Team:NTNU Trondheim/Project
From 2012.igem.org
| Line 21: | Line 21: | ||
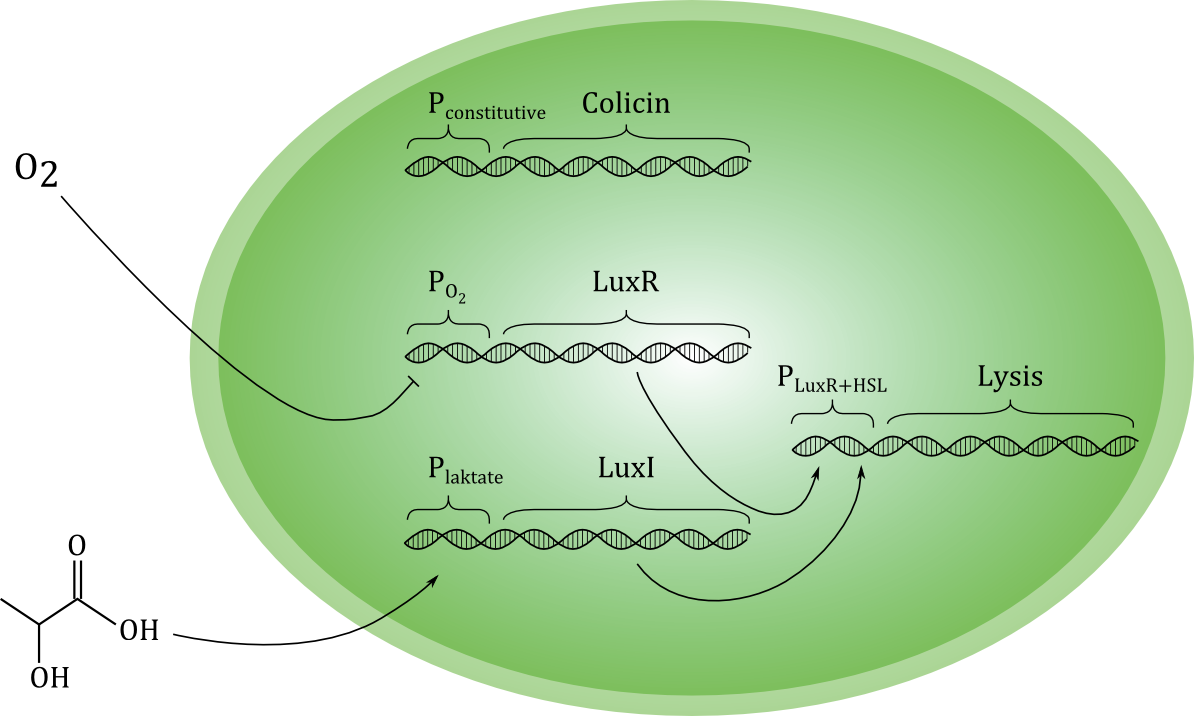
Differentiated cells (“normal” cells) can produce high amount of lactate in an anaerobic environment, but this is not a normal environment for the cells in our body. Under normal circumstances, the differentiated cells will primarily metabolize glucose to carbon dioxide in the citric acid cycle (TCA), and have a very low production of lactate. Cancer cells behave differently. They produce large amounts of lactate regardless of the oxygen concentration in their surroundings [http://www.ncbi.nlm.nih.gov/pubmed/19460998], [http://cancerres.aacrjournals.org/content/58/7/1408.full.pdf+html]. The reason that we want to use oxygen as a trigger factor is that tumors exists in an environment deprived of oxygen [http://www.springerlink.com/content/a60537w7085574v4/]. | Differentiated cells (“normal” cells) can produce high amount of lactate in an anaerobic environment, but this is not a normal environment for the cells in our body. Under normal circumstances, the differentiated cells will primarily metabolize glucose to carbon dioxide in the citric acid cycle (TCA), and have a very low production of lactate. Cancer cells behave differently. They produce large amounts of lactate regardless of the oxygen concentration in their surroundings [http://www.ncbi.nlm.nih.gov/pubmed/19460998], [http://cancerres.aacrjournals.org/content/58/7/1408.full.pdf+html]. The reason that we want to use oxygen as a trigger factor is that tumors exists in an environment deprived of oxygen [http://www.springerlink.com/content/a60537w7085574v4/]. | ||
| - | == | + | ==Overview== |
Our goal for this years competition is to create a genetic circuit which enables ''E. coli'' cells to detect and attack cancer cells. To do this, the cells should produce a tumor inhibitor or toxin effective at killing cancer cells, and respond to environmental cues indicating the presence of cancer cells by undergoing [http://en.wikipedia.org/wiki/Lysis lysis], releasing the anti-cancer agent into the surrounding environment. In principle, bacteria could be used in "search and destroy" missions against cancer inside the human body. We wish to develop our genetic circuit as a proof-of-concept of one such strategy. | Our goal for this years competition is to create a genetic circuit which enables ''E. coli'' cells to detect and attack cancer cells. To do this, the cells should produce a tumor inhibitor or toxin effective at killing cancer cells, and respond to environmental cues indicating the presence of cancer cells by undergoing [http://en.wikipedia.org/wiki/Lysis lysis], releasing the anti-cancer agent into the surrounding environment. In principle, bacteria could be used in "search and destroy" missions against cancer inside the human body. We wish to develop our genetic circuit as a proof-of-concept of one such strategy. | ||
| Line 35: | Line 35: | ||
As shown in the sketch, we plan to express the toxin-producing gene with a constitutive promoter. This means that the toxin production always be on. Ideally, the toxin should be maximally effective against cancer cells and minimally toxic to the toxin-producing cell. We have evaluated several candidate molecules. At the moment, we are leaning towards using the protein [http://proteopedia.org/wiki/index.php/Colicin_E1 Colicin E1]. | As shown in the sketch, we plan to express the toxin-producing gene with a constitutive promoter. This means that the toxin production always be on. Ideally, the toxin should be maximally effective against cancer cells and minimally toxic to the toxin-producing cell. We have evaluated several candidate molecules. At the moment, we are leaning towards using the protein [http://proteopedia.org/wiki/index.php/Colicin_E1 Colicin E1]. | ||
| - | == | + | ==Details== |
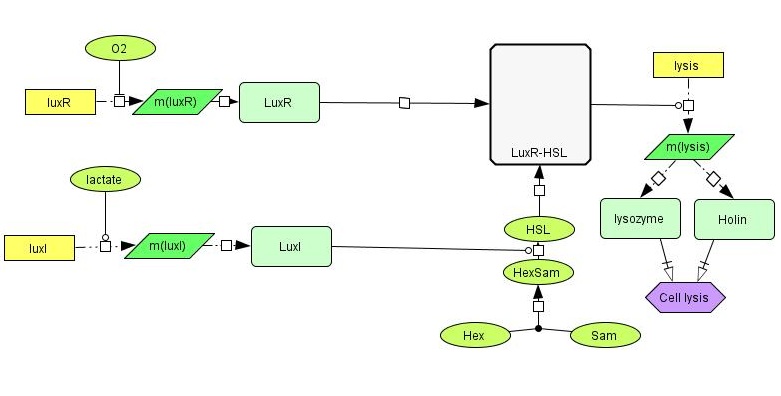
Below is a more detailed sketch of the circuit design ([https://2012.igem.org/File:NTNU_sketchlegend.jpg legend]). The environmental cues oxygen and lactate regulate the production of two regulatory proteins, LuxR and LuxI. Oxygen inhibits the production of LuxR, while lactate activates the production of LuxI. LuxI in turn catalyzes the formation of a homo-serine lactone (HSL) compound from S-Adenosyl Methionine (SAM) and Hexanoyl-ACP (Hex). When both LuxR and HSL is produced, they combine irreversibly to form a complex which activates the promoter in front of the lysis device (BioBrick part <partinfo>BBa_K112808</partinfo>, designed by the [https://2008.igem.org/Team:UC_Berkeley University of California Berkeley iGEM 2008] team). | Below is a more detailed sketch of the circuit design ([https://2012.igem.org/File:NTNU_sketchlegend.jpg legend]). The environmental cues oxygen and lactate regulate the production of two regulatory proteins, LuxR and LuxI. Oxygen inhibits the production of LuxR, while lactate activates the production of LuxI. LuxI in turn catalyzes the formation of a homo-serine lactone (HSL) compound from S-Adenosyl Methionine (SAM) and Hexanoyl-ACP (Hex). When both LuxR and HSL is produced, they combine irreversibly to form a complex which activates the promoter in front of the lysis device (BioBrick part <partinfo>BBa_K112808</partinfo>, designed by the [https://2008.igem.org/Team:UC_Berkeley University of California Berkeley iGEM 2008] team). | ||
Revision as of 17:07, 25 September 2012
 "
"