Team:Peking/Project/Luminesensor/Future
From 2012.igem.org
Jintanshadow (Talk | contribs) |
|||
| (37 intermediate revisions not shown) | |||
| Line 9: | Line 9: | ||
<div class="PKU_context floatR first"> | <div class="PKU_context floatR first"> | ||
<h3 id="title1">Motivation</h3> | <h3 id="title1">Motivation</h3> | ||
| - | <p>Light-oxygen-voltage (LOV) domain containing fusion proteins are validated photosensing devices that have been widely utilized and well characterized in the field of optogenetics. The use of Vivid (VVD) in the construction of an ultra-sensitive | + | <p>Light-oxygen-voltage (LOV) domain containing fusion proteins are validated photosensing devices that have been widely utilized and well characterized in the field of optogenetics. The use of the Vivid (VVD) protein in the construction of an ultra-sensitive <i>Luminesensor</i> (LexA-VVD) has opened the gate to a new generation of optogentics. (See <a href="/Team:Peking/Modeling/Channel">Project Luminesensor Spectrum Expanding</a>) |
<br/><br/>Functional the existing biobrick may be, there are two major concerns about it: </p> | <br/><br/>Functional the existing biobrick may be, there are two major concerns about it: </p> | ||
<ul> | <ul> | ||
| - | <li>1. Blue light absorption at 455nm ( | + | <li>1. Blue light absorption at 455nm (Figure 1) suffers from a disability of penetrating tissues for further applications such as phototherapy, because of its short wavelength. |
<div class="floatC"> | <div class="floatC"> | ||
<img src="/wiki/images/0/09/Peking2012_Sensor_Future_Redshift.jpg" alt="figure1" style="width:400px" /> | <img src="/wiki/images/0/09/Peking2012_Sensor_Future_Redshift.jpg" alt="figure1" style="width:400px" /> | ||
| - | <p class="description" style="text-align:center;font-style:normal;">Figure 1. Absorption Spectrum of VVD protein | + | <p class="description" style="text-align:center;font-style:normal;">Figure 1. Absorption Spectrum of VVD protein<sup>[3]</sup></p> |
</div> | </div> | ||
</li> | </li> | ||
| Line 22: | Line 22: | ||
</ul> | </ul> | ||
<p> | <p> | ||
| - | Thus, | + | Thus, we are greatly motivated by the thought of developing a red-shifted VVD domain for the <i>Luminesensor</i>. And such a dual channel system will sure see a bright future in its complexity and diversity. |
</p> | </p> | ||
</div> | </div> | ||
| Line 28: | Line 28: | ||
<h3 id="title2">Rationale</h3> | <h3 id="title2">Rationale</h3> | ||
<p> | <p> | ||
| - | The spectroscopic characters of VVD is mostly determined by its chromophore—Flavin Adanine Dinucleotide (FAD) substrate. | + | The spectroscopic characters of VVD is mostly determined by its chromophore—Flavin Adanine Dinucleotide (FAD) substrate. Therefore, any changes to the absorption spectrum should be focused on modifications to the conjugating substructure of FAD.<br/><br/> |
| - | One essential prerequisite for the incorporation of an FAD analogue is that the VVD pocket is big enough to accommodate a structurally extended FAD. | + | One essential prerequisite for the incorporation of an FAD analogue is that the VVD pocket is big enough to accommodate a structurally extended FAD. The protein structure of VVD shows it’s possible for substitution in 8 position of riboflavin -- the light absorbing group of FAD (Fig. 2&3). We can see a large space, measured by dash lines (unit: Angstrom), is vacant for future incorporation of an 8-substituted FAD analogue. |
</p> | </p> | ||
<div class="floatC"> | <div class="floatC"> | ||
<img src="/wiki/images/1/14/Peking2012_luminesensor_future_2.gif" alt="figure2 goes here" /> | <img src="/wiki/images/1/14/Peking2012_luminesensor_future_2.gif" alt="figure2 goes here" /> | ||
| - | <p class="description"> | + | <p class="description" style="text-align:center;">Figure 2 Vancancy in the pocket of VVD<sup><a href="#ref2">[2]</a></sup></p> |
</div> | </div> | ||
<div class="floatC"> | <div class="floatC"> | ||
| - | <img src="/wiki/images/7/75/Peking2012_luminesensor_future_3.png" alt="figure3 goes here" /> | + | <img src="/wiki/images/7/75/Peking2012_luminesensor_future_3.png" alt="figure3 goes here" style="width:400px"/> |
| - | <p class="description"> | + | <p class="description" style="text-align:center;">Figure 3 Molecular structure of FAD</p> |
</div> | </div> | ||
</div> | </div> | ||
| Line 43: | Line 43: | ||
<h3 id="title3">Design</h3> | <h3 id="title3">Design</h3> | ||
<p> | <p> | ||
| - | The choice of the FAD analogue should have the following features: a red-shifted absorption spectrum and a substitution at 8 position. After selecting from of a pool of over 500 chemicals listed by similarity search on SciFinder, we identified 8- | + | The choice of the FAD analogue should have the following features: a red-shifted absorption spectrum and a substitution at 8 position. After selecting from of a pool of over 500 chemicals listed by similarity search on SciFinder, we identified 8-tetrahydropyrroriboflavin (Fig. 4) as a promising candidate for incorporation, which has a red shift of absorption at 510nm. |
</p> | </p> | ||
<div class="floatC"> | <div class="floatC"> | ||
| - | <img src="" alt="figure4 goes here" /> | + | <img src="/wiki/images/5/5c/Peking2012_luminesensor_future_4.png" alt="figure4 goes here" style="width:400px"/> |
| - | <p class="description"> | + | <p class="description" style="text-align:center;">Figure 4 Molecular Structure of 8-tetrahydropyrrole riboflavin</p> |
</div> | </div> | ||
<p> | <p> | ||
| - | On the other hand, in order to attain bio-orthoganality and to efficiently accommodate the analogue, necessary mutations should be done with the pocket of VVD. Molecular docking results (Fig. 5) indicate that this analogue interacts with the protein at four specific sites. These side chains serve as spatial hindrances in that they are within a distance of 3 | + | On the other hand, in order to attain bio-orthoganality and to efficiently accommodate the FAD analogue, necessary mutations should be done with the pocket of VVD. Molecular docking results (Fig. 5) indicate that this analogue interacts with the protein at four specific sites(residues marked in blue). These side chains serve as spatial hindrances in that they are within a distance of 3 angstrom (dash lines). Mutations of these residues will hopefully create enough space for the 8-substituted FAD analogue, in this case, a tetrahydropyrrole at the 8 position. |
</p> | </p> | ||
<div class="floatC"> | <div class="floatC"> | ||
| - | <img src="" alt=" | + | <img src="/wiki/images/c/c9/Peking2012_luminesensor_future_005.gif" alt="Spatial interactions" /> |
| - | <p class="description"> | + | <p class="description" style="text-align:center;">Figure 5 Spatial interactions between FAD analogue and the pocket of VVD<sup><a href="#ref2">[2]</a></sup></p> |
</div> | </div> | ||
</div> | </div> | ||
| Line 60: | Line 60: | ||
<h3 id="title4">Perspectives</h3> | <h3 id="title4">Perspectives</h3> | ||
<p> | <p> | ||
| - | A newly developed, dual channel photosensing system based on LexA-VVD will greatly broaden the territory of optogentics. | + | A newly developed, dual channel photosensing system based on LexA-VVD will greatly broaden the territory of optogentics. A red-shifted <i>Luminesensor</i> could be used in deep tissue phototherapy, non-invasive light control of neuron and many other fields. What’s more, the concept used in this design could be instructive for future endeavors in developing a spectroscopically different photosensor. |
</p> | </p> | ||
| + | </div> | ||
| + | <div class="PKU_context floatR"> | ||
| + | <h3 id="title5">Reference</h3> | ||
| + | <p></p> | ||
| + | <ul class="refer"><li id="ref1"> | ||
| + | 1. Wu Y. <i>et al.</i>(2012). Dynamic Modeling of Human 5-Lipoxygenase−Inhibitor Interactions Helps To Discover Novel Inhibitors. <i>J. Med. Chem.</i>, 55: 2597: 605 | ||
| + | </li><li id="ref2"> | ||
| + | 2. Zoltowski, B.D. <i>et al.</i>(2007). Conformational switching in the fungal light sensor Vivid. <i>Science</i>, 316: 1054-1057 | ||
| + | </li><li id="ref3"> | ||
| + | 3. Wang, X., Chen, X., and Yang, Y.(2012). Spatiotemporal control of gene expression by a light-switchable transgene system. <i>Nature Methods</i>, 9:266–269. | ||
| + | </li></ul> | ||
</div> | </div> | ||
</html>{{Template:Peking2012_Color_Epilogue}} | </html>{{Template:Peking2012_Color_Epilogue}} | ||
Latest revision as of 11:30, 26 October 2012
Motivation
Light-oxygen-voltage (LOV) domain containing fusion proteins are validated photosensing devices that have been widely utilized and well characterized in the field of optogenetics. The use of the Vivid (VVD) protein in the construction of an ultra-sensitive Luminesensor (LexA-VVD) has opened the gate to a new generation of optogentics. (See Project Luminesensor Spectrum Expanding)
Functional the existing biobrick may be, there are two major concerns about it:
- 1. Blue light absorption at 455nm (Figure 1) suffers from a disability of penetrating tissues for further applications such as phototherapy, because of its short wavelength.
Figure 1. Absorption Spectrum of VVD protein[3]
- 2. Mono-channel manipulation of cellular behavior is not enough for future extensions of more complexity
Thus, we are greatly motivated by the thought of developing a red-shifted VVD domain for the Luminesensor. And such a dual channel system will sure see a bright future in its complexity and diversity.
Rationale
The spectroscopic characters of VVD is mostly determined by its chromophore—Flavin Adanine Dinucleotide (FAD) substrate. Therefore, any changes to the absorption spectrum should be focused on modifications to the conjugating substructure of FAD.
One essential prerequisite for the incorporation of an FAD analogue is that the VVD pocket is big enough to accommodate a structurally extended FAD. The protein structure of VVD shows it’s possible for substitution in 8 position of riboflavin -- the light absorbing group of FAD (Fig. 2&3). We can see a large space, measured by dash lines (unit: Angstrom), is vacant for future incorporation of an 8-substituted FAD analogue.
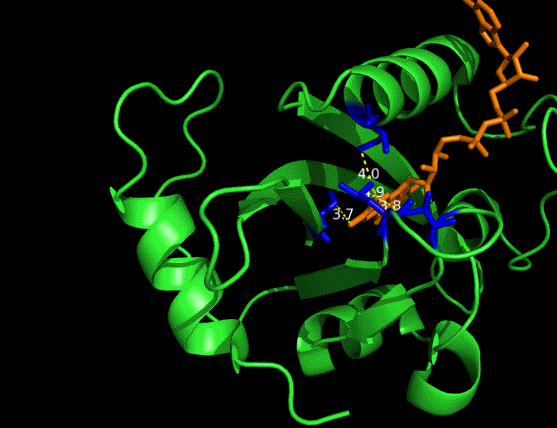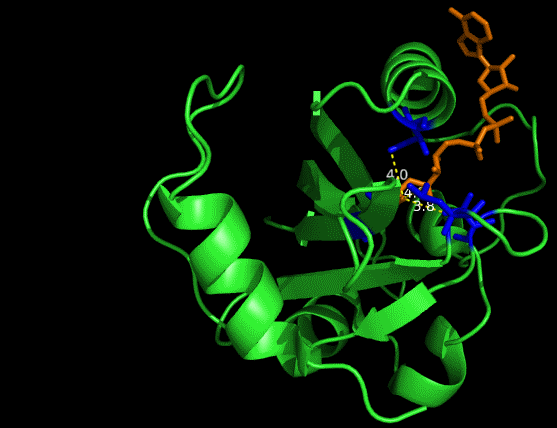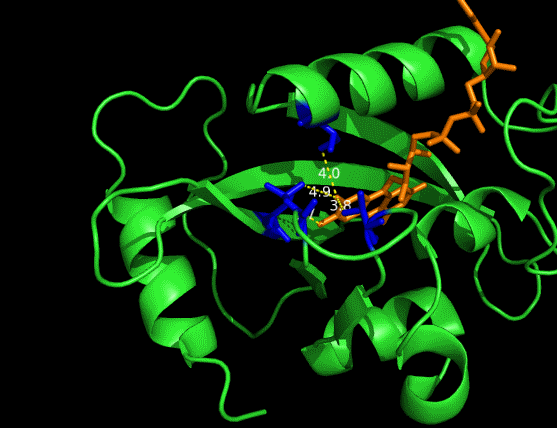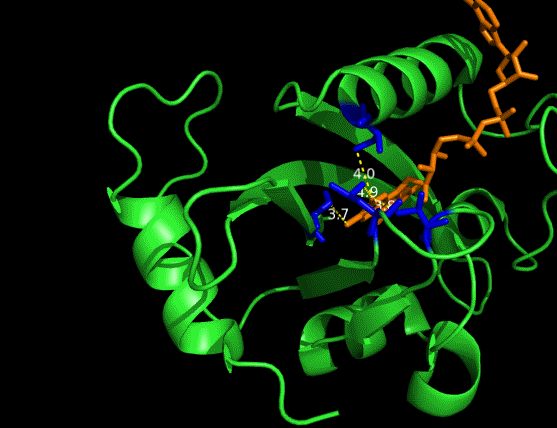
Figure 2 Vancancy in the pocket of VVD[2]
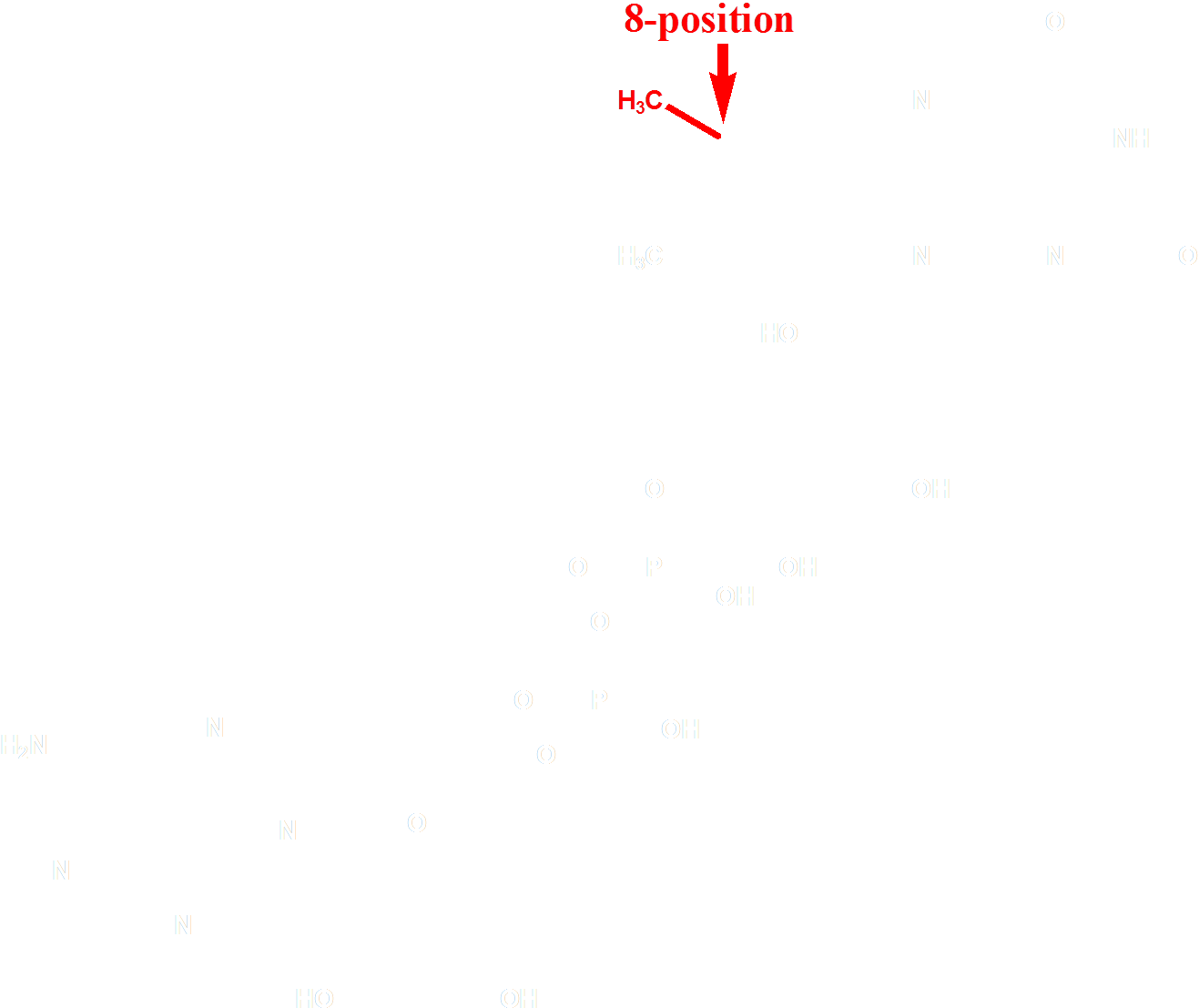
Figure 3 Molecular structure of FAD
Design
The choice of the FAD analogue should have the following features: a red-shifted absorption spectrum and a substitution at 8 position. After selecting from of a pool of over 500 chemicals listed by similarity search on SciFinder, we identified 8-tetrahydropyrroriboflavin (Fig. 4) as a promising candidate for incorporation, which has a red shift of absorption at 510nm.
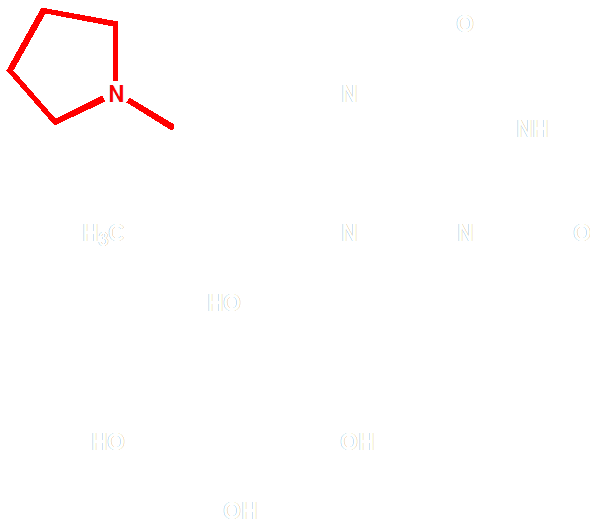
Figure 4 Molecular Structure of 8-tetrahydropyrrole riboflavin
On the other hand, in order to attain bio-orthoganality and to efficiently accommodate the FAD analogue, necessary mutations should be done with the pocket of VVD. Molecular docking results (Fig. 5) indicate that this analogue interacts with the protein at four specific sites(residues marked in blue). These side chains serve as spatial hindrances in that they are within a distance of 3 angstrom (dash lines). Mutations of these residues will hopefully create enough space for the 8-substituted FAD analogue, in this case, a tetrahydropyrrole at the 8 position.
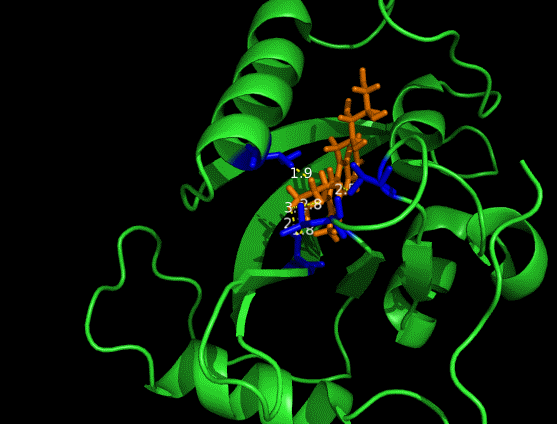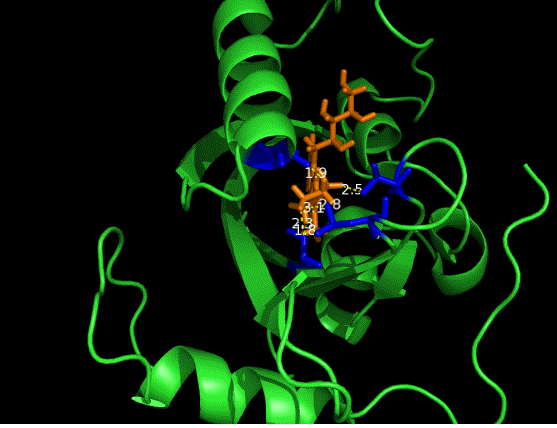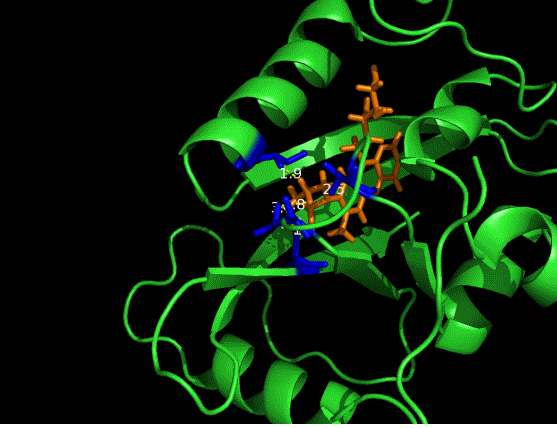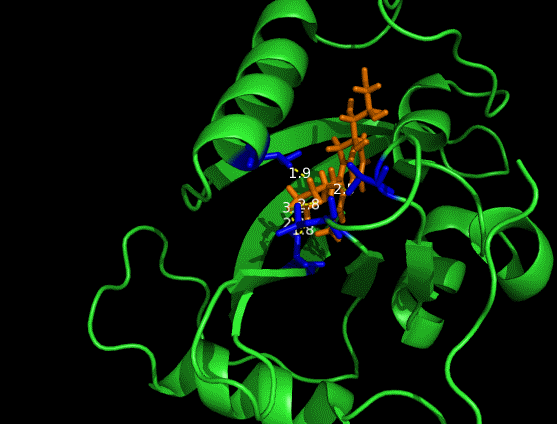
Figure 5 Spatial interactions between FAD analogue and the pocket of VVD[2]
Perspectives
A newly developed, dual channel photosensing system based on LexA-VVD will greatly broaden the territory of optogentics. A red-shifted Luminesensor could be used in deep tissue phototherapy, non-invasive light control of neuron and many other fields. What’s more, the concept used in this design could be instructive for future endeavors in developing a spectroscopically different photosensor.
Reference
- 1. Wu Y. et al.(2012). Dynamic Modeling of Human 5-Lipoxygenase−Inhibitor Interactions Helps To Discover Novel Inhibitors. J. Med. Chem., 55: 2597: 605
- 2. Zoltowski, B.D. et al.(2007). Conformational switching in the fungal light sensor Vivid. Science, 316: 1054-1057
- 3. Wang, X., Chen, X., and Yang, Y.(2012). Spatiotemporal control of gene expression by a light-switchable transgene system. Nature Methods, 9:266–269.
 "
"