Team:Peking/Project/Overview
From 2012.igem.org
m |
|||
| Line 11: | Line 11: | ||
<p> | <p> | ||
This summer, we have<br /> | This summer, we have<br /> | ||
| - | 1. helped 4 other iGEM teams by sharing DNA materials, characterizing their parts and modeling! <br /><br /> | + | 1. helped 4 other iGEM teams by sharing DNA materials, characterizing their parts and modeling! <a href="/Team:Peking/HumanPractice/Outreach/Collaboration"></i>Click Here</i></a><br /><br /> |
2. outlined and detailed a new approach of human pratice called "Sowing Tomorrow Synthetic Biologists"!<br /><br /> | 2. outlined and detailed a new approach of human pratice called "Sowing Tomorrow Synthetic Biologists"!<br /><br /> | ||
3. presented all fresh iGEMers with a collection and praise of historic iGEM projects to share and learn from each other!<br /><br /> | 3. presented all fresh iGEMers with a collection and praise of historic iGEM projects to share and learn from each other!<br /><br /> | ||
Revision as of 13:38, 21 September 2012
Achievements
This summer, we have
1. helped 4 other iGEM teams by sharing DNA materials, characterizing their parts and modeling! Click Here
2. outlined and detailed a new approach of human pratice called "Sowing Tomorrow Synthetic Biologists"!
3. presented all fresh iGEMers with a collection and praise of historic iGEM projects to share and learn from each other!
4. submitted 11 high quality and well-characterized standard biobricks!
5. improved the ease-of-use of "Lux Brick" that primarily constructed by Cambridge iGEM 2010 and carefully characterized its dynamics of functionality!
6. rationally constructed an ultra-sensitive genetically encoded sensor of luminance – what we call the Luminesensor and comprehensively characterized it. Luminesensor proved to be as sensitive to sense natural light and even bioluminescence!
7. successfully implemented spatiotemporal control of cellular behavior, such as high-resolution 2-D and 3-D bio-printing using dim light, and even utilizing the luminescence of an iPad!
8. successfully implemented cell-cell communication using light for the very first time in synthetic biology!
Therefore, we believe that we deserve a Gold Medal Prize.
1. Luminesensor
-- An Ultrasensitive Light Sensor
Based on comprehensive literature review, Peking iGEM has concluded that an ultra-sensitive light-sensing transcription factor may circumvent serious issues of current optogenetic methods, e.g. cytotoxicy, narrow dynamic range, and dependency on laser and exogenous chromophore.
Inspired by the general design principle of genetically encoded sensor in chemical biology, we fused a light-sensing domain, the smallest LOV (light, oxygen, or voltage) protein- Vivid (VVD) from Neurospora, to the DNA binding domain of bacterial LexA, a paradigm of bacterial repressor. When illuminated by blue light (440~480nm), driven by the dimerization of VVD, DNA binding domain of LexA will dimerize and bind to DNA, thus to repress the expression of downstream gene (Figure 1).
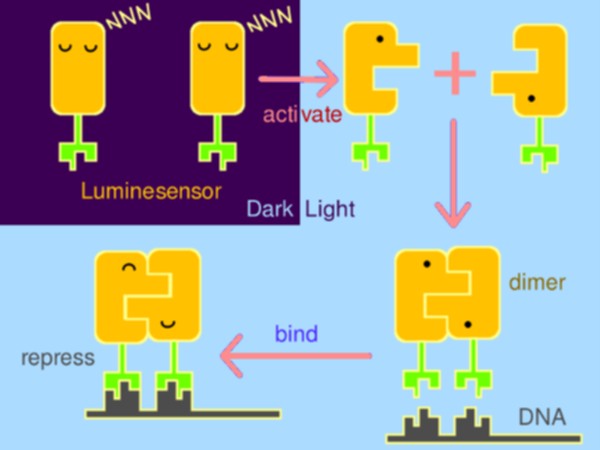
Figure 1. The basic design of Luminesensor.
After primary construction of fusion protein, protein structure inspection and kinetic simulations were conducted to rationally perform optimization. (If you would like to know details of Luminesensor design, please see Design page.)
Due to the consideration of minimal interference with host genetic context, a special mutation at LexA DNA binding domain was introduced to ensure the orthogonality with bacterial endogenous LexA protein. Resulted light-sensing protein proved to work very well in wild type E. coli strain without any necessity to genetically modify host (data in Characterization).
Consistent with the results of kinetic simulation (details in Modeling), mutagenesis based on kinetic data of VVD mutation significantly improved the performance of our light-sensing protein, called as Luminesensor, now with an amazing dynamic range reaching 300 (data in Characterization).
The high sensitivity of VVD protein is preserved in our Luminesensor. Amazingly, the Luminesensor proved to be sensitive to natural light and even bioluminescence (data in Characterization). Moreover, Luminesensor is able to respond to light signals with broad illuminance scales (Figure 2).

Figure 2. The broad illuminance scales which the Luminesensor could respond to.
To expand the toolkit of Luminesensor, we also tried to red-shift the absorption spectrum of VVD through molecular docking simulation (see Modeling) and VVD mutation (see Extension), which would largely extend the future application of Luminesensor, e.g. a multicolor Luminesensor control system.
2. Synthetic Biology in 2D and 3D
-- A Practical Application
Few optogenetics methods have been applied to practical application until now. To fully demonstrate spatiotemporally precise manipulation of cell behavior using our Luminesensor, Peking iGEM has deeply explored the application of Luminesensor in 2D and 3D bio-printing.
High resolution image could be easily obtained by 2D bioprinting on a bacterial lawn bearing Luminesensor (Figure 3). (more on 2D Printing).

Figure 3. High resolution of 2D bioprinting.
What's more, sharp images could be printed using an Apple iPad as light source (see iPrinting), which indicates that our Luminesensor could serve as the interface between biological systems and electrical devices.
A very promising applications of Luminesensor is 3D printing, using light signals of spatially high resolution. 3D images could be elegantly printed into media mixed with bacteria expressing Luminesensor (see 3D Printing). A new design strategy of 3D printer using holographic technology instead of layer-by-layer printing, was also proposed. With such a power tool, 3D printing regarding biological material has endless potentials for medical use (crazy ideas on Future).
3. Cell-Cell Light Communication
-- A Novel Strategy of Signaling
Cell-cell communication based on quorum sensing systems, e.g. AHL, has been largely utilized by synthetic biologists to construct gene circuits with complex functions, e.g. pattern formation and edge detection. However, the diffusion and saturation of quorum sensing chemicals limit the delivery of communication signals, which in turn requires high spatiotemporal resolution and long range interaction.
The ultrasensitivity of Luminesensor encouraged our team to explore the possibility of cell-cell light communication (Figure 4). By employing bacteria expressing lux operon from V. fisheri as light sender and those bearing Luminesensor as light receiver, we successfully demonstrated, for the first time ever, that light communication between cells could be achieved beyond direct physical contact (see video in Results). Through measuring the dynamics of light communication, it was proved that bioluminescence was sufficient to trigger the response of Luminesensor (data in Results).
Figure 4.
Our Light-Off system could be upgraded into Light-On system through simple design (details in Design), which enables us to achieve more flexible cell-cell light communication (creative ideas in Future).
4. Phototaxis
--Controlling Cell Motion by Light
What makes Luminesensor outstanding is not only its ultra-sensitivity, but also the particularly high dynamic range (data on Characterization). Such switch-like property makes Luminesensor a very reliable and beneficial module in synthetic biology. Similar to the light-controlled animal behavior in neuroscience, by coupling Luminesensor with endogenous systems, it is possible to integrate the light signal with cell motion.
Figure 5.
"Phototatic" bacteria can be built by reprogramming chemotaxis system in E. coli through light with Luminesensor (Figure 5). By controlling the expression of cheZ protein with light, the tumbling frequency is coupled to intensity of light signals (details in Design). On the border of light and dark fields, the motility ability of the cells in a single colony on the two sides is sufficient to result in an uneven colony (see Demonstration). The light-controlled cell motion has a very promising environmental and medical application for the future, e.g. the delivery of drugs to target concerns.
 "
"