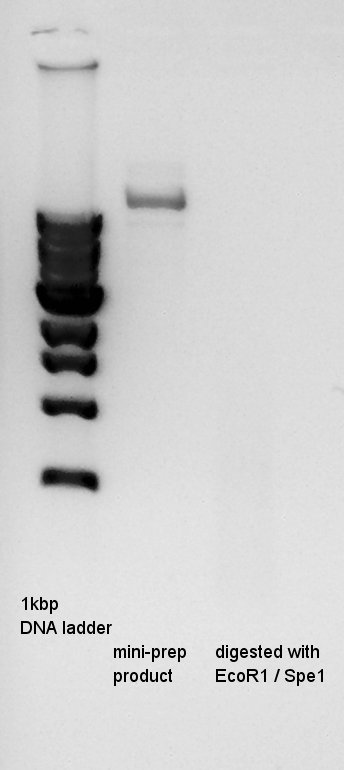
Plasmid extraction
Mini-prep of pT7-RBS-Ag43-dT which had been incubated from yesterday. We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions.
Digestion
pT7-RBS(20 ng/ul) =No. 9 SpeI using 10xH
| DNA solution | 4.5 ul |
| SpeI | 1 ul |
| 10xH buffer | 1 ul |
| DW | 3.5 ul |
| Total | 10 ul |
SpeI using 10xM
| DNA solution | 4.5 ul |
| SpeI | 1 ul |
| 10xM buffer | 1 ul |
| DW | 3.5 ul |
| Total | 10 ul |
pT7-RBS(30 ng/ul) =No. 10
SpeI using 10xH
| DNA solution | 3 ul |
| SpeI | 1 ul |
| 10xH buffer | 1 ul |
| DW | 5 ul |
| Total | 10 ul |
SpeI using 10xM
| DNA solution | 3 ul |
| SpeI | 1 ul |
| 10xM buffer | 1 ul |
| DW | 5 ul |
| Total | 10 ul |
We couldn't digested them exactly, so we tried to digest once more time.
pT7-RBS(20 ng/ul) =No. 9 SpeI 10xH
| DNA solution | 4.5 ul |
| SpeI | 1 ul |
| 10xM buffer | 2 ul |
| DW | 12.5 ul |
| Total | 20 ul |
Liquid culture
Liquid culture for pBAD-RBS on pSB1K3.
- Added 2 ml of LBK into culture tube.
- Scraped the surface of glycerol stock of construct.
- Incubated the tube at 33C.
August 2nd
Preparing chemical competent cell
Preparing chemical competent cell of BL21, JM109 and DH5α. Chemical competent cell made in each E. coli strains.
Our competent cell Protocol
- Single colony isolation on LB plate.
- Incubated the plate for 15-19 hours at 37C.
- Lift colony of E. coli into 2 ml LB
- Incubate at 37C for 12-16 hours at 180-200 rpm
- Transfer 30 ul, 100 ul, 300 ul of the culture into 100 ml of LB.
- Incubate cells at 20C (for 24 hours over) at 140 rpm
- Select culture by measuring OD 600.(OD 600=0.5)
- Incubate the 300 ml flask for 10 min on ice
- Transfer the culture into two 50 ml Falcon tube
- Centrifuge at 3000 rpm, 4C for 20 min (TOMY LX-120 rotor), and discard sup.
- Suspend the pellet in ice-cold 15 ml of TB (Transformation Buffer).(7.5 ml/tube)
- Collect them to one tube.
- Centrifuge at 3000 rpm, 4C for 20 min (TOMY LX-120 rotor), and discard sup.
- Suspend the pellet in ice-cold 3.2 ml of TB.
- Instill 0.24 ml of DMSO in precipitant.
- Incubate the 50 ml Falcon tube for 10 min on ice.
- Divide 50 ul of solutions in each 0.5 ml tubes.
- Freezed the suspension by liquid nitrogen.
- Stored at –80C.
Electrophoresis
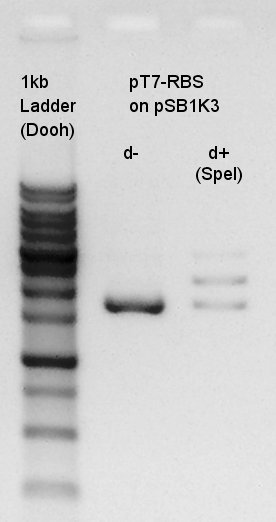
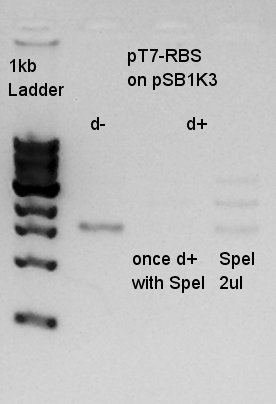
Electrophoresis of digestion result of August 1st.(pT7-RBS on pSB1K3 digested by SpeI)
There were low concentration band in above of thick band. This thick band was same as digestion minus band.
Digestion
Digestion of pT7-RBS on pSB1K3 (more fresh one)
| DNA solution | 4 ul |
| SpeI | 1 ul |
| 10xM buffer | 2 ul |
| DW | 13 ul |
| Total | 20 ul |
August 3rd
Electrophoresis
Electrophoresis for the result of digestion.
Gel extraction
Gel extraction for digestion product. We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50 ul of DNA solution.
Ethanol precipitation
Ethanol precipitation for digestion and gel extraction product.
- Added 5ul of NaoAc, 1.5 ul of glycogen and 125ul of 100% ethanol.
- Centrifuged at 15000 rpm, 10 min at 4C.
- Removed supernatant and added 220ul of 70% ethanol.
- Centrifuged at 15000 rpm, 5 min at 4C.
- Removed supernatant and air drying at room temperature then added 10 ul of DW.
Ligation
We ligated pT7-RBS on pSB1K3 (digested Spe1) as vector and Ag43-dT (digested Spe1 and Xba1 and HindIII) as insert.
| pT7-RBS | 2 ul |
| Ag43-dT | 4 ul |
| Ligation Mighty Mix(TAKARA) | 6 ul |
| Total | 12 ul |
Ligation reaction time was written below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
Transformation
Transformation of plasmid DNA ligation product (pT7-RBS-Ag43-dT on pSB1K3) in DH5α.
- Added 1 ul of plasmid DNA to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30min.
- Added 600 ul of LB then incubated the cells for 2 hrs at 37C.
- Prepared and Labeled two petri dishes with LBK.
- Plate 300 ul of the transformation onto first dish and spread.
- Added 900 ul of LB to 100 ul of the transformation and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for 15 hrs 45 min.
PCR
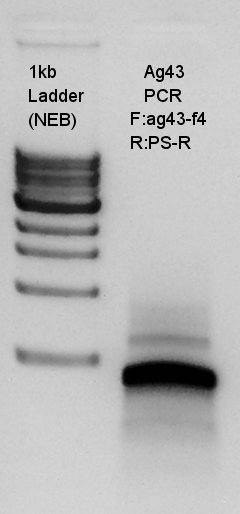
PCR to confirm Ag43-f4 primer.
| DNA solution | 1 ul |
| KOD-Plus-NEO(Taq polymerase) | 1 ul |
| dNTP | 5 ul |
| MgSO4 | 3 ul |
| KOD-Plus-NEO Buffer | 5 ul |
| Ag43-f4 primer | 1 ul |
| PS-R primer | 1 ul |
| DW | 33 ul |
| Total | 50 ul |
| Number | Degree | Second |
| 1 | 94 | 120 |
| 2 | 98 | 10 |
| 3 | 58 | 30 |
| 4 | 68 | 60 |
| 5 | 4 | HOLD |
Cycle:2~4 x 45
From this result, we could see that the ag43-f4 primer had worked and DNA of last 500~600 bp of ag43 to BioBrick suffix were increased.
August 4th
Colony PCR
Colony PCR to confirm that whether the Ag43-dT was successfully ligated with pT7-RBS on pSB1K3 or not.
| DNA solution | 4 ul |
| Kapa-Taq(Taq polymerase) | 5 ul |
| Forward Primer(Ag43-f4 primer) | 0.5 ul |
| Reverse Primer(PS-R primer) | 0.5 ul |
| Total | 10 ul |
| Number | Degree | Second |
| 1 | 95 | 120 |
| 2 | 95 | 30 |
| 3 | 58 | 30 |
| 4 | 72 | 60 |
| 5 | 72 | 60 |
| 6 | 4 | HOLD |
Cycle:2~4 x 35
We used N1 (DW only) and N2 (Ag43-dT on pSB1AK3) as controls. Desired product is about 500~600bp.
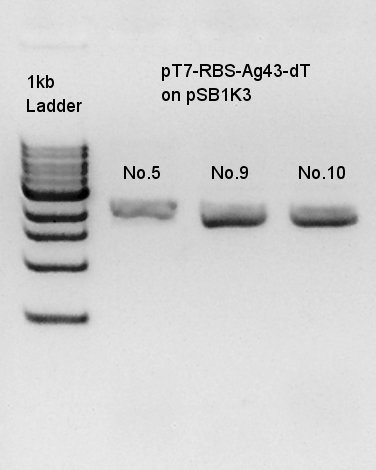
We thought that No. 5 and 9 colonies were made in exactly transformed in E. coli. and No. 10 colony had high concentration band. Next step, we resuspended these three colonies and cultured.
Liquid culture
Liquid culture of colonies passed the colony PCR test.
- Added 2 ml of LBK into culture tubes.
- Resuspended 3 colonies.
- Incubated the tubes at 37C for 13 hrs.
August 5th
Plasmid extraction
Mini-prep of incubated medium from yesterday. We used FastGene Plasmid Mini Kit(Nippon Genetics) and got 50 ul of DNA solutions.
Digestion
Digested pT7-RBS on pSB1K3 and pT7-RBS (once digestioned with SpeI) with speI. pT7-RBS on pSB1K3
| DNA solution | 8 ul |
| SpeI | 2 ul |
| 10xM buffer | 2 ul |
| DW | 8 ul |
| Total | 20 ul |
pT7-RBS (once digestioned with SpeI)
| DNA solution | 4 ul |
| SpeI | 1 ul |
| 10xM buffer | 1 ul |
| DW | 4 ul |
| Total | 10 ul |
| DNA solution | 4 ul |
| SpeI | 1 ul |
| 10xM buffer | 2 ul |
| DW | 13 ul |
| Total | 20 ul |
From this result, the bottom band was misdigested band and middle band was digested band we thought. Was the above band something contaminated in the DNA solution?
Gel extraction
Gel extraction of middle band. We thought this band would be the exactly digested DNA fragment. We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50 ul of DNA solution.
Sequencing
Sequencing to confirm what kind of DNA we made.
| DNA | primer | ||||
| Ag43 mini-prep product | 100bp-up forward primer, | ag43-f1, | ag43-f2, | ag43-f3, | ag43-f4 |
| K542009 | 100bp-up forward primer, | ag43-f1, | ag43-f2, | ag43-f3, | ag43-f4 |
| pT7-RBS on pSB1K3 | 100bp-up forward primer | ||||
| pT7-RBS on pSB1C3 | 100bp-up forward primer | ||||
| Ag43-dT on pSB1AK3 | 100bp-up forward primer, | ag43-f1, | ag43-f2, | ag43-f3, | ag43-f4 |
| Ag43-dT on pSB1T3 | 100bp-up forward primer, | ag43-f1, | ag43-f2, | ag43-f3, | ag43-f4 |
| pT7-RBS on pSB1K3 | 100bp-up forward primer |
Sequencing PCR
| template DNA | 1 ul |
| Ready Reaction Premix | 1 ul |
| 5x Sequencing Buffer | 1.5 ul |
| H2O | 5 ul |
| Primer(1 pmol/ul) | 1.5 ul |
| Total | 10 ul |
| Number | Degree | Second |
| 1 | 96 | 10 |
| 2 | 50 | 5 |
| 3 | 60 | 240 |
| 4 | 4 | HOLD |
Cycle:1~3 x 25
To purify the PCR product, we did ethanol precipitation.
Ethanol precipitation for sequencing.
- Added 10 ul of H2O, 2 ul of NaoAc, 1 ul of glycogen and 50 ul of 100% ethanol.
- Centrifuged at 15000 rpm, 15 min at 26C.
- Removed supernatant and added 100ul of 70% ethanol.
- Centrifuged at 15000 rpm, 5 min at 26C.
- Removed supernatant and air drying at room temperature then added 10 ul of Hi-Di.
Then run a sequencing machine.(ByoDye Terminator v3.1 Cycle Sequencing Kit)
We could not get the sequencing data.
Digestion
Digesting of Ag43-dT (digested by SpeI and XbaI) by HindIII.
| DNA solution | 10 ul |
| HindIII | 1 ul |
| 10xM buffer | 2 ul |
| DW | 7 ul |
| Total | 20 ul |
From this result, we confirmed that the pSB1AK3 was successfully digested by HindIII.
Gel extraction
Gel extraction for digestion production. We used FastGene Gel&PCR extraction kit(NipponGenetics)and got 50 ul of DNA solution.
 "
"