Team:Chalmers-Gothenburg/Biodetection of hCG
From 2012.igem.org
Biodetection of hCG hormone
Saccharomyces cerevisiae will be modified in such a way that the yeast cell should function as a biosensor for the human chorionic gonadotropin hormone (hCG). This hormone is produced in the body during pregnancy and consequently, the idea for the biosensor is to function as a simple pregnancy test. To construct the biosensor, the Ste2 receptor, member of the G protein-coupled receptor (GPCR) family, in the yeast pheromone signaling pathway will be replaced by the human luteinizing hormone receptor (LH/CG). The LH/CG receptor binds to hCG with high affinity. As an output signal, the expression of indigo producing enzymes will be coupled with the pheromone pathway. The picture below shows an overviewed illustration of our biological system.
The yeast strain that will be used is provided by the Kondo group from Kobe University in Japan. It has the endogenous GPCR Ste2 deleted in order to prevent it from isolating G proteins in inactive complexes. Sst2, a negative feedback regulator of the pheromone pathway is also knocked out. The strain contains a yeast/human chimeric Gα subunit which should ensure the interaction of the human GPCR with the yeast pheromone pathway. Binding of hCG to the LH/CG receptor will result in the activation of the chimeric G protein, the activation of the pheromone pathway and consequently the expression of indigo synthesizing enzymes.
Our project includes the constitutive expression of the human luteinizing hormone receptor (LHCGR). Two signal peptides will be tested when expressing the receptor, the native signal and a yeast membrane protein signal. The genes tnaA and fmo, encoding tryptophanase and flavin-containing monooxygenase respectively, will also be introduced into the yeast strain. These enzymes catalyze the conversion of tryptophan to indigo. TnaA will be coupled with the pheromone pathway by fusing it to the pheromone-induced FIG1 promoter whereas the fmo will be expressed constitutively. Another part of the project is the deletion of the ‘’CWP2’’ gene, encoding a cell wall mannoprotein, in order to increase the cell wall permeability. This is done in oder to enhance the chances of the hCG hormone to pass the yeast cell wall and to bind to the receptor.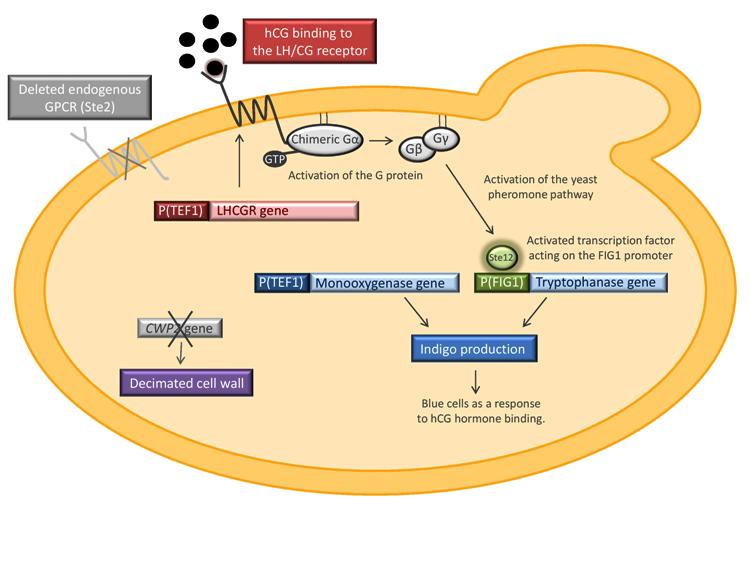
’’’An overview of the biological construct that should serve as a biosensor for hCG.’’’ The LH/CG receptor is expressed in yeast. STE2 and SST2 are deleted and the strain expresses a human/yeast chimeric Gα subunit. The tryptophanase gene is under the control of the pheromone-induced FIG1 promoter whereas the monooxygenase is expressed constitutively. CWP2 is removed in order to increase the cell wall permeability.
 "
"