Team:HokkaidoU Japan/Notebook/aggregation Week 11
From 2012.igem.org
(Difference between revisions)
(10th digestionImage) |
(10th ethapre-transformation 11th colop) |
||
| Line 133: | Line 133: | ||
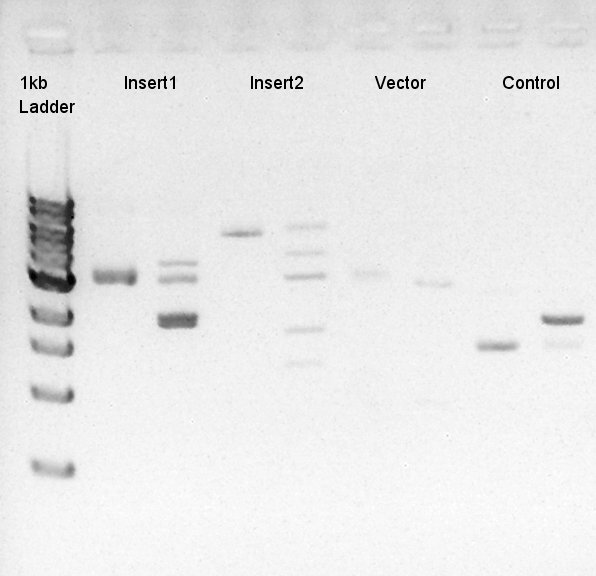
[[image:HokkaidoU2012 120910 digestion Insert1-pBAD-RBS-eCFP-RBS-pSB1A2 Insert2-Ag43-dT-pSB1AK3 Vector-ptet-RBS-eYFP-dT-pSTV28 Control-pT7-RBS-pSB1A2.jpg|thumb|digestion result]] | [[image:HokkaidoU2012 120910 digestion Insert1-pBAD-RBS-eCFP-RBS-pSB1A2 Insert2-Ag43-dT-pSB1AK3 Vector-ptet-RBS-eYFP-dT-pSTV28 Control-pT7-RBS-pSB1A2.jpg|thumb|digestion result]] | ||
| + | </p> | ||
| + | |||
| + | ==Ethanol precipitation of ptet-RBS-eYFP-dT-pSB1A2 and pSTV28== | ||
| + | <p> | ||
| + | #Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol. | ||
| + | #Centrifuged in 14000 rpm, 30 min at 4C. | ||
| + | #Remove supernatant and added 220 ul of 70% ethanol. | ||
| + | #Centrifuged in 15000 rpm, 15 min at 4C. | ||
| + | #Remove supernatant and air drying in room temperature then added 5 ul of DW. | ||
| + | |||
| + | |||
| + | [[image:|thumb|ethanol precipitation result]] | ||
| + | We confirmed that the concentration of Insert1 DNA solution is 50 ng/ul, Insert2 DNA solution is 10~15 ng/ul and Vector DNA solution is 10 ng/ul. | ||
| + | </p> | ||
| + | |||
| + | ==Ligation of pBAD-RBS-eCFP-RBS, Ag43-dT and pSTV28== | ||
| + | <p> | ||
| + | {|class="hokkaidou-table-ligation" | ||
| + | |- | ||
| + | |Vector DNA (10 ng/ul) | ||
| + | |4 ul | ||
| + | |- | ||
| + | |Insert1 DNA (50 ng/ul) | ||
| + | |2 ul | ||
| + | |- | ||
| + | |Insert2 DNA (10~15 ng/ul) | ||
| + | |4 ul | ||
| + | |- | ||
| + | |Ligation Mighty Mix | ||
| + | |10 ul | ||
| + | |- | ||
| + | |Total | ||
| + | |20 ul | ||
| + | |} | ||
| + | |||
| + | |||
| + | Ligation reaction time was in detail below. | ||
| + | |||
| + | {|class="hokkaidou-table-ligation" | ||
| + | |- | ||
| + | |Degree | ||
| + | |Minute | ||
| + | |- | ||
| + | |16 | ||
| + | |30 | ||
| + | |- | ||
| + | |65 | ||
| + | |10 | ||
| + | |- | ||
| + | |4 | ||
| + | |Hold | ||
| + | |} | ||
| + | |||
| + | </p> | ||
| + | |||
| + | ==Transformation of pBAD-RBS-eCFP-RBS-Ag43-dT-pSTV28== | ||
| + | <p> | ||
| + | #Mixed 2 ul pBAD-RBS-eCFP-RBS-Ag43-dT-pSTV28 ligation product to 50 ul of thawed competent cells on ice. | ||
| + | #Incubated on ice for 30 min. | ||
| + | #Mixed 350 ul of LB. | ||
| + | #Incubated for 2 hrs to get the resistance to Chloramphenicol. | ||
| + | #Prepared and Labeled two plastic plates with LB plate medium which contained appropriate antibiotics (LBC). | ||
| + | #Plated 300 ul of the culture onto first dish and spread. | ||
| + | #Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread. | ||
| + | #Incubated the plates at 37C for 14 hours. | ||
| + | </p> | ||
| + | </div></div> | ||
| + | |||
| + | |||
| + | <div class="hokkaidou-notebook-daily"> | ||
| + | ==September 10th== | ||
| + | <div> | ||
| + | ==Colony PCR of pBAD-RBS-eCFP-RBS-pSB1A2== | ||
| + | <p> | ||
| + | |||
| + | |||
| + | {|class="hokkaidou-table-pcr-reagent" | ||
| + | |- | ||
| + | |DNA solution | ||
| + | |4 ul | ||
| + | |- | ||
| + | |Kapa-Taq(Taq polymerase) | ||
| + | |5 ul | ||
| + | |- | ||
| + | |Forward Primer(pbad-f2 primer) | ||
| + | |0.5 ul | ||
| + | |- | ||
| + | |Reverse Primer(PS-R down primer) | ||
| + | |0.5 ul | ||
| + | |- | ||
| + | |Total | ||
| + | |10 ul | ||
| + | |} | ||
| + | |||
| + | |||
| + | {|class="hokkaidou-table-pcr-time" | ||
| + | |- | ||
| + | |Number | ||
| + | |Degree | ||
| + | |Second | ||
| + | |- | ||
| + | |1 | ||
| + | |95 | ||
| + | |120 | ||
| + | |- | ||
| + | |2 | ||
| + | |95 | ||
| + | |30 | ||
| + | |- | ||
| + | |3 | ||
| + | |53.3 | ||
| + | |30 | ||
| + | |- | ||
| + | |4 | ||
| + | |72 | ||
| + | |60 | ||
| + | |- | ||
| + | |5 | ||
| + | |72 | ||
| + | |60 | ||
| + | |- | ||
| + | |6 | ||
| + | |4 | ||
| + | |HOLD | ||
| + | |} | ||
| + | Cycle:2~4 x 35 | ||
| + | |||
| + | We used N1 (DW only) and N2(pBAD-RBS-Ag43-dT-pSB1A2)as controls. | ||
| + | Desired product is about 542bp. | ||
| + | |||
| + | [[image:|thumb|Colony PCR result]] | ||
| + | |||
</p> | </p> | ||
</div></div> | </div></div> | ||
Revision as of 06:07, 11 September 2012
Contents |
September 10th
Digestion of eCFP-RBS-pSB1A2 and pBAD-RBS-pSB1A2
To make a construct of pBAD-RBS-eCFP-RBS-Ag43-dT-pSTV28 by 3piece ligation, we digested pBAD-RBS-eCFP-RBS-pSB1A2 with EcoRI & SpeI, Ag43-dT-pSB1AK3 (previously digested with HindIII) with XbaI & NotI and pSTV28 with EcoRI and NotI. Then we digested pT7-RBS-pSB1C3 with XbaI & SpeI as a control for confirmation of the ability of restriction enzyme. Insert1 (pBAD-RBS-eCFP-RBS-pSB1A2)
| DNA solution ( 35ng/ul) | 17 ul |
| EcoRI | 1 ul |
| SpeI | 1 ul |
| 10xH buffer | 3 ul |
| DW | 8 ul |
| Total | 30 ul |
Insert2 (Ag43-dT-pSB1AK3)
| DNA solution ( 35ng/ul) | 25 ul |
| XbaI | 1 ul |
| NotI | 1 ul |
| 10xK buffer | 1.5 ul |
| 100xBSA | 0.3ul |
| DW | 1.5 ul |
| Total | 30 ul |
Vector(pSTV28)
| DNA solution ( 15ng/ul) | 9 ul |
| EcoRI | 1 ul |
| NotI | 1 ul |
| 10xH buffer | 2 ul |
| 10xBSA | 0.2ul |
| DW | 7 ul |
| Total | 20 ul |
control (pT7-RBS-pSB1C3)
| DNA solution (30~40 ng/ul) | 10 ul |
| XbaI | 1 ul |
| SpeI | 1 ul |
| 10xM buffer | 2 ul |
| DW | 6 ul |
| Total | 20 ul |
| Number | Degree | Minute |
| 1 | 37 | 120 |
| 2 | 70 | 20 |
| 3 | 4 | HOLD |
Ethanol precipitation of ptet-RBS-eYFP-dT-pSB1A2 and pSTV28
- Added 5 ul of NaoAc, 1.5 ul of glycogen and 125 ul of 100% ethanol.
- Centrifuged in 14000 rpm, 30 min at 4C.
- Remove supernatant and added 220 ul of 70% ethanol.
- Centrifuged in 15000 rpm, 15 min at 4C.
- Remove supernatant and air drying in room temperature then added 5 ul of DW.
Ligation of pBAD-RBS-eCFP-RBS, Ag43-dT and pSTV28
| Vector DNA (10 ng/ul) | 4 ul |
| Insert1 DNA (50 ng/ul) | 2 ul |
| Insert2 DNA (10~15 ng/ul) | 4 ul |
| Ligation Mighty Mix | 10 ul |
| Total | 20 ul |
Ligation reaction time was in detail below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
Transformation of pBAD-RBS-eCFP-RBS-Ag43-dT-pSTV28
- Mixed 2 ul pBAD-RBS-eCFP-RBS-Ag43-dT-pSTV28 ligation product to 50 ul of thawed competent cells on ice.
- Incubated on ice for 30 min.
- Mixed 350 ul of LB.
- Incubated for 2 hrs to get the resistance to Chloramphenicol.
- Prepared and Labeled two plastic plates with LB plate medium which contained appropriate antibiotics (LBC).
- Plated 300 ul of the culture onto first dish and spread.
- Mixed 450 ul of LB to 50 ul of the culture and plated 300 ul of it onto second dish and spread.
- Incubated the plates at 37C for 14 hours.
September 10th
Colony PCR of pBAD-RBS-eCFP-RBS-pSB1A2
| DNA solution | 4 ul |
| Kapa-Taq(Taq polymerase) | 5 ul |
| Forward Primer(pbad-f2 primer) | 0.5 ul |
| Reverse Primer(PS-R down primer) | 0.5 ul |
| Total | 10 ul |
| Number | Degree | Second |
| 1 | 95 | 120 |
| 2 | 95 | 30 |
| 3 | 53.3 | 30 |
| 4 | 72 | 60 |
| 5 | 72 | 60 |
| 6 | 4 | HOLD |
Cycle:2~4 x 35
We used N1 (DW only) and N2(pBAD-RBS-Ag43-dT-pSB1A2)as controls. Desired product is about 542bp.
[[image:|thumb|Colony PCR result]]
 "
"