Team:TU-Delft/Modeling
From 2012.igem.org
(→Research Cycle) |
(→Research Cycle) |
||
| Line 41: | Line 41: | ||
[[File:Model-Experiment Cycle.png|350px|right|thumb|'''Figure 2''': Model-Experiment Cycle.]] | [[File:Model-Experiment Cycle.png|350px|right|thumb|'''Figure 2''': Model-Experiment Cycle.]] | ||
| - | At the beginning, model hypotheses of system structures are proposed for signal transduction and ligand binding based on biological expertise | + | At the beginning, model hypotheses of system structures are proposed for signal transduction and ligand binding based on biological expertise, literature surveys and database. However, due to the inadequate descriptions of dynamics and lack of information, analyses of hypothesized models are necessary to investigate the properties of models, such as parameter sensitivities and structural stability in pathway model. Then, experiments can be comprehended and designed by the knowledge of model. For example, based on the understanding of diffusion model, a practical device is designed, and the output of the device is provisional. |
Next, the measured data from "wet" experiment, in turn, modifies the model to fit the reality by tuning the parameters and eliminating the inconsistent assumptions. For instance, ambiguous assumptions of the stability of pathway model is resolved by analysing the measured output GFP. | Next, the measured data from "wet" experiment, in turn, modifies the model to fit the reality by tuning the parameters and eliminating the inconsistent assumptions. For instance, ambiguous assumptions of the stability of pathway model is resolved by analysing the measured output GFP. | ||
Revision as of 00:08, 27 September 2012


Contents |
Overview
We decided to use the modeling expertise of the team members to achieve three key objectives for our project.
- Develop scientific understanding of the yeast pheromone response pathway.
- Test the effects of the changes to the system.
- Aid decision making in the laboratory.
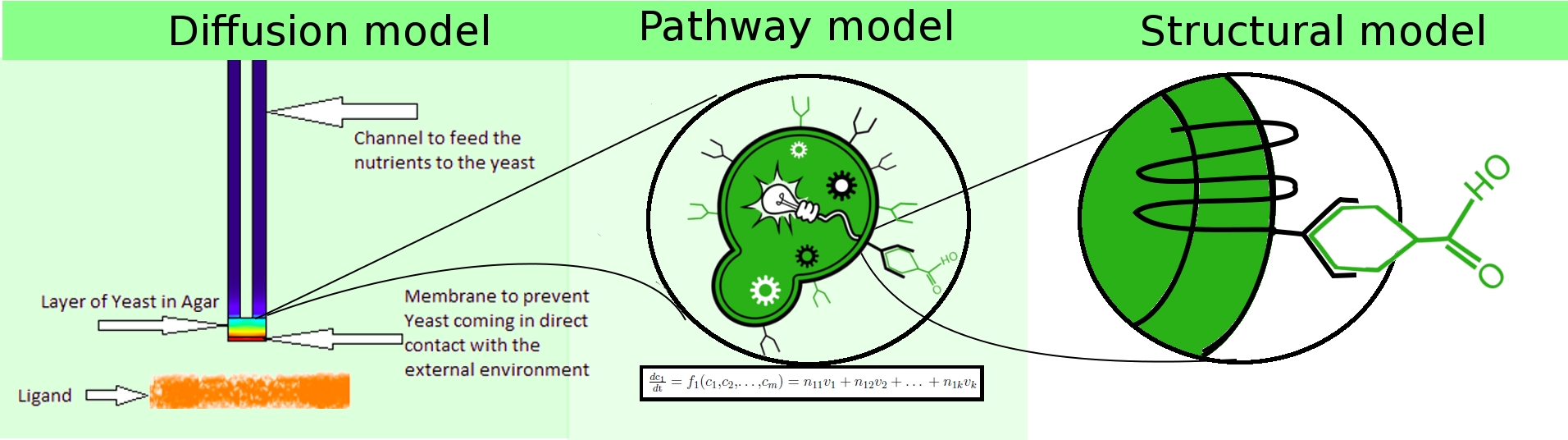
To achieve the stated objectives, we adopted a hierarchical modeling approach, in which we constructed heterogeneous models spanning
- population dynamics (diffusion model).
- extra-cellular interactions (structural model).
- intra-cellular dynamics (pathway model).
Connected Multi-level Models
These three models interact with each other to represent the mechanism of our project in different perspectives. At the system level, diffusion model sheds light on how the oder in the petridish diffuses through gas into the disk of olfactory yeasts, which imitates the diffusion process in real life such as smelling banana by human. As a downstream of receiving odor ligands, the pathway model is built to functionally simulate the intra-cellular biochemical reactions in the signalling pathway. Parameters in pathway were fitted to the experimental data thus it can give a good estimation on how the important intra-cellular species evolve and predict the producing fluorescence in the experiment. Moreover, the structural model is designed to understand the ligand docking at the molecular level. It indicates how well the hydrogen bonds are and how stable of activated receptor, which give an insight into rates of receptor inactivation in the pathway model. Further research of structural model on receptor binding and niche shrink in volume may provide more knowledge about binging rate and G protein release.
Based on these models, a practical device, Snifferometer is designed to achieve the purpose of odor detection. The model of this device is developed to give an idea of the application of our project.
The models span a broad spectrum of techniques: PDE, MD (molecular dynamics), SDE and ODE, as well as alignment techniques. Various and diverse tools have been employed: COMSOL, YASARA, MATLAB, COPASI, BLAST, ClustalW2, Ensembl.
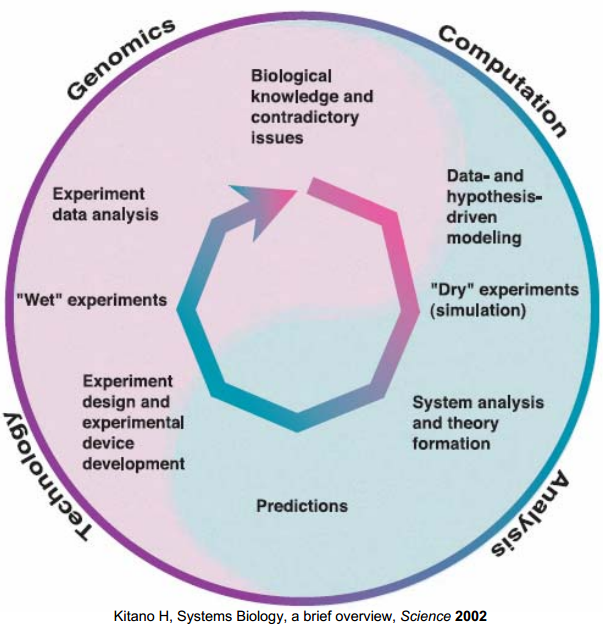
Research Cycle
This research cycle is introduced in our project as an interactive boundary work between wetlab and "dry" modeling to gain a hierarchical in-depth understanding of our biological system. [1]
At the beginning, model hypotheses of system structures are proposed for signal transduction and ligand binding based on biological expertise, literature surveys and database. However, due to the inadequate descriptions of dynamics and lack of information, analyses of hypothesized models are necessary to investigate the properties of models, such as parameter sensitivities and structural stability in pathway model. Then, experiments can be comprehended and designed by the knowledge of model. For example, based on the understanding of diffusion model, a practical device is designed, and the output of the device is provisional.
Next, the measured data from "wet" experiment, in turn, modifies the model to fit the reality by tuning the parameters and eliminating the inconsistent assumptions. For instance, ambiguous assumptions of the stability of pathway model is resolved by analysing the measured output GFP.
Later, the verified model gives a good comprehension for the biological system and useful predictions in different conditions can be simulated by "cheap" computations in silco instead of doing the time-consuming experiments.
Diffusion Model
One of the main objectives of the project is to synthesize a practical device, the Snifferometer for tuberculosis detection. As a first step towards achieving this goal, we built a temporal model of the system using PDE's which was simulated in matlab. A 2D reaction-diffusion system was then implemented in COMSOL multiphysics using the knowledge obtained from single cell pathway model, combining the behaviours of the which helped us get a better understanding of how such a device could be implemented and the response times involved in such a process.
Structural Model
Pathway Model
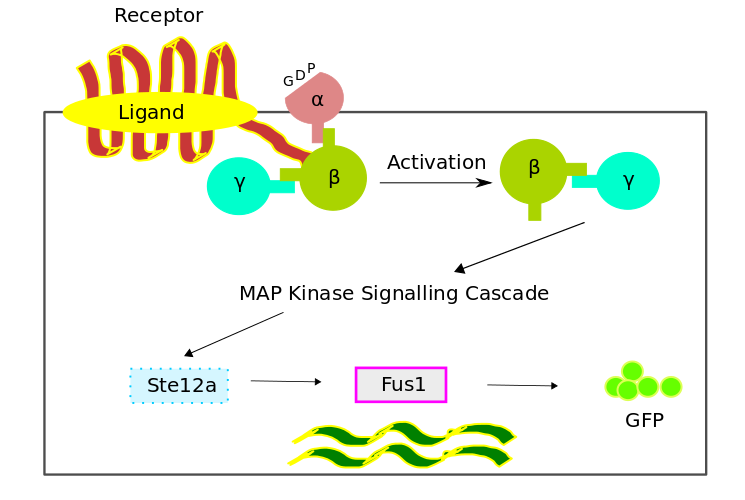
The bio-chemical pathway model was developed based on a scheme favouring the temporal order of the processes, which involved the four fundamental modules,
- Receptor Activation
- G - Protein Cycle
- MAPK Cascade
- Gene Expression
Three different models were used for the analysis of different aspects of the pathway. The dynamics of the system in these models were described using a set of differential equations governing the concentration changes of individual components and of complexes over time. On account of the gene expression module being noisy, a stochastic model of the gene expression module was implemented using stochastic differential equations.
Sensitivity and stability analysis were permformed to determine the sensitivity coefficients which were used to study the parametric dependence of the biological models. The results from the sensitivity analysis were then used in the parameter estimation to fit the model to the data provided by the experimentalists.
References
1.Kitano, H. Systems biology: a brief overview. Science 295, 1662–1664 (2002)
2.L.Gelis et al, Prediction of a Ligand-Binding Niche within a Human Olfactory Receptor by Combining Site-Directed Mutagenesis with Dynamic Homology Modeling, Wiley-VCH, Angew. Chem. Int. Ed., 51,1274–1278 (2012)

 "
"