Team:ETH Zurich/UVR8/Results
From 2012.igem.org
| (22 intermediate revisions not shown) | |||
| Line 2: | Line 2: | ||
{{:Team:ETH_Zurich/Templates/SubmenuUVR8}} | {{:Team:ETH_Zurich/Templates/SubmenuUVR8}} | ||
| + | |||
| + | == TetR<sub>DBD</sub>-UVR8 expression studies == | ||
| + | |||
| + | [[File:UVR8 SDS native page.jpg|frameless|350px|right|thumb|Figure 1: UVR8 expression: a) SDS-page of TetR<sub>DBD</sub>-dUVR8 expression. An additional 65 kDa protein appears after introducing a TetR<sub>DBD</sub>-dUVR8 gene into the cells. b) Native page of a purified TetR<sub>DBD</sub>-dUVR8-Histag fusion. In native state, TetR<sub>DBD</sub>-dUVR8 forms a dimer which is split by heat denaturation]] | ||
| + | |||
| + | We tested whether TetR<sub>DBD</sub>-UVR8 fusions can be expressed in as a proper folded proteins in ''E.coli''. SDS-page analysis clearly shows, that introducing TetR<sub>DBD</sub>-dUVR8 results in expression of 65kDa size protein - the same theoretical size as TetR<sub>DBD</sub>-dUVR8 (fig. 1a). Furthermore, we added 7xHistag to C-terminus of the TetR<sub>DBD</sub>-dUVR8 construct and purified it through Ni affinity chromatography column. However, contrarily to literature <span class='eth_reference'>[Heijde2012]</span>, TetR<sub>DBD</sub>-dUVR8 dimer was not seen in SDS-page gels(data not shown). Nevertheless, dimer is observed in the native page analysis, indicating that ''E.coli'' synthesizes a functional TetR<sub>DBD</sub>-dUVR8. | ||
== Repression studies == | == Repression studies == | ||
| - | |||
| - | |||
In order to test whether a UVR8 fusion with the TetR DNA binding domain (TetR<sub>DBD</sub>) is reasonable, we first had to test, if our used TetR<sub>DBD</sub> is not able to repress the expression of GFP that is controlled by a P<sub>tet</sub> promoter. For this purpose we measured the fluorescence from three different cultures: reporter only, reporter co-transfected with full length TetR or TetR<sub>DBD</sub>. Cells without any plasmid termed ''WT'' were used as a background fluorescence control. | In order to test whether a UVR8 fusion with the TetR DNA binding domain (TetR<sub>DBD</sub>) is reasonable, we first had to test, if our used TetR<sub>DBD</sub> is not able to repress the expression of GFP that is controlled by a P<sub>tet</sub> promoter. For this purpose we measured the fluorescence from three different cultures: reporter only, reporter co-transfected with full length TetR or TetR<sub>DBD</sub>. Cells without any plasmid termed ''WT'' were used as a background fluorescence control. | ||
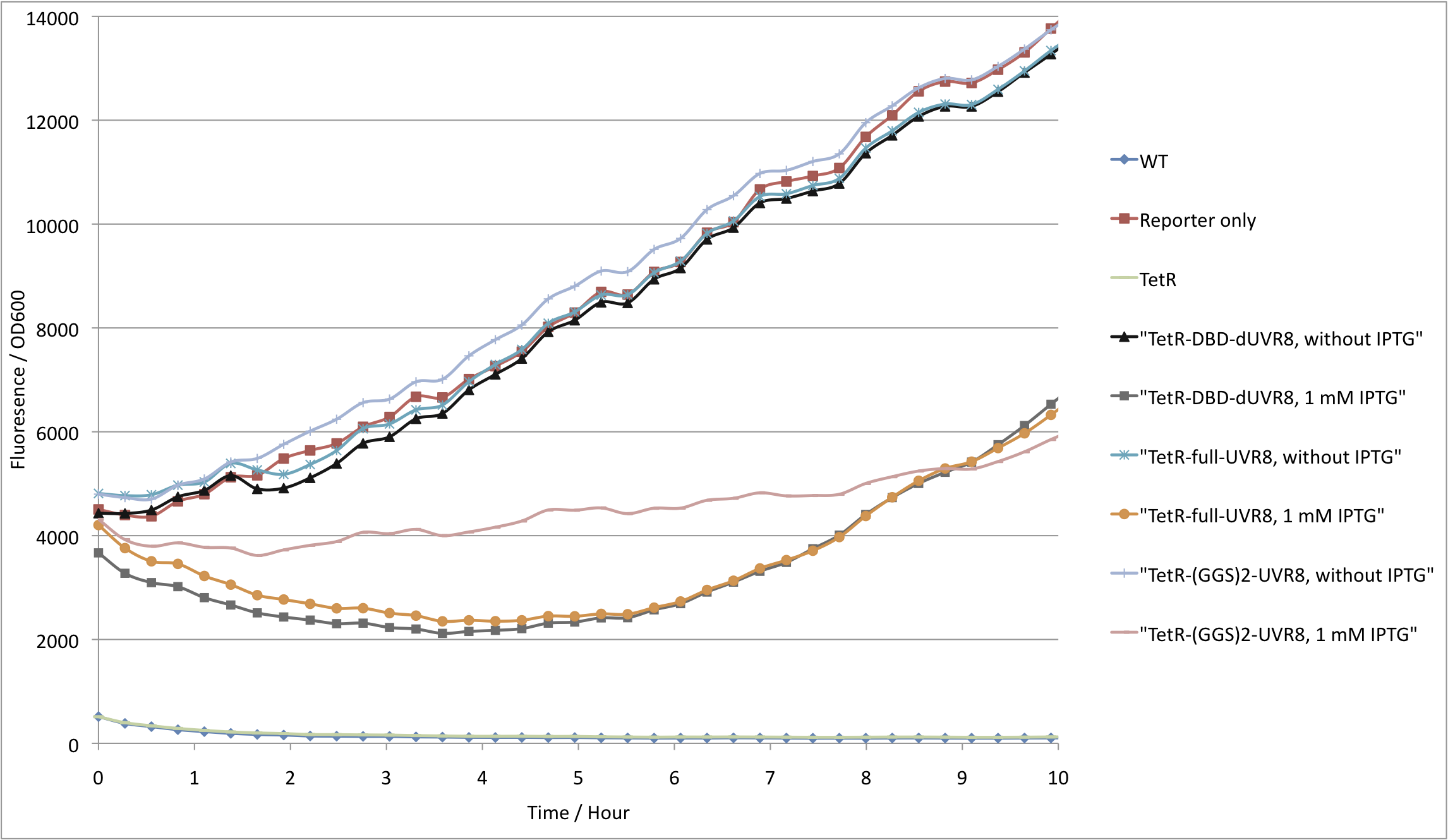
| - | As expected, cells with ''reporter only'' showed high GFP fluorescence, whereas cells containing full length tetR have a tightly repressed GFP production. In addition, cells expressing TetR<sub>DBD</sub> also express GFP at similar levels to those with just the reporter plasmid (see figure | + | As expected, cells with ''reporter only'' showed high GFP fluorescence, whereas cells containing full length tetR have a tightly repressed GFP production. In addition, cells expressing TetR<sub>DBD</sub> also express GFP at similar levels to those with just the reporter plasmid (see figure 2). These results can be explained in two ways: dimerizing domain is required for proper TetR<sub>DBD</sub> folding. Another explanation would be that TetR can bind P<sub>tet</sub> more efficiently due to cooperativity. If the latter is true, fusion of the TetR<sub>DBD</sub> with proteins, that are able to dimerize, would restore its activity to repress the P<sub>tet</sub> promoter. |
| + | [[File:TetRDBD_1.png|frameless|600px|center|thumb|Figure 2: Fluorescence normalized to the OD for wt, reporter only, TetR and TetR<sub>DBD</sub>. TetR shows the fluorescence level as the wildtype.]] | ||
<br style="clear: both" /> | <br style="clear: both" /> | ||
| - | Thus we fused TetR<sub>DBD</sub> with three versions of UVR8 proteins each varying in linker length ([[Team:ETH_Zurich/UVR8/Design|see Design]]). Again, fluorescence from cells cotransfected with fusion proteins and reporter ([[Team:ETH_Zurich/UVR8/Design#Plasmids|see Plasmids]]) were compared with reporter only and wildtype. | + | Thus we fused TetR<sub>DBD</sub> with three versions of UVR8 proteins each varying in linker length ([[Team:ETH_Zurich/UVR8/Design|see Design]]). Again, fluorescence from cells cotransfected with fusion proteins and reporter ([[Team:ETH_Zurich/UVR8/Design#Plasmids|see Plasmids]]) were compared with reporter only and wildtype. Figure 3 shows that we are able to repress GFP production by fusing TetR<sub>DBD</sub> with UVR8. We limit our further studies to the best performing chimera – TetR<sub>DBD</sub>-truncated-UVR8 ([[Team:ETH_Zurich/UVR8/Design|see 3.]]). |
| - | [[File:UVR8_3.png|frameless|600px|center|thumb|Figure | + | [[File:UVR8_3.png|frameless|600px|center|thumb|Figure 3: Comparison of different TetR<sub>DBD</sub>-UVR8 fusions with TetR and reporter only ]] |
| - | [[File:IPTG_1.png|frameless|600px|center|thumb|Figure | + | [[File:IPTG_1.png|frameless|600px|center|thumb|Figure 4: Cell growth dependent on UVR8 expression ]] |
| - | However, high inductions of UVR8 are toxic for cells and impair cell growth (fig. | + | However, high inductions of UVR8 are toxic for cells and impair cell growth (fig. 4). Nevertheless, for our purposes we do not need high overexpression of the protein thus we tested repression dependency on protein induction. |
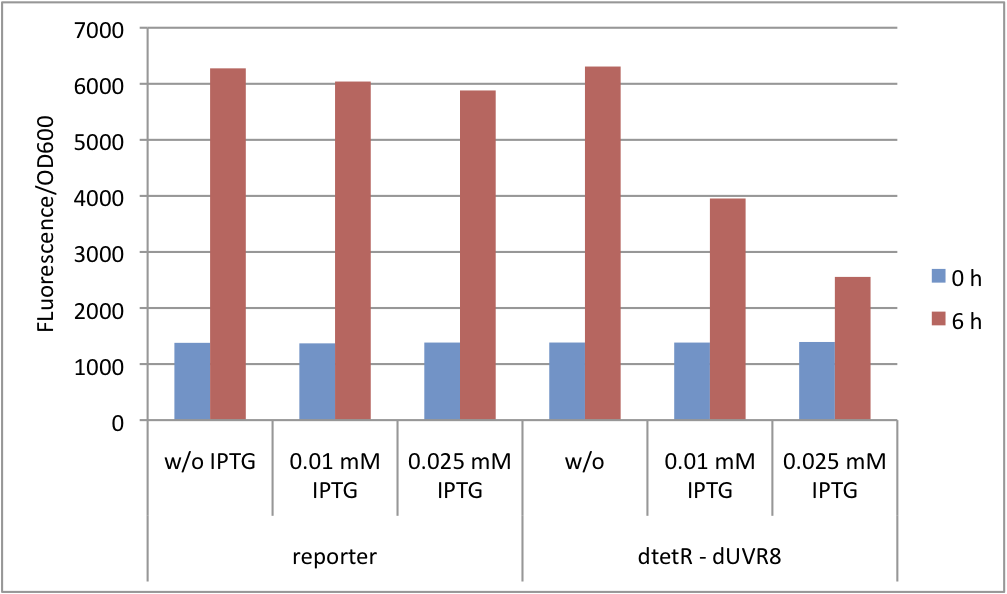
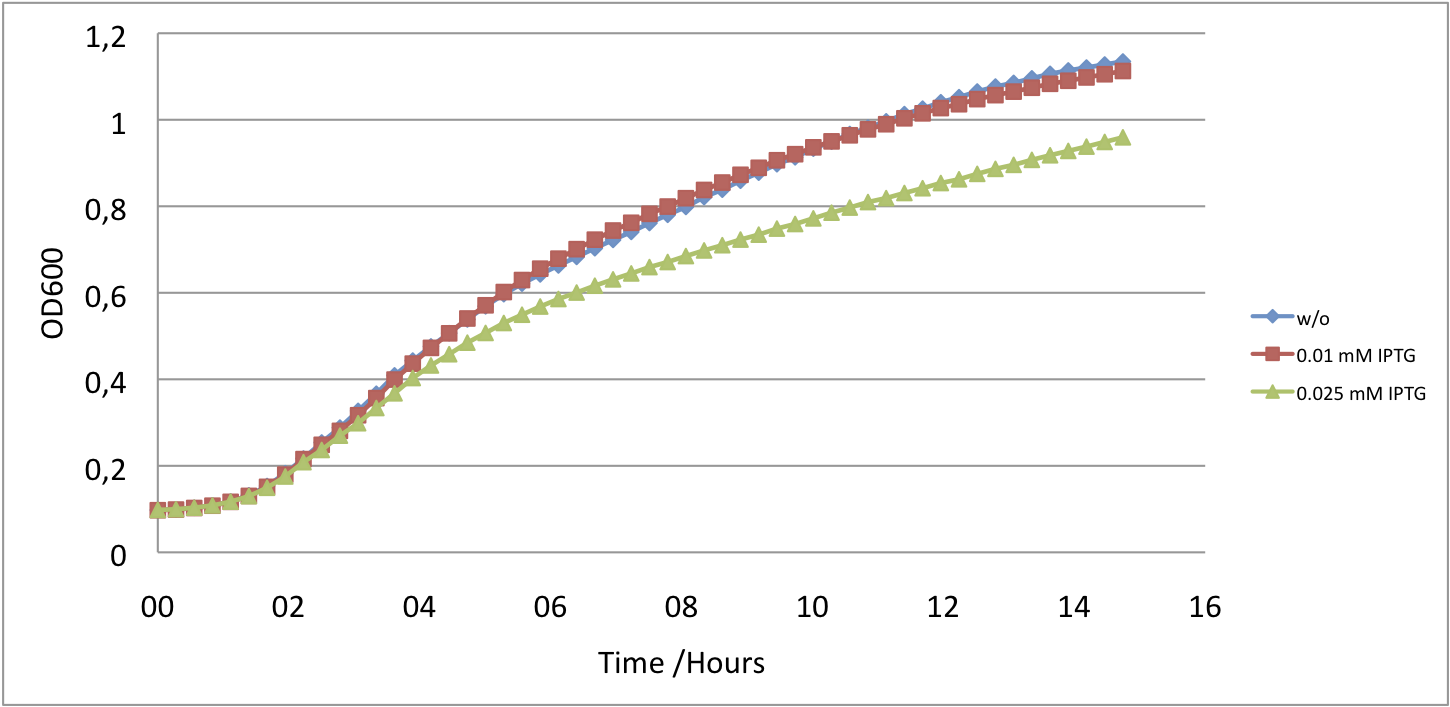
| - | Since the expression of UVR8 is induced by IPTG, we varied IPTG concentrations. The results show that even small amounts, 0.01-0.025 mM, of inducer results in GFP repression which does not impair cell growth (fig. | + | Since the expression of UVR8 is induced by IPTG, we varied IPTG concentrations. The results show that even small amounts, 0.01-0.025 mM, of inducer results in GFP repression which does not impair cell growth (fig. 5 and fig. 6). |
| - | [[File: | + | [[File:UVR8_4.png|frameless|600px|center|thumb|Figure 5: Repression dependent on TetR-DBD-UVR8 induction via different IPTG concentrations. left: reporter only as negative control]] |
| - | [[File: | + | [[File:UVR8_5.png|frameless|600px|center|thumb|Figure 6: Cells expressing TetR<sub>DBD</sub>-UVR8 grow at low inducer IPTG concentrations, thus low TetR-DBD-UVR8 concentration]] |
== New part TetR<sub>DBD</sub> == | == New part TetR<sub>DBD</sub> == | ||
| - | We showed that the TetR monomer is unable to repress the P<sub>tet</sub> promoter. This phenomenon makes our new part [http://partsregistry.org/Part:BBa_K909007 BBa_K909007] a powerful tool. Any protein can be fused in case the coding DNA contains a BamHI restriction site. By that fusion, any protein able to dimerize acts as a transcriptional repressor. By combining the P<sub>tet</sub> promoter and any reporter gene, one can test whether certain proteins are able to dimerize in a cell or not. | + | We showed that the TetR monomer is unable to repress the P<sub>tet</sub> promoter. This phenomenon makes our new part [http://partsregistry.org/Part:BBa_K909007 BBa_K909007] a powerful tool. Any protein can be fused in case the coding DNA contains a BamHI restriction site. By that fusion, any protein able to dimerize acts as a transcriptional repressor. By combining the P<sub>tet</sub> promoter and any reporter gene, one can test whether certain proteins are able to [[Team:ETH_Zurich/UVR8#TetRDBD_-_a_novel_two-hybrid_screening_in_E.coli|dimerize]] in a cell or not. |
| - | + | ||
==Achievements so far== | ==Achievements so far== | ||
| Line 45: | Line 49: | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
{{:Team:ETH_Zurich/Templates/Footer}} | {{:Team:ETH_Zurich/Templates/Footer}} | ||
Latest revision as of 02:46, 27 October 2012
Contents |
TetRDBD-UVR8 expression studies
We tested whether TetRDBD-UVR8 fusions can be expressed in as a proper folded proteins in E.coli. SDS-page analysis clearly shows, that introducing TetRDBD-dUVR8 results in expression of 65kDa size protein - the same theoretical size as TetRDBD-dUVR8 (fig. 1a). Furthermore, we added 7xHistag to C-terminus of the TetRDBD-dUVR8 construct and purified it through Ni affinity chromatography column. However, contrarily to literature [Heijde2012], TetRDBD-dUVR8 dimer was not seen in SDS-page gels(data not shown). Nevertheless, dimer is observed in the native page analysis, indicating that E.coli synthesizes a functional TetRDBD-dUVR8.
Repression studies
In order to test whether a UVR8 fusion with the TetR DNA binding domain (TetRDBD) is reasonable, we first had to test, if our used TetRDBD is not able to repress the expression of GFP that is controlled by a Ptet promoter. For this purpose we measured the fluorescence from three different cultures: reporter only, reporter co-transfected with full length TetR or TetRDBD. Cells without any plasmid termed WT were used as a background fluorescence control.
As expected, cells with reporter only showed high GFP fluorescence, whereas cells containing full length tetR have a tightly repressed GFP production. In addition, cells expressing TetRDBD also express GFP at similar levels to those with just the reporter plasmid (see figure 2). These results can be explained in two ways: dimerizing domain is required for proper TetRDBD folding. Another explanation would be that TetR can bind Ptet more efficiently due to cooperativity. If the latter is true, fusion of the TetRDBD with proteins, that are able to dimerize, would restore its activity to repress the Ptet promoter.
Thus we fused TetRDBD with three versions of UVR8 proteins each varying in linker length (see Design). Again, fluorescence from cells cotransfected with fusion proteins and reporter (see Plasmids) were compared with reporter only and wildtype. Figure 3 shows that we are able to repress GFP production by fusing TetRDBD with UVR8. We limit our further studies to the best performing chimera – TetRDBD-truncated-UVR8 (see 3.).
However, high inductions of UVR8 are toxic for cells and impair cell growth (fig. 4). Nevertheless, for our purposes we do not need high overexpression of the protein thus we tested repression dependency on protein induction.
Since the expression of UVR8 is induced by IPTG, we varied IPTG concentrations. The results show that even small amounts, 0.01-0.025 mM, of inducer results in GFP repression which does not impair cell growth (fig. 5 and fig. 6).
New part TetRDBD
We showed that the TetR monomer is unable to repress the Ptet promoter. This phenomenon makes our new part [http://partsregistry.org/Part:BBa_K909007 BBa_K909007] a powerful tool. Any protein can be fused in case the coding DNA contains a BamHI restriction site. By that fusion, any protein able to dimerize acts as a transcriptional repressor. By combining the Ptet promoter and any reporter gene, one can test whether certain proteins are able to dimerize in a cell or not.
Achievements so far
- TetRDBD only binds to DNA as a dimer
- enables to use TetRDBD in a two-hybrid screening or as a switch
- TetRDBD-UVR8 fusion is able to act as a repressor
References
- Brown, B. a, Headland, L. R., & Jenkins, G. I. (2009). UV-B action spectrum for UVR8-mediated HY5 transcript accumulation in Arabidopsis. Photochemistry and photobiology, 85(5), 1147–55.
- Christie, J. M., Salomon, M., Nozue, K., Wada, M., & Briggs, W. R. (1999): LOV (light, oxygen, or voltage) domains of the blue-light photoreceptor phototropin (nph1): binding sites for the chromophore flavin mononucleotide. Proceedings of the National Academy of Sciences of the United States of America, 96(15), 8779–83.
- Christie, J. M., Arvai, A. S., Baxter, K. J., Heilmann, M., Pratt, A. J., O’Hara, A., Kelly, S. M., et al. (2012). Plant UVR8 photoreceptor senses UV-B by tryptophan-mediated disruption of cross-dimer salt bridges. Science (New York, N.Y.), 335(6075), 1492–6.
- Cloix, C., & Jenkins, G. I. (2008). Interaction of the Arabidopsis UV-B-specific signaling component UVR8 with chromatin. Molecular plant, 1(1), 118–28.
- Cox, R. S., Surette, M. G., & Elowitz, M. B. (2007). Programming gene expression with combinatorial promoters. Molecular systems biology, 3(145), 145. doi:10.1038/msb4100187
- Drepper, T., Eggert, T., Circolone, F., Heck, A., Krauss, U., Guterl, J.-K., Wendorff, M., et al. (2007). Reporter proteins for in vivo fluorescence without oxygen. Nature biotechnology, 25(4), 443–5
- Drepper, T., Krauss, U., & Berstenhorst, S. M. zu. (2011). Lights on and action! Controlling microbial gene expression by light. Applied microbiology, 23–40.
- EuropeanCommission (2006). SCIENTIFIC COMMITTEE ON CONSUMER PRODUCTS SCCP Opinion on Biological effects of ultraviolet radiation relevant to health with particular reference to sunbeds for cosmetic purposes.
- Elvidge, C. D., Keith, D. M., Tuttle, B. T., & Baugh, K. E. (2010). Spectral identification of lighting type and character. Sensors (Basel, Switzerland), 10(4), 3961–88.
- GarciaOjalvo, J., Elowitz, M. B., & Strogatz, S. H. (2004). Modeling a synthetic multicellular clock: repressilators coupled by quorum sensing. Proceedings of the National Academy of Sciences of the United States of America, 101(30), 10955–60.
- Gao Q, Garcia-Pichel F. (2011). Microbial ultraviolet sunscreens. Nat Rev Microbiol. 9(11):791-802.
- Goosen N, Moolenaar GF. (2008) Repair of UV damage in bacteria. DNA Repair (Amst).7(3):353-79.
- Heijde, M., & Ulm, R. (2012). UV-B photoreceptor-mediated signalling in plants. Trends in plant science, 17(4), 230–7.
- Hirose, Y., Narikawa, R., Katayama, M., & Ikeuchi, M. (2010). Cyanobacteriochrome CcaS regulates phycoerythrin accumulation in Nostoc punctiforme, a group II chromatic adapter. Proceedings of the National Academy of Sciences of the United States of America, 107(19), 8854–9.
- Hirose, Y., Shimada, T., Narikawa, R., Katayama, M., & Ikeuchi, M. (2008). Cyanobacteriochrome CcaS is the green light receptor that induces the expression of phycobilisome linker protein. Proceedings of the National Academy of Sciences of the United States of America, 105(28), 9528–33.
- Kast, Asif-Ullah & Hilvert (1996) Tetrahedron Lett. 37, 2691 - 2694., Kast, Asif-Ullah, Jiang & Hilvert (1996) Proc. Natl. Acad. Sci. USA 93, 5043 - 5048
- Kiefer, J., Ebel, N., Schlücker, E., & Leipertz, A. (2010). Characterization of Escherichia coli suspensions using UV/Vis/NIR absorption spectroscopy. Analytical Methods, 9660. doi:10.1039/b9ay00185a
- Kinkhabwala, A., & Guet, C. C. (2008). Uncovering cis regulatory codes using synthetic promoter shuffling. PloS one, 3(4), e2030.
- Krebs in Deutschland 2005/2006. Häufigkeiten und Trends. 7. Auflage, 2010, Robert Koch-Institut (Hrsg) und die Gesellschaft der epidemiologischen Krebsregister in Deutschland e. V. (Hrsg). Berlin.
- Lamparter, T., Michael, N., Mittmann, F., & Esteban, B. (2002). Phytochrome from Agrobacterium tumefaciens has unusual spectral properties and reveals an N-terminal chromophore attachment site. Proceedings of the National Academy of Sciences of the United States of America, 99(18), 11628–33.
- Levskaya, A. et al (2005). Engineering Escherichia coli to see light. Nature, 438(7067), 442.
- Mancinelli, A. (1986). Comparison of spectral properties of phytochromes from different preparations. Plant physiology, 82(4), 956–61.
- Nakasone, Y., Ono, T., Ishii, A., Masuda, S., & Terazima, M. (2007). Transient dimerization and conformational change of a BLUF protein: YcgF. Journal of the American Chemical Society, 129(22), 7028–35.
- Orth, P., & Schnappinger, D. (2000). Structural basis of gene regulation by the tetracycline inducible Tet repressor-operator system. Nature structural biology, 215–219.
- Parkin, D.M., et al., Global cancer statistics, 2002. CA: a cancer journal for clinicians, 2005. 55(2): p. 74-108.
- Rajagopal, S., Key, J. M., Purcell, E. B., Boerema, D. J., & Moffat, K. (2004). Purification and initial characterization of a putative blue light-regulated phosphodiesterase from Escherichia coli. Photochemistry and photobiology, 80(3), 542–7.
- Rizzini, L., Favory, J.-J., Cloix, C., Faggionato, D., O’Hara, A., Kaiserli, E., Baumeister, R., et al. (2011). Perception of UV-B by the Arabidopsis UVR8 protein. Science (New York, N.Y.), 332(6025), 103–6.
- Roux, B., & Walsh, C. T. (1992). p-aminobenzoate synthesis in Escherichia coli: kinetic and mechanistic characterization of the amidotransferase PabA. Biochemistry, 31(30), 6904–10.
- Strickland, D. (2008). Light-activated DNA binding in a designed allosteric protein. Proceedings of the National Academy of Sciences of the United States of America, 105(31), 10709–10714.
- Sinha RP, Häder DP. UV-induced DNA damage and repair: a review. Photochem Photobiol Sci. (2002). 1(4):225-36
- Sambandan DR, Ratner D. (2011). Sunscreens: an overview and update. J Am Acad Dermatol. 2011 Apr;64(4):748-58.
- Tabor, J. J., Levskaya, A., & Voigt, C. A. (2011). Multichromatic Control of Gene Expression in Escherichia coli. Journal of Molecular Biology, 405(2), 315–324.
- Thibodeaux, G., & Cowmeadow, R. (2009). A tetracycline repressor-based mammalian two-hybrid system to detect protein–protein interactions in vivo. Analytical biochemistry, 386(1), 129–131.
- Tschowri, N., & Busse, S. (2009). The BLUF-EAL protein YcgF acts as a direct anti-repressor in a blue-light response of Escherichia coli. Genes & development, 522–534.
- Tschowri, N., Lindenberg, S., & Hengge, R. (2012). Molecular function and potential evolution of the biofilm-modulating blue light-signalling pathway of Escherichia coli. Molecular microbiology.
- Tyagi, A. (2009). Photodynamics of a flavin based blue-light regulated phosphodiesterase protein and its photoreceptor BLUF domain.
- Vainio, H. & Bianchini, F. (2001). IARC Handbooks of Cancer Prevention: Volume 5: Sunscreens. Oxford University Press, USA
- Quinlivan, Eoin P & Roje, Sanja & Basset, Gilles & Shachar-Hill, Yair & Gregory, Jesse F & Hanson, Andrew D. (2003). The folate precursor p-aminobenzoate is reversibly converted to its glucose ester in the plant cytosol. The Journal of biological chemistry, 278.
- van Thor, J. J., Borucki, B., Crielaard, W., Otto, H., Lamparter, T., Hughes, J., Hellingwerf, K. J., et al. (2001). Light-induced proton release and proton uptake reactions in the cyanobacterial phytochrome Cph1. Biochemistry, 40(38), 11460–71.
- Wegkamp A, van Oorschot W, de Vos WM, Smid EJ. (2007 )Characterization of the role of para-aminobenzoic acid biosynthesis in folate production by Lactococcus lactis. Appl Environ Microbiol. Apr;73(8):2673-81.
 "
"