Team:University College London/LabBook/Week17
From 2012.igem.org
(→Material from cells containing our Biosafety BioBrick is not able to transform commercial competent cells) |
|||
| Line 2: | Line 2: | ||
</html>{{:Team:University_College_London/templates/labbookmenu}}<html> | </html>{{:Team:University_College_London/templates/labbookmenu}}<html> | ||
<img src="https://static.igem.org/mediawiki/2012/d/d4/Ucl2012-labbook-monfri.png" /> | <img src="https://static.igem.org/mediawiki/2012/d/d4/Ucl2012-labbook-monfri.png" /> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
<img src="http://www.plasticrepublic.org/wikifiles/lab17-2.png"" /><div class="experimentContent"> | <img src="http://www.plasticrepublic.org/wikifiles/lab17-2.png"" /><div class="experimentContent"> | ||
</html> | </html> | ||
| Line 22: | Line 17: | ||
We suggest periplasmic nuclease expression provides a promising strategy for preventing transfer of genetically modified DNA. This is particularly valuable for the application of synthetic biology in environmental contexts. | We suggest periplasmic nuclease expression provides a promising strategy for preventing transfer of genetically modified DNA. This is particularly valuable for the application of synthetic biology in environmental contexts. | ||
| - | |||
| - | |||
<html> | <html> | ||
</div><div class="experiment"></div> | </div><div class="experiment"></div> | ||
Revision as of 22:28, 26 October 2012



Material from cells containing our Biosafety BioBrick is not able to transform commercial competent cells
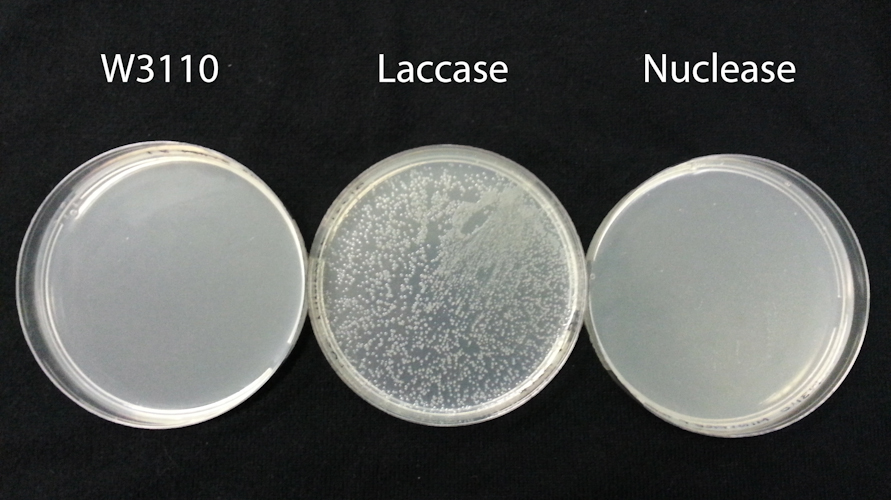
We used sonication to disrupt three cell types: unmodified W3110 strain E. coli and W3110 strains harbouring chloramphenical resistance plasmids encoding a laccase (BBa_K729006) or the DsbA-Nuclease (BBa_K729019). All three strains were grown to OD600= 2.0 prior to sonication, in 2mL LB broth which contained 100ug/mL chloramphenical for plasmid-harbouring cells.
After sonication, disruptates were incubated for 10min at room temperature. 5uL of the disruptate was used to transform an aliquot of TOP10 chemically competent cells, following manufacturer instructions. As a control, we also spread 20uL of the disruptate material directly onto LB agar plates with and without 100ug/mL Chloramphenical. No colonies were observed on the control plates for any of the three strains (data not shown), indicating sonication had successfully disrupted all cells.
As expected, transformation of TOP10 cells with disruptate of unmodified W3110 did not yield any cells able to grow on chloramphenicol plates (FigXA). In contrast transformation with disruptate of W3110 harbouring the BBa_K729006 laccase plasmid yielded many TOP10 colonies on chloramphenicol plates (FigXA), indicating that plasmid DNA in the disruptate was able to transform TOP10 cells.
Transformation with disruptate of W3110 harbouring the BBa_K729019 nuclease plasmid yielded no colonies on chloramphenicol plates (FigXC). BBa_K729004 differs from BBa_K729006 only in that it contains an ORF encoding the periplasmic nuclease instead of laccase. As such, we conclude the periplasmic nuclease has sufficient DNAase activity in the disruptate to reduce the amount of plasmid DNA present to below the threshold capable of transforming TOP10 cells.
We suggest periplasmic nuclease expression provides a promising strategy for preventing transfer of genetically modified DNA. This is particularly valuable for the application of synthetic biology in environmental contexts.

17-3

17-4
 "
"