Team:Peking/Project/Luminesensor/Characterization
From 2012.igem.org
Sensitivity
1. Set-up & Brief Procedure
We tested the sensitivity of Luminesensor by examine the light-dependent transcriptional activity of a GFP-ssrA reporter. ssrA is a protein tag that induces fast degradation of protein, which in our case facilitated the observation of transcriptional activity. Based on the consideration of guaranteeing accuracy and precision, our setup (Figure 2):
Figure 2. Setup
consists of three central parts: light source, incubator and 48-well plate. On account of high sensitivity, protecting the system from the preventable light exposure with the purpose of acquiring the accurate results which is the true reflection of our sensitivity is necessary. In order to solve the problems, we focus on two foremost aspects: utilizing attenuators to weaken the light intensity and using tin foil to avoid light leakage. For more details about how we conducting the experiment, see experiment procedures. In our experiments, illumination with different light intensity conditions at 460nm peak light from blue LED arrays for 16 hours show marked light-depressed reporter gene transcription, which indicates that under different blue light exposure conditions, there was hardly any light-induced reporter gene transcriptional activity. But when in the dark environment (packaged with three layers of aluminum foil), our systems showed extremely high GFP expression (Figure 3)
Figure 3. Expression of GFP.
There are a couple of plausible and conceivable reasons for why our system possesses such high sensitivity that can sense unbelievably weak light intensity. First, it includes the unusually stable, photo-activated state of VVD, which causes the system to be extremely sensitive to light. Secondly, without relying on the addition of chromophores, which might impede the growth of cells, our system can directly regulate gene expression without trade-off.
2. Results and Conclusions
Cells exposed to different light intensity expressing Luminesensor showed manifest light-repressed reporter gene transcription. As shown in the figure, all of the cells with dissimilar attenuators showed incredible repression efficiency (figure 4: luminance measurement of different attenuator).
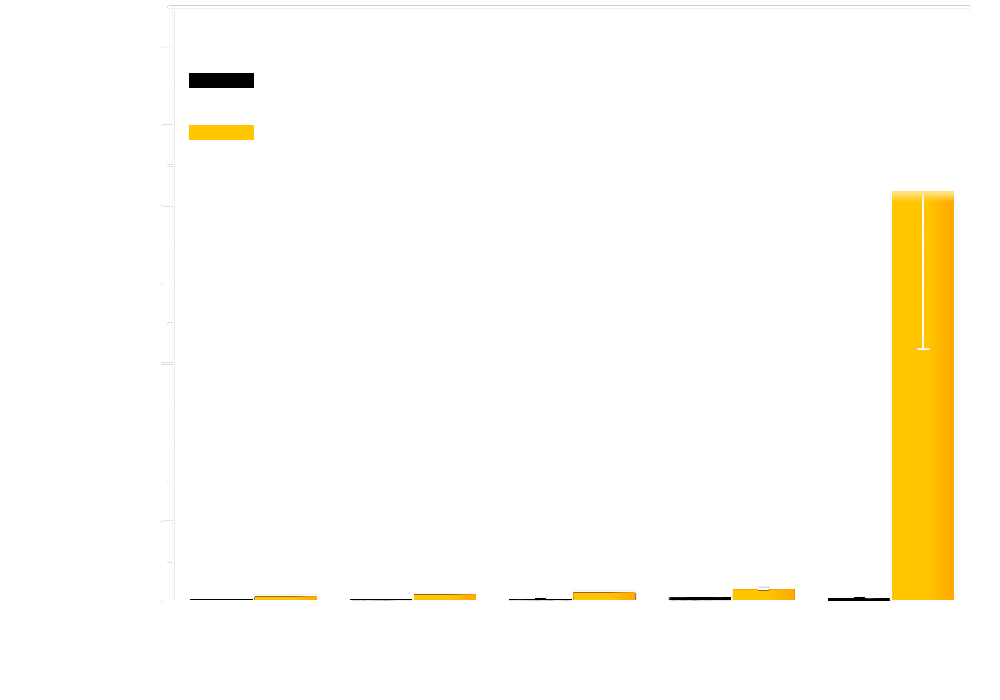
Figure 4. luminance measurement of different attenuator.
It proves that once the cells are exposed to natural light, the transcription of reporter gene will be strongly repressed, although still presents as a dose response. Besides, in the negative control group, which was entirely in the dark state, the expression of GFP ran up to a high degree of 50,000. As a matter of fact, as you can see later in "Results of light communication between cells", when we serially diluted light-emitting cells which expresses bacterial luciferase, the cells expressing Luminesensor presents significant dose response. Taking everything into account, our luminesensor does possess high sensitivity across several orders of magnitude.
We are impartially proud of accomplishing the goal of controlling gene expression using light without excessive or unnecessary energy or substrates, and we have respectable and appropriate reasons to have complete faith in our Luminesensor that it has imposed specific innovative challenges of light-dependent process for the future researchers.
Orthogonality Test
It is our biggest concern whether lexA408 Luminesensor works independently of endogenous LexA. If yes, maximizing its biological orthogonality will make Luminesensor a really plug-and-play device.
Two sections of testing expriments were carried out simultaneously. GFP was selected as a reporter and was fused downstream to 2 promoters (psulA408 and precA408) controlled by Luminesensor with 408 mutation and to 2 promoters (psulA and precA) controlled by Luminesensor without mutation. GFP expression is expected to have a negative relation with repression activity. To be more specific, higher level of green fluorescent indicates weaker repression effect; lower expression of GFP stands for stronger repression.
1. Experimental Design
To prove the orthogonality, facts that LexA408-VVD and endogenous LexA work totally independently are needed. Considering practical efficiency, 2 points of evidence are to collect:
- 1. Promoters psulA and precA are repressed in wild-type Ecoli strains while promoters psulA408 and precA408 are not blocked in wild-type strains;
- 2. LexA408-VVD Luminesensor efficiently represses its target under blue illumination, while it does not repress targets in total dark.
Figure 5. Shows observable color of plates where cell cultures growing under totally dark or light conditions. Different colors are used to indicate three levels of GFP expression.
2. Detailed Methods
Experiments were carried out in parallel: taking photos of plates as visual evidence; measuring GFP to quatitify the effect. Detailed protocols are as follows:
- 1. Visible display:
-
- Inoculate a single colony into 1.5ml centifuge tube, shaking cultivate cells at 250 rpm/min for 2-3 h at 30℃, then the cell culture are prepared for streaking;
- Streak cell culture onto right plates;
- Ioncubate in totally dark and light state at 30℃ for 1 or 2 days;
- Take photos of right plates;
- 2. Data collection:
-
- Inoculate a proper colony in liquid media;
- Shaking cultivate cells until platform stage at 30 ℃;
- Dilute celll cultures with selective media in proportion of 1:500;
- Shaking cultivate cells until platform stage at 30 ℃ in fully dark or under blue light;
- Harvest cells by centrifugation, resuspend pellets in PBS;
- Measuring GFP expression with 96-well-microplate reader;
3. Results
Below is a collage of our plates. The plates are placed in groups at 30℃ in either total dark or blue illumination. Visual results fit well with Figure 1.Visual look of plates were positive evidence for us.
Figure 6. Plates display. Streaking certain strain onto selected media.
Apart from those photos, more accurate results were obtained with Fluorescent Microplate Reader. Below are histograms based on our experimmental data. They clarify how fluoresence changes with lighting condition in different strains.
Figure 7. GFP expression under different promoters. The tests were carried out in strains capable of expressing LexA408-VVD.
It repeartedly turned out that:
- 1. reporters under wild-type promoters (psulA and precA) hardly express in either illuminated or dark state; their colonies are yellow. In contrast, GFPs fused to promoters psulA408 and precA408 were not blocked in strains, thus they looks bright green.
- 2. BL21 (DE3) strains expressing both Luminesensor and its reporters twist swiftly and sensitively from one states to the other, giving high definition output including lighting condition.
To sum up, LexA408-VVD Luminesensor works independently of host genetic context.
Improving Dynamic Performance
To test whether our designated mutations would improve the dynamic performance of Luminesensor in respect to reversibility and the on/off ratio, we co-transformed the psulA-GFP (GFP driven by luminesensor repressible promoter psulA) plasmid with four versions of Luminesensor plasmid into BL21 (ΔLexA ΔSulA): the original LexA-VVD(WT), LexA-VVD(I74V), LexA-VVD(M135I), and LexA-VVD(I74V+M135I). Resulted four strains were designated as LV-WT, LV-74, LV-135, LV-74-135, respectively. The overnight culture of the four strains were diluted 500 times and divided into two groups: one exposed to blue light and the completely wrapped with aluminum foil. After incubation for 16 hours, GFP expression levels were measured. As shown in figure below, LV-135 has an increased on/off ratio compared to the original LexA-VVD, while the LV-74 and LV-74-135 show reduced on/off ratios, which is in accordance with our modeling result.

Figure 8. Effects of introduced mutations on the performance (on/off ratio) of Luminesensor.
In order to determine whether the LexA-VVD(M135I) Luminesensor has enhanced reversibility in comparison to the original LexA-VVD, the temporal change of GFP expression level of dilated overnight culture of the strains LV-WT and LV-135 under blue light were measured at 2 hour intervals for 26 hours. As shown in figure below, the GFP expression level of both of the two strains began to rise after incubating at dark for about 10 hour. We speculated that the GFP expression level of the two strains is mainly determined by the equilibrium between GFP production and degradation and the dimerized LexA-VVD(WT) and LexA-VVD(135) dissociate in a shorter time scale compared to the time needed to establish the equilibrium of the GFP production and degradation.
Figure 9. The time course of GFP expression controlled by LV-WT and LV-135

Figure 9. The time course of GFP expression controlled by LV-WT and LV-135
Conclusion
In the end, we have finally acquired final version of Luminesensor that will function as a transcription repressor in response to blue light. And since we have introduced mutations into LexA DNA binding domain, the Luminesensor proved orthogonal to host genetic context. Futher introduced mutations in VVD photosensitive domain significantly improved the dynamic range of our optogenetic module. Equipped with this optimized LexA408-VVD74 Luminesensor, we are now able to apply it to practices, such as 2D bio-printing and cell-cell communication through light, as you will see later.
Reference
reference here
 "
"