Team:Tokyo Tech/Experiment/PHB
From 2012.igem.org
| Line 4: | Line 4: | ||
<div class="whitebox"> | <div class="whitebox"> | ||
| - | =PHB production= | + | =PHB production by <I>E.coli</I> & Confirmation of PHB= |
| + | |||
| + | To synthesize PHB by <I>E.coli</I>, we transformed <I>E.coli</I> JM109 with the constructed <I>PHA</I>C1-A-B1 part on pSB1C3 ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K934001 BBa_K934001]). <I>E.coli</I> JM109 is used to synthesize PHB, because it tends to have a high density accumulation of PHB([[#Reference|[5]]] | ||
| + | ). As a negative control, we transformed <I>E.coli</I> JM109 with PlasI-gfp on pSB1C3. | ||
| + | |||
| + | ==4-1 Confirmation of PHB synthesized on colonies== | ||
| + | |||
| + | We observed the accumulation of PHB in the <I>E.coli</I> colonies on Nile red positive medium under UV. Nile red has been widely used to stain colonies and distinguish between <I>PHA</I>-accumulating and non-accumulating colonies. Nile red in the agar medium doesn’t affect the growth of the cells, and the accumulation of <I>PHA</I>s in the colonies can be directly monitored([[#Reference|[3][4][5]]] | ||
| + | ). We cultured the transformant on LB agar medium plates with Nile red. After several days, colonies storing PHB were stained orange by Nile red when observed under UV. This result indicates that transformant synthesized and stored PHB. | ||
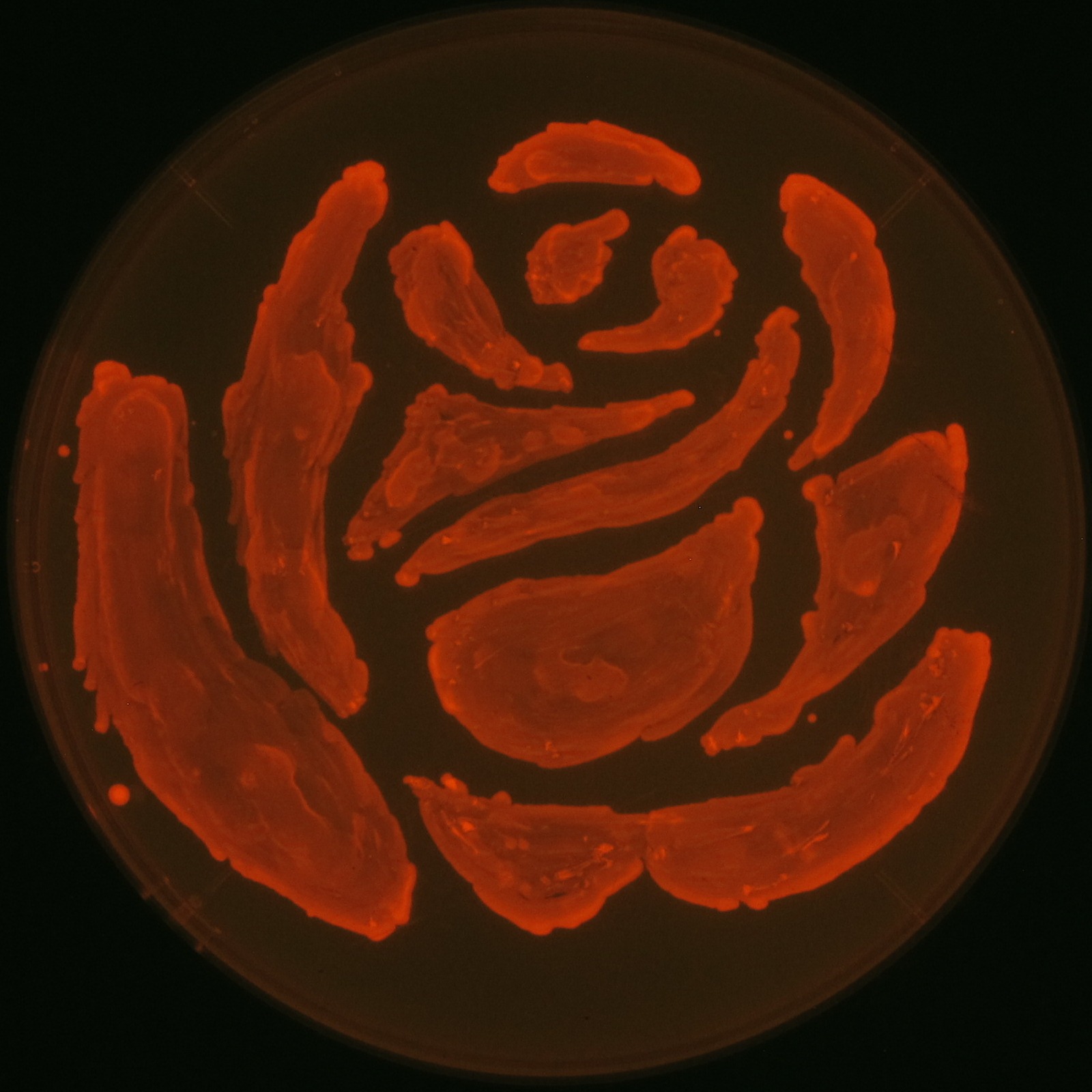
| + | Fig2-2-4-1 is the photographs of <I>E.coli</I> colonies on Nile red positive medium taken under UV. The orange colonies in Fig2-2-4-1A show that the accumulated PHB in cells was stained by Nile red. This result indicates that part [http://partsregistry.org/wiki/index.php?title=Part:BBa_K934001 BBa_K934001] synthesized PHB. Fig2-2-4-1B is the photograph of negative control cells. In this figure we observed that there were no remarkable colored colonies. Fig2-2-4-1-2 shows the difference between cells storing PHB and those not storing PHB on one plate. The cells in blue rectangle area are the cells with PHB synthesis gene and the cells in green rectangle area are the cells with PlasI-gfp gene as a negative control. Using the cells storing PHB, we drew a rose silhouette on the LB agar plate containing Nile red (Fig2-2-4-1-3).[[https://2012.igem.org/Team:Tokyo_Tech/Projects/PHAs/detail/index.htm#A_.PHB_production_on_colonies_and_preparation_before_confirmation_with_Nile_red_under_UV Protocol]] | ||
| + | [[File:tokyotech PHA Nilered1.png|300px|thumb|left|Fig2-2-4-1-1 <br>Fig2-2-4-1-1A: <I>E.coli</I> JM109 colonies with [http://partsregistry.org/wiki/index.php?title=Part:BBa_K934001 BBa_K934001] gene, PHB accumulation | ||
| + | <br>Fig2-2-4-1-1B: <I>E.coli</I> JM109 colonies with PlasI-gfp gene, no PHB accumulation]] | ||
| + | |||
| + | [[File:tokyotech PHA Nilered3.png|300px|thumb|left|Fig2-2-4-1-2, Difference between cells storing PHB and cells not storing PHB. <br>Blue rectangle: with [http://partsregistry.org/wiki/index.php?title=Part:BBa_K934001 BBa_K934001] gene, PHB accumulation. <br>Green rectangle: with PlasI-gfp gene, no PHB accumulation]] | ||
| + | [[File:tokyotech PHA make rose.png|150px|thumb|right|Fig2-2-4-1-3, Rose silhouette on the LB agar plate containing Nile red.]] | ||
| + | <br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br><br> | ||
| + | <br><br><br> | ||
| + | |||
| + | ==Confirmation of PHB accumulated in cells== | ||
| + | |||
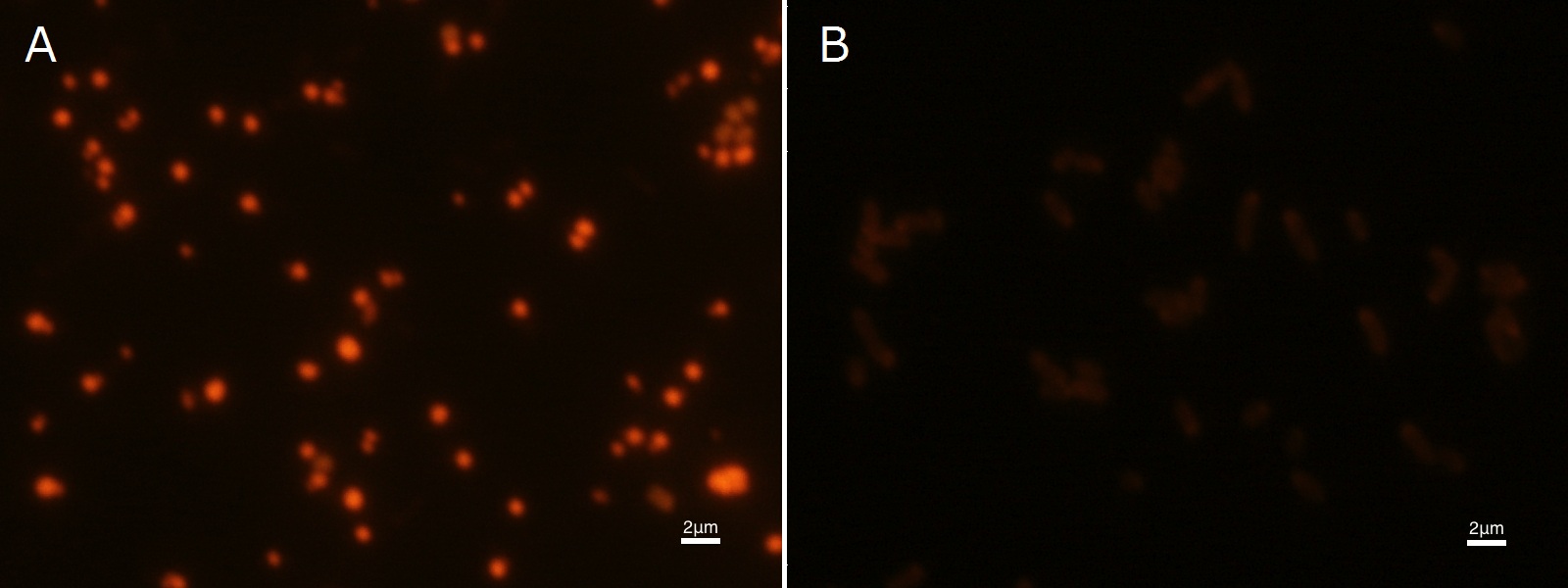
| + | To confirm the accumulation condition of PHB in <I>E.coli</I> with a microscope, we stained the PHB with Nile blue A reagent. Nile blue A is also used to detect the existence of PHB and has no toxicity to the cells([[#Reference|[5]]]). Before the observation, we stained the dried cells with Nile blue A solution. We then took photographs of the sample under fluorescence microscope. | ||
| + | Fig2-2-4-2-1 is the photograph of dried <I>E.coli</I> (with <I>PHA</I>C1-A-B1 gene) cells dyed with Nile blue A solution taken by fluorescence microscope. The fluorescent areas in Fig2-2-4-2-1A are the accumulated PHB in the cells. This result also indicates that part [http://partsregistry.org/wiki/index.php?title=Part:BBa_K934001 BBa_K934001] synthesized PHB. In the photograph of negative control (Fig2-2-4-2-1B), no remarkable fluorescent area was observed.[[https://2012.igem.org/Team:Tokyo_Tech/Projects/PHAs/detail/index.htm#B.PHB_production_in_cells_and_preparation_before_the_confirmation_with_Nile_blue_A Protocol]] | ||
| + | |||
| + | [[File:tokyotech PHA Nileblue1.png|500px|thumb|center| | ||
| + | Fig2-2-4-2-1A, <I>E.coli</I> JM109 dried cells with PHB accumulation stained by Nile blue A | ||
| + | Fig2-2-4-2-1B, <I>E.coli</I> JM109 dried cells without PHB accumulation stained by Nile blue A | ||
| + | ]] | ||
| + | |||
| + | <div id="tokyotech" style=" font:bold ;left ; font-size: 30px; color: #0000FF; padding: 2px;"> | ||
| + | 5.</div> | ||
Revision as of 04:28, 17 October 2012
Contents |
PHB production by E.coli & Confirmation of PHB
To synthesize PHB by E.coli, we transformed E.coli JM109 with the constructed PHAC1-A-B1 part on pSB1C3 (BBa_K934001). E.coli JM109 is used to synthesize PHB, because it tends to have a high density accumulation of PHB([5] ). As a negative control, we transformed E.coli JM109 with PlasI-gfp on pSB1C3.
4-1 Confirmation of PHB synthesized on colonies
We observed the accumulation of PHB in the E.coli colonies on Nile red positive medium under UV. Nile red has been widely used to stain colonies and distinguish between PHA-accumulating and non-accumulating colonies. Nile red in the agar medium doesn’t affect the growth of the cells, and the accumulation of PHAs in the colonies can be directly monitored([3][4][5] ). We cultured the transformant on LB agar medium plates with Nile red. After several days, colonies storing PHB were stained orange by Nile red when observed under UV. This result indicates that transformant synthesized and stored PHB. Fig2-2-4-1 is the photographs of E.coli colonies on Nile red positive medium taken under UV. The orange colonies in Fig2-2-4-1A show that the accumulated PHB in cells was stained by Nile red. This result indicates that part BBa_K934001 synthesized PHB. Fig2-2-4-1B is the photograph of negative control cells. In this figure we observed that there were no remarkable colored colonies. Fig2-2-4-1-2 shows the difference between cells storing PHB and those not storing PHB on one plate. The cells in blue rectangle area are the cells with PHB synthesis gene and the cells in green rectangle area are the cells with PlasI-gfp gene as a negative control. Using the cells storing PHB, we drew a rose silhouette on the LB agar plate containing Nile red (Fig2-2-4-1-3).[Protocol]
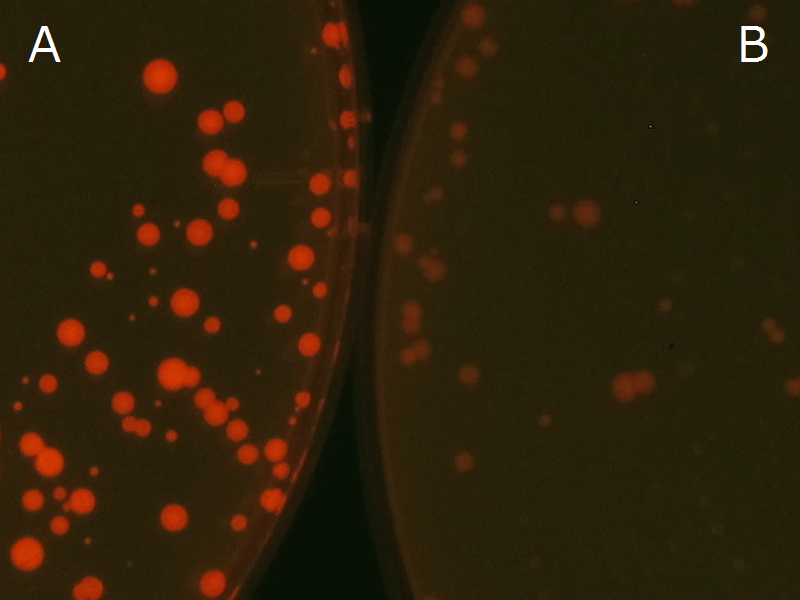
Fig2-2-4-1-1A: E.coli JM109 colonies with BBa_K934001 gene, PHB accumulation
Fig2-2-4-1-1B: E.coli JM109 colonies with PlasI-gfp gene, no PHB accumulation

Blue rectangle: with BBa_K934001 gene, PHB accumulation.
Green rectangle: with PlasI-gfp gene, no PHB accumulation
Confirmation of PHB accumulated in cells
To confirm the accumulation condition of PHB in E.coli with a microscope, we stained the PHB with Nile blue A reagent. Nile blue A is also used to detect the existence of PHB and has no toxicity to the cells([5]). Before the observation, we stained the dried cells with Nile blue A solution. We then took photographs of the sample under fluorescence microscope. Fig2-2-4-2-1 is the photograph of dried E.coli (with PHAC1-A-B1 gene) cells dyed with Nile blue A solution taken by fluorescence microscope. The fluorescent areas in Fig2-2-4-2-1A are the accumulated PHB in the cells. This result also indicates that part BBa_K934001 synthesized PHB. In the photograph of negative control (Fig2-2-4-2-1B), no remarkable fluorescent area was observed.[Protocol]
Construction of pha-C1-A-B1 in Biobrick format
[Back to "Construction of phaC1-A-B1 in Biobrick format"]
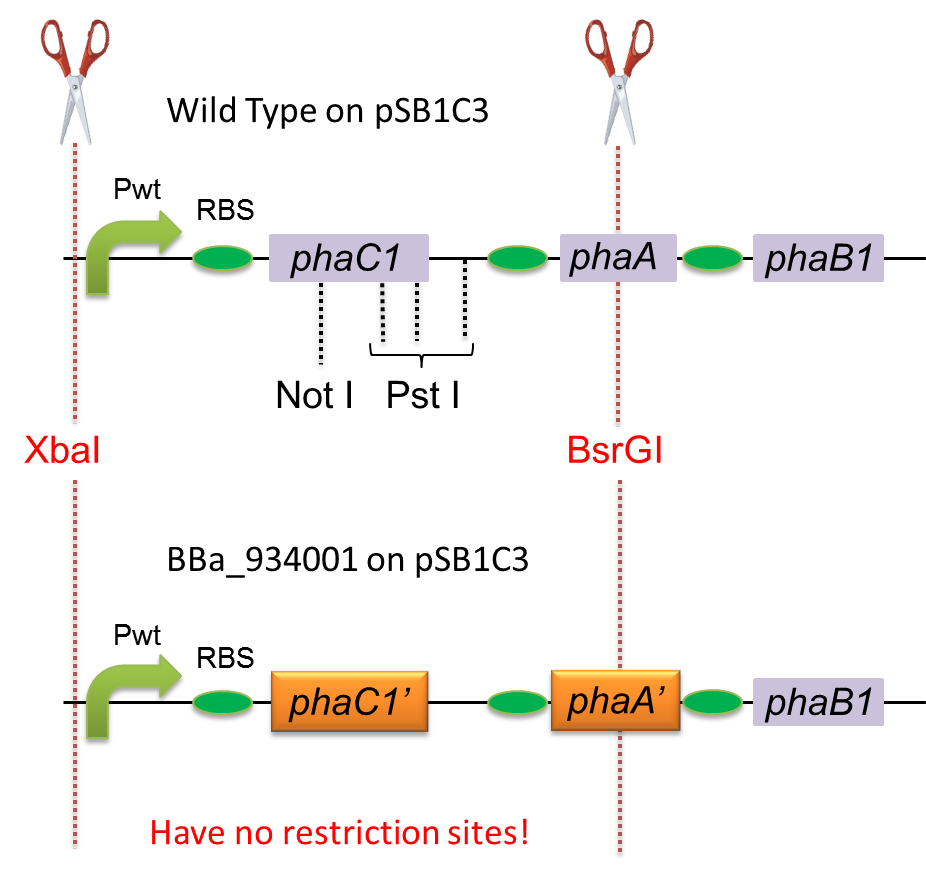
To construct a part that meets Biobrick format, we have modified the phaC1-A-B1 operon not to contain forbidden restriction enzyme sites. First, we cloned the wild type gene phaC1-A-B1 from R.eutropha H16 by using PCR and inserted the gene into pSB1C3. However, wild type phaC1-A-B1 gene sequence contains one NotI and three PstI recognition sites that are not allowed in Biobrick format. To get phaC1-A-B1 sequence without these recognition sites, we ordered the chemically synthesized DNA from IDT/MBL. In this chemically synthesized DNA, coding is optimized for E.coli. We used restriction enzyme XbaI (on pSB1C3) and BsrGI (on phaC1-A-B1) to insert sequence. That is to say, we got Poly[(R)-3-hydroxybutyrate] synthesizing gene in Biobrick format (BBa_K934001).
[Back to "Construction of phaC1-A-B1 in Biobrick format"] "
"