Team:Wageningen UR/OutsideModification
From 2012.igem.org
m (→Polerovirus) |
|||
| Line 1: | Line 1: | ||
{{Template:WUR}} | {{Template:WUR}} | ||
| + | |||
| + | {{Template:WUR_PO}} | ||
= Outside Modification = | = Outside Modification = | ||
Revision as of 22:27, 26 September 2012
Contents |
Outside Modification
Introduction
The monomers of virus-like particles (VLPs) have been subject to many modifications of which some are aimed at changing the appearance of the particle. By changing the outside, the VLP acquires new properties which have been used mainly in vaccine development [1,2]. The modification we pursue is adding a coil to the protein subunits, at any location that is exposed on the outside of the VLP, as schematically shown in figure 1. This can be a fusion to a C or N-terminal, but a modification in a loop is possible as well. The goal of the coil is to serve as a docking site for ligands which are modified to contain a coil subunit. A coil on the outside plays a rather important role expanding the applications of VLPs. It forms the link between the VLP and a functional group, such as a ligand or antigen.
Aim: Construct a Virus-Like Particle that has the K-coil exposed on the outside
CCMV
While Hepatitis B is modified multiple times and Polerovirus shows great opportunities to do so too, CCMV has not been modified on the outside yet. We have designed two possible modifications which might lead to the desired VLPs as described in the aim.
- Modification of an outside loop. This has shown to be possible with Hepatitis B, but the loops in CCMV seem to have more interactions
- Construct a mutant by deleting the first 25 amino acids, after which the K-coil is added. This might expose the K-coil on the outside of the VLP.
Results:
The full construct of the delta 25 mutant was amplified and cloned. After sequencing and conformation of intended modifications, we found out that one of the many primers contained a minor mistake having major implications: a frameshift in the CCMV coat protein gene.
The primer that was supposed to add the flexible linker to the coat protein contained an minor mistake: One ‘G’ was not taken up in the primer, causing a frameshift. After all the subsequent steps were undertaken, we found that the whole coil was on the coat protein as intended. However, due to the mistake, the open-reading frame of the coat protein was a mess, creating an early STOP-codon. The figure below shows the new protein created, compared to wild-type CCMV.
Frame shift in delta 25 mutant primers
After receiving our samples from the sequencing, we saw that there was a frame shift in the sequence, leading to the faulty translation of the CCMV delta 25 coat protein, with an early stop codon.
| Faulty | Intended | |
| BP sequence |
GTTTCTTCGAATTCGCGGCCGCTTCTAGATGAAGATAGCGG
CGTTGAAGGAGAAAATCGCAGCACTAAAAGAAAAGATAGCG
GCGTTGAAGGAGCTTGGTGGTGGTTCTGGTGGTGGTGGTTC
TGCTGCTGCTGTGTGGTCCAACCTGTTATTGTAGAACCCAT
CGCTTCAGGCCAAGGCAAGGCTATTAAAGCATGGACCGGTT
ACAGCGTATCGAAGTGGACCGCCTCTTGTGCGGCTGCCGAA
GCTAAAGTAACCTCGGCTATAACTATCTCTCTCCCTAATGA
GCTATCGTCCGAAAGGAACAAGCAGCTCAAGGTAGGTAGAG
TTTTATTATGGCTTGGGTTGCTTCCCAGTGTTAGTGGCACA
GTGAAATCCTGTGTTACAGAGACGCAGACTACTGCTGCTGC
CTCCTTTCAGGTGGCATTAGCTGTGGCCGACAACTCGAAAG
ATGTTGTCGCTGCTATGTACCCCGAGGCGTTTAAGGGTATA
ACCCTTGAACAACTCACCGCGGATTTAACGATCTACTTGTA
CAGCAGTGCGGCTCTCACTGAGGGCGACGTCATCGTGCATT
TGGAGGTTGAGCATGTCAGACCTACGTTTGACGACTCTTTC |
GTTTCTTCGAATTCGCGGCCGCTTCTAGATGAAGATAGCGG
CGTTGAAGGAGAAAATCGCAGCACTAAAAGAAAAGATAGCG
GCGTTGAAGGAGCTTGGTGGTGGTTCTGGTGGTGGTGGTTC
TGCTGCTGCTGGTGTGGTCCAACCTGTTATTGTAGAACCCA
TCGCTTCAGGCCAAGGCAAGGCTATTAAAGCATGGACCGGT
TACAGCGTATCGAAGTGGACCGCCTCTTGTGCGGCTGCCGA
AGCTAAAGTAACCTCGGCTATAACTATCTCTCTCCCTAATG
AGCTATCGTCCGAAAGGAACAAGCAGCTCAAGGTAGGTAGA
GTTTTATTATGGCTTGGGTTGCTTCCCAGTGTTAGTGGCAC
AGTGAAATCCTGTGTTACAGAGACGCAGACTACTGCTGCTG
CCTCCTTTCAGGTGGCATTAGCTGTGGCCGACAACTCGAAA
GATGTTGTCGCTGCTATGTACCCCGAGGCGTTTAAGGGTAT
AACCCTTGAACAACTCACCGCGGATTTAACGATCTACTTGT
ACAGCAGTGCGGCTCTCACTGAGGGCGACGTCATCGTGCAT
TTGGAGGTTGAGCATGTCAGACCTACGTTTGACGACTCTTT |
| Primer FW1 FL-Delta26 |
TGGTGGTGGTTCTGGTGGTGGTGGTTCTGCTGCTGC TGTGTGGTCCAACCTGTTATTGTAG | TGGTGGTGGTTCTGGTGGTGGTGGTTCTGCTGCTGC
TGGTGTGGTCCAACCTGTTATTGTAG |

The new primer is ordered to still make this part available for the registry, but submission before the wiki deadline will not be possible anymore. However, we think that iGEM should not be the only reason to deliver bricks, and we intend to deliver all bricks that did not make it just in time, still after the iGEM deadlines.
However, this showed that the technique of extension-PCR worked, so the primers for TuYV and Hepatitis B should work as well. This provides a standardized approach to attach a coil to any protein; coat protein, ligand or epitope.
Polerovirus
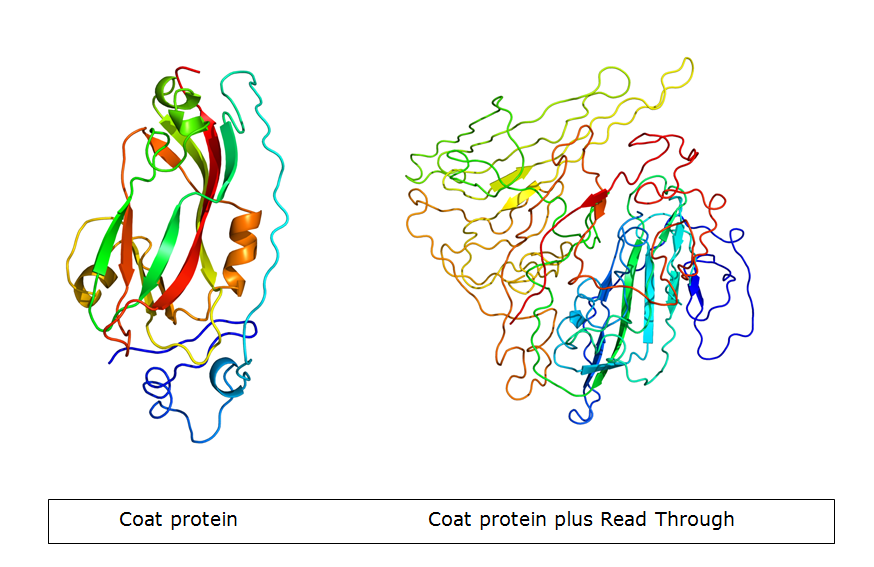
The Potato Leaf Roll Virus (PLRV) and Turnip Yellows Virus (TuYV) have a very interesting feature. Part of the subunits is expressed with a read-through product which forms spike-like structures on the outside of the VLP. The idea is adding the K-coil downstream of the coat protein gene, so it ends up in the spike. Because the spike is not involved in formation of the VLP, it will not change the wild-type properties of the particle during VLP formation. However, the spike structure makes more parts on the C terminus stick out. Consequently, the modification can be done more freely.
Results:
Both viruses have been isolated from either nature or provided plasmids. We cloned both the full gene including read-through and the gene without read-through. The primers have been designed to elongate the different versions of the gene, adding the K-coil. Transforming E.coli with the wild-type gene succeeded, but we did not manage to produce the subunits.
Hepatitis B
A loop exposed on the outside of Hepatitis B is known to accept modifications and for VLPs still [3]. Formation is improved when a mixture is made from wild-type and modified subunits. There is one problem concerning the insertion of the k-coil in the external loop: The coil will be bended, while it is designed to be linear. This can be solved by inserting a protease specific site next to the coil, as illustrated in figure 7. In this way, the coil can be linearized by cutting the protein after VLP formation [4]. This exposes the coil in a linearized formation, allowing attachment of the ligand. We designed a set of primers that adds the modification while amplifying the whole plasmid. By using 5'phosphorylated primers, it should be possible to ligate the new linearized backbones, yielding whole plasmids. Figure 8 shows this technique.
Results:
Expression and formation of wild-type VLPs succeeded. Multiple ways to modify the loop were tried, but without success.
References
1. Wang, Y.S., et al., Virus-like particles of hepatitis B virus core protein containing five mimotopes of infectious bursal disease virus (IBDV) protect chickens against IBDV. Vaccine, 2012. 30(12): p. 2125-30.
2. Crisci, E., J. Barcena, and M. Montoya, Virus-like particles: The new frontier of vaccines for animal viral infections. Vet Immunol Immunopathol, 2012. 148(3-4): p. 211-25.
3. Karpenko, L.I., et al., Insertion of foreign epitopes in HBcAg: how to make the chimeric particle assemble. Amino Acids, 2000. 18(4): p. 329-37.
4. Walker, A., et al., Internal core protein cleavage leaves the hepatitis B virus capsid intact and enhances its capacity for surface display of heterologous whole chain proteins. Journal of Biological Chemistry, 2008. 283(48): p. 33508-15.
 "
"