Synechocystis PCC 6803 growth curve
When we started working in the project, we realized that although we could manage
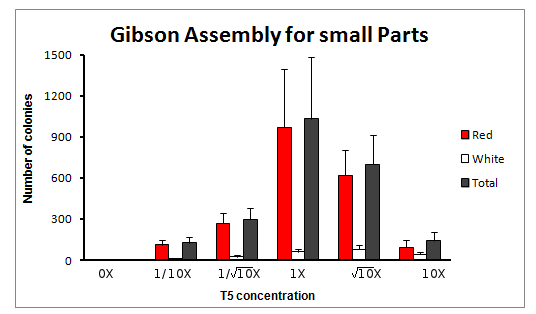
Gibson Assembly for small parts
During the construction of our first plasmid, we had many problems dealing with getting correct assemblies of the constructs which involved small parts (< 200bp). We found that for those kind of assemblies, people would recommend using as much as 5 times more DNA from the smaller insert in relation to the amount of plasmid backbone, arguing that such a small insert would be completely chewed up by the T5 exonuclease when in small amount.
Our analysis of the reaction gave us notion that theoretically, the number of resulting colonies would be in direct relation to the amount of available backbone. However, the total number of true positives would only be influenced by the ratio at which the inserts could compete with the assembly of aberrant constructs, such as the one described by the Washington University 2011 iGEM Team about selfcomplementarity of the NotI sequences embedded in the prefix and suffix.
Having in mind that the small part would be chewed up by the T5 exonuclease and that with no insert available the remaining backbone would end up assembling with its selfcomplementary ends, we decided to see if we could test if by changing the amount of T5 exonuclease in the reaction we could improve our rate of true positives.
To challenge this, we designed a Gibson Assembly which involved a small part of mRFP1 and a large backbone for it to assemble. Our design included a strategy to quickly identify those colonies which where true positives from false positive.
We used pSB4K5-J04450 and selected a portion of mRFP1 which would measure 100bp plus the 40bp from the overlaps. The part being in the middle of the mRFP1, that way if assemble is succesful, then you'll obtain the wild type mRFP1 making the colony visibly red.
We situated the small amplicon in such a way that if by any chance there was recircularization of the backbone, the resulting juncture would provide a helpful in-frame TAA stop codon to finish translation and evidence a false positive.
The experiment was done for 6 different concentrations of T5 exonuclease, in squareroot of 10 magnitude changes of the original T5 concentration. The concentrations were: 0X, 1/10X, 1/sqrt(10)X, 1X, sqrt(10)X and 10X T5 exonuclease.
Methods for the preparation of the experiment can be found in the protocols section.
Total count of colonies
We obtained varying amount of number of colonies, with a clear tendency to increase towards the reaction with 1X concentration of T5 exonuclease in the Master Mix.
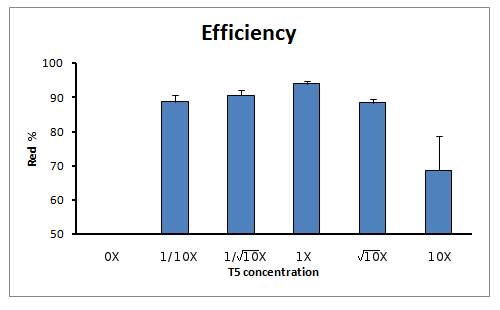
Efficiency of reaction
When calculating the efficiency of the reaction (in terms of true positive rate) we also see a clear tendency towards the reaction with 1X concentration of T5 exonuclease in the Master Mix.
Conclusion
According to the results we obtained in this experiment we conclude that according to our DNA concentration conditions (0.00125 pmoles/uL) and ratio of insert to backbone (5:1), the Gibson Assembly reaction is already optimized for small parts.
LuxBrick Characterization - (BBa_K325909)
As a part of our project, we set out to investigate the conditions which affect the output of the LuxBrick. As we are using various elements from this Biobrick, we believe that is cornersome to know how we may increase its output by modifying the conditions that affect its behaviour.
We set out to understand diverse factors that could be influencing the total bioluminescence yield of the brick. Some of them have direct relation with some of the aspects of our project. These factors are:
Glucose concentration
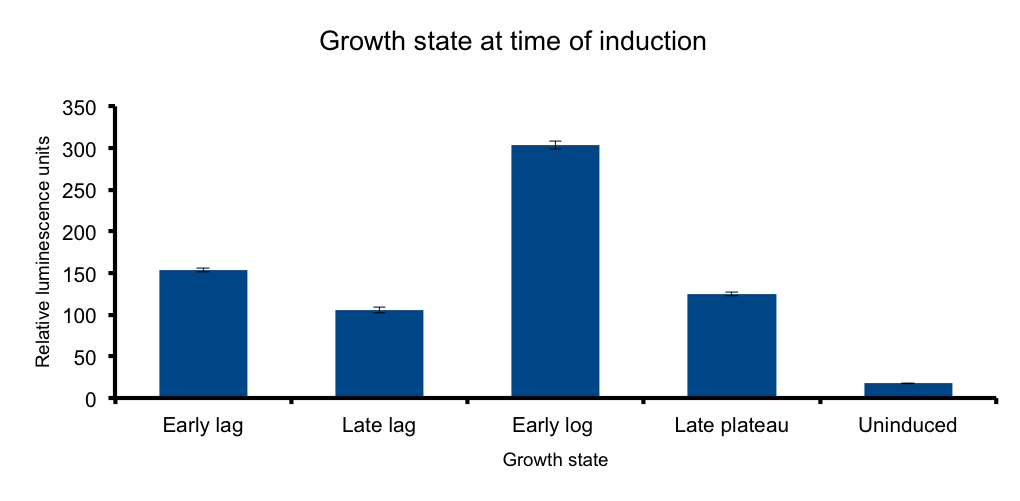
Growth state at time of induction
Cell mass
Temperature
Here you'll find detailed the characterization methods we used in this experiment.
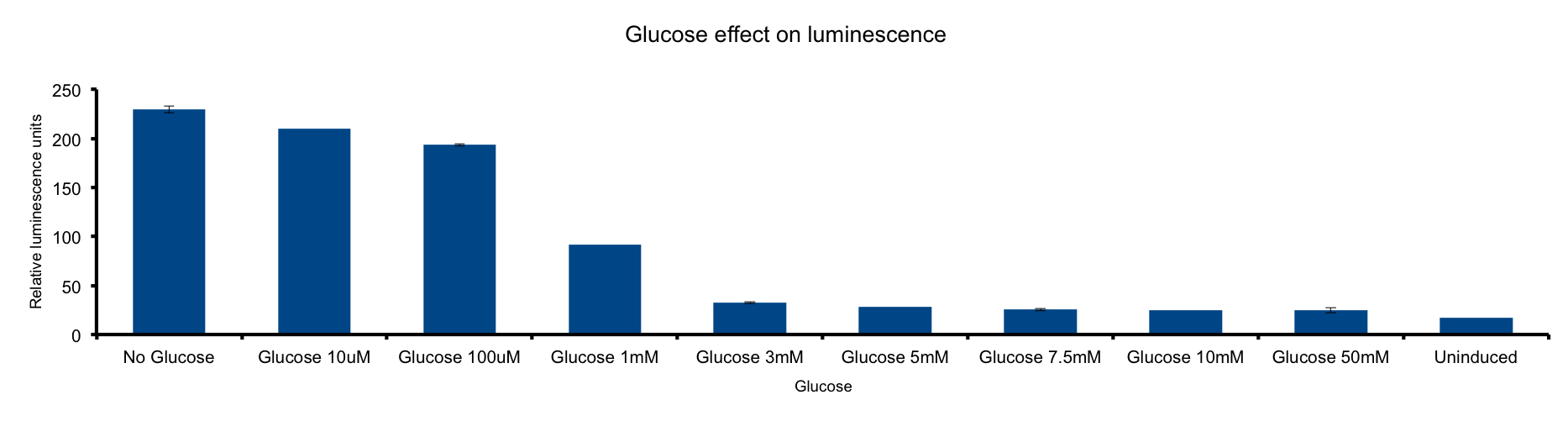
Glucose concentration in media
HERE THERE SHOULD BE SOME TEXT SUPPORTING THE EXPERIMENT HOPEFULLY WITH CITATION.
FOR EXAMPLE:
Glucose is most important energy source of bacteria. As glucose is catabolized into pyrovate ...
Growth state at-time-of-induction
HERE THERE SHOULD BE SOME TEXT SUPPORTING THE EXPERIMENT HOPEFULLY WITH CITATION.
It has been know for some time that general bacterial metabolism is principally determined its growth state. Growth states involve the expression of different sigma factors according to the metabolic status of the bacteria. The transcription of certain sequences directly depend on this, as some promoters are only read by the RNA polymerase when sigma70 is present in the cell. (TO BE CONTINUED)
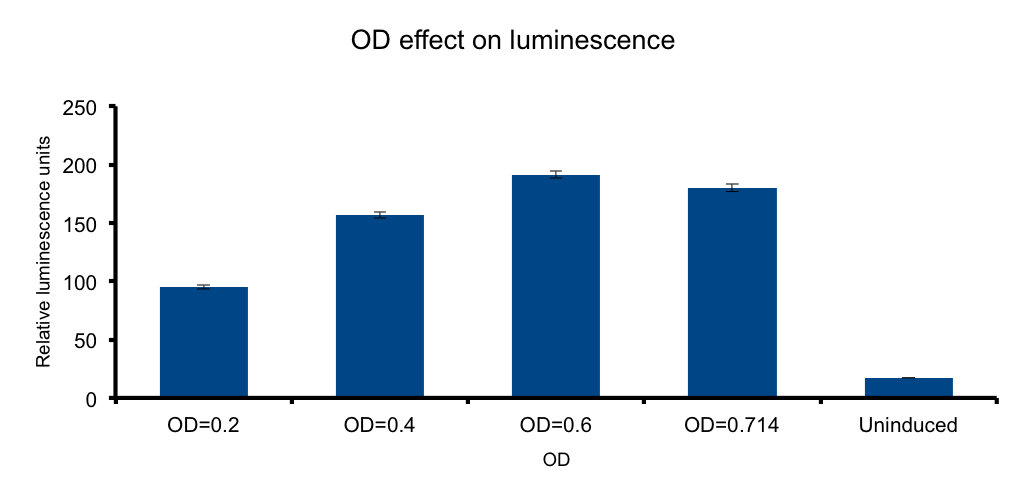
Cell mass (Optical Density)
Photons, cells. Impedance?
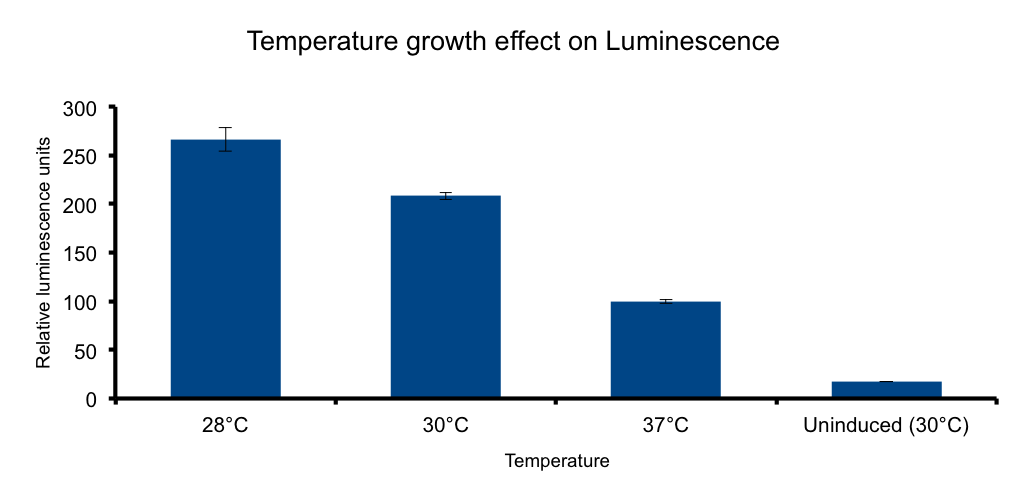
Growth temperature
HERE THERE SHOULD BE SOME TEXT SUPPORTING THE EXPERIMENT HOPEFULLY WITH CITATION.
Protein folding, vibrio fisherii free living organism... blablabla
sfGFP vs mRFP1
Improvement of an existing BioBrick
Our chosen target was Biobrick J04450 which is a RFP Coding Device (link: http://partsregistry.org/Part:BBa_J04450). Its usage as a cloning tool has increased in time due to the easy detection of transformed colonies to the naked eye. Nonetheless, the advancement of automatic measurement systems (and yada yada) opens up a whole new range of applications in which markers are detected by highly sensitive machines and not by human observers. In these cases mRFP does not provide a significant advantage over other marker systems. In fact, it could be detrimental for the efficiency of the experiment (?)
Therefore, we propose to exchange part mRFP for sfGFP. sfGFP would prove to be a better part for the following reasons:
sfGFP is of shorter length than mRFP1 (proposed implication: Lower metabolic cost. )
sfGFP has higher quantum yield/faster maturation rate or something of the sort!
To demonstrate the improvement of the biobrick, we designed and run the following experiments:
Experiment : Culture growth
Three flasks were inoculated in equal concentrations with: psB4K5-J04450, psB4K5 – ours and wildtype TOP 10 cells respectively. Inoculum concentrations were checked by culture OD measurement at 600 nm to normalize the amount of bacteria that would be initially present.
Then, OD measurements at 600 nm, 584 nm and 492 nm were reported every 30 minutes for each of the flasks. 584 nm and 492 nm are the peak absortion wavelengths of mRFP1 and sfGFP respectively.
Insert:Explanation of why this should prove improvement
Results 1. Growth rate
Experimental data was adjusted in order to….
Waiting for results ☺
Results 2.
Experimental data was adjusted in order to….
Waiting for results ☺
References
 "
"