Team:Slovenia/Notebook
From 2012.igem.org
Experimental methods


|
| Figure 1. Schematic presentation of methods used for cloning and culturing eukaryotic cells. |
|
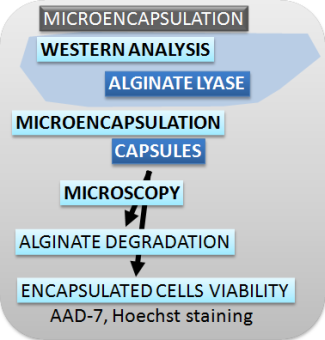
| Figure 2. Schematic presentation of methods used for characterizing the switch, safety mechanisms, microencapsulation and effectors. |
Cloning
Plasmid DNA isolation
A. MINI PREPs for analysis and sequencing- A single colony was picked from a LB-agar plate or glycerol stock and inoculated 10 mL LB-medium with appropriate antibiotic for selection (100 mg/L ampicillin, 50 mg/L kanamycin, 35 mg/L chloramphenicol).
- Bacteria were grown over night at 37 °C while shaking.
- Plasmid DNA was isolated from 2-6 mL of over-night culture with GeneJET plasmid miniprep kit according to manufacturer's protocol.
- Around 6-10 µg plasmid DNA was obtained.
- Purity and amount of DNA was analysed using a NanoDrop.
Fragment DNA isolation from agarose gel
A. AGAROSE ELECTROPHORESIS- A mixture of DNA fragments with different sizes were separated with agarose gel (0.7 and 2% agarose in 0.5x TAE buffer and 0.1 µg/ml ethidium bromide) at constant voltage of 100 V.
- UV light (λ = 254 nm) was used to visualize ethidium bromide intercalated into DNA.
- Position of DNA fragments are documented and cut out from agarose.
B. FRAGMENT ISOLATION from agarose gel
- The band with the desired DNA fragment was excised from the gel, using a clean scalpel.
- DNA was isolated from the gel slice with GeneJet Gel Extraction Kit according to manufacturers protocol.
- Purity and amount of DNA was determined using a NanoDrop.
Restriction digest
- Set up a restriction mixture:
- for analysis of cloned DNA
- 1X optimized Restriction buffer (10X)
- 0.5 µL restriction enzyme (10 U/µL)
- Bring volume to 20 µL with nuclease-free water.
- for isolation of specific DNA
- 1X optimized Restriction buffer 10X
- up to 2 µL restriction enzyme 10 U/µL
- Bring volume to 50 µL with nuclease-free water.
- The sample was incubated at optimal temperature for the restriction enzyme(s)
- Analysis of fragmented DNA was done by gel electrophoreses (loading dye was added to the samples before loaded on a agarose gel)
- Results were documented with
- Desired DNA fragment was excised and purified using suitable DNA purification kit.
PCR
A. PCR REACTIONAccuPrime and Phusion polymerase were used for DNA amplification. Colony PCR was preformed with Taq polymerase.
- For Phusion polymerase: master mix contained
- DNA (1-10 ng)
- both primers (0,4 pmol/µl )
- 1x Phusion HF buffer
- 0,2 µM dNTPs
- Phusion polymerase (0,02 U/ µl) and
- MQ up to final volume of 25 µl
-
vAccuPrime reaction contained:
- DNA (10 ng),
- both primers (0,4 pmol/µl ),
- 1xRnx mix,
- enzyme (0,05 U/ µl) and
- MQ up to final volume of 50 µl.
-
Taq polymerase mix contained:
- both primers (0,4 pmol/µl),
- 1x Taq PCR buffer II,
- 0,2 µM dNTPs,
- 5mM MgSO4,
- enzyme (0,125 U/ µl) and
- MQ up to total volume of 20 µl.
- Then the bacterial colony was added to the reaction mix.
- All temperature programs were designed according to manufacturer protocol, primers melting temperature and length of desired PCR products. Reactions were preformed in Applied Biosystems Veriti 96 well thermal cycler.
B. PCR product purification
Desired PCR product was purified by GeneJet Gel Extraction Kit according to manufacturer's protocol.
DNA concentration. An aliquot of DNA isolation was analyzed using a NanoDrop.
Gibson Assembly
Gibson assembly master mix was prepared as by Gibson at al., 2009.- 50 ng of each PCR product were added to Gibson assembly master mix and incubated at 50 °C.
- After incubation, the transformation in bacteria was preformed.
Ligation
T4 ligase joins the 5' phosphate and the 3'-hydroxyl groups of DNA.- Estimate the vector and insert concentrations and set molar ratio of insert to vector 3:1 (100-150ng Vector DNA).
- Set up a ligation mixture:
1X ligase buffer (10X)
1 µL T4 ligase (3 U/µL)
Bring volume to 10 or 20 µL with nuclease-free water. - Incubate sample for sticky-end ligation reactions at room temperature for 3 hours (or at 4 to 8 °C, overnight).
or
Incubate blunt-end ligation reactions at 17 °C for 4 to 18 hours. - After incubation part of the ligation mixture is used for transformation of bacterial cells (see: transformation of bacteria)
Culturing clones
A. BACTERIAFor plasmid DNA propagation two bacterial strains were used: DH5alpha [fhuA2Δ(argF-lacZ)U169 phoA glnV44 Φ80 Δ(lacZ)M15 gyrA96 recA1 relA1 endA1 thi-1 hsdR17] and TOP10 [mcrA, Δ(mrr-hsdRMS-mcrBC), Phi80lacZ(del)M15, ΔlacX74, deoR, recA1, araD139, Δ(ara-leu)7697, galU, galK, rpsL(SmR), endA1,nupG].
B. GROWTH MEDIA for BACTERIA
Luria Broth (LB)
- 10 g/L tryptone
- 5 g/L yeast extract
- 10 g/L NaCl
- media is supplemented with suitable antibiotics depending on selection marker on transfected plasmid: ampicilin 100 mg/L or kanamycin 50 mg/L
- LB with 1.5% agar
- media is supplemented with suitable antibiotics depending on selection marker on transfected plasmid
Transformation of bacteria
For enrichment of vectors E. coli DH5alpha and TOP10 were used.- 100 µL of competent cells were thawed on ice.
- 50 – 400 ng DNA solution was added to competent bacterial cells (depending on the concentration of the DNA solution).
- A mixture of cells and DNA solution was incubated on ice for 30-60 minutes.
- Than the mixture was heat-shocked for 45 seconds at 42 °C.
- Cooled down for 3 minute on ice.
- The mixture were than rescued by adding 500 µL preheated antibiotic free LB-medium and incubated for one hour at 37 °C while shaking for induction of the antibiotic resistance.
- The selection for plasmid containing and therefore antibiotic resistant bacteria was conducted by plating them on antibiotic containing LB-agar plates.
Glycerol stock for long term storage of bacteria
- 1 mL of an overnight culture was added to 150 µL of 80% glycerol into a cryo-tube.
- Mixed and incubated at room temperature for 30 min.
- Afterwards the glycerol stock was stored at -80 °C.
Cell cultures
Eucaryotic cell lines and cultivation
HEK293 is a human cell line derived from kidney cells and grows in a monolayer culture. Cells were grown in DMEM medium supplemented with 10% FBS.
HEK293T cell line is derived form HEK293 cells. HEK293T cells express the SV40 large T-antigen that enables episomal replication of plasmids containing the SV40 origin of replication in transfected cells. Cells were grown in DMEM medium supplemented with 10% FBS.
NK-92 is an interleukin-2 (IL-2) dependent natural killer cell line derived from peripheral blood mononuclear cells from patient with non-Hodgkin's lymphoma. The cell line is cytotoxic to a wide range of malignant cells. Cells were grown in RPMI medium supplemented with 20% FBS and 100 U/ml IL-2.
Cultivation
A. SUBCULTURING MONOLAYER CELL CULTURES- Remove and discard culture medium from a T-75 flask containing a monolayer of HEK293 or HEK293T cells.
- Rinse the T-75 flask with 10 ml of PBS buffer to remove all traces of growth medium (DMEM + 10% FBS) which otherwise inhibits trypsin function. Remove and discard the PBS buffer.
- Add 2-3 ml of trypsine solution and gently tilt the flask to ensure the trypsine solution covers all the cells. Incubate the cells in trypsin for 0,5 - 3 min.
- When the cells start to detach from the surface, add 7 ml of growth medium to the trypsin solution. Resuspend all remaining cells from the bottom of the T-75 flask by pipetting.
- Transfer the cell suspension to a 15 ml centrifuge tube.
- Centrifuge the cell suspension for 5 min at 1200 rpm.
- Remove the trypsin-containing medium from the centrifuge tube.
- Resuspend the cell pellet in fresh medium.
- Take as much cells as you need and add fresh medium to a total volume of 10 ml.
- Return the cells in a T-75 flask to the incubator (37 °C, 5 % CO2).
B. CELL PLATING
- Count cells.
- Calculate the desired number of cells per well. Dilute cells in DMEM with 10% FBS.
- Transfer the cells into an appropriate plate and place in a cell culture incubator.
MEDIA and BUFFERS
DMEM supplemented with
- 1 % L-Glutamine (GlutaMax)
- 10 % FBS
- Optionally: 1% Pen/Strep
RPMI supplemented with
- 1 % L-Glutamine (GlutaMax)
- 20 % FBS
Transfection
TABLE: Transfection mixtures for different culture format| Culture format | jetPEI reagent per µg of DNA (µL) | Typical amount of DNA (ng) | Volume of 150 mM NaCl solution for DNA and jetPEI (µL) | Total transfection mixture volume (µL) |
|---|---|---|---|---|
| 96-well | 2 | 200 | 10 | 20 |
| 24-well and 8-well microscope chamber | 2 | 500 | 50 | 100 |
| 12-well | 2 | 1000 | 50 | 100 |
| 6-well | 2 | 2000 | 100 | 200 |
| 10 cm | 2 | 15000 | 250 | 500 |
- Dilute plasmid DNA to desired concentration in 150 mM NaCl, vortex gently and spin down briefly.
- Dilute jetPEI (PolyPlus) in 150mM NaCl, vortex gently and spin down briefly.
- Add the jetPEI solution to the DNA solution.
- Vortex the solution immediately and spin down briefly.
- Incubate for 15 to 30 minutes at room temperature.
- Add the jetPEI/DNA mix to the cells in and gently swirl the plate.
- Return the plate to a cell culture incubator.
Next: Lablog >>
 "
"