Team:SJTU-BioX-Shanghai/Project/project3.2
From 2012.igem.org
Huanan1991 (Talk | contribs) (Created page with "{{Template:12SJTU_header}} {{Template:12SJTU_nav_project}} <table><tr> <td valign="top" width="200"> {{Template:12SJTU_floatnav_head}} <html> <script type="text/javascript"> // ...") |
Huanan1991 (Talk | contribs) |
||
| Line 35: | Line 35: | ||
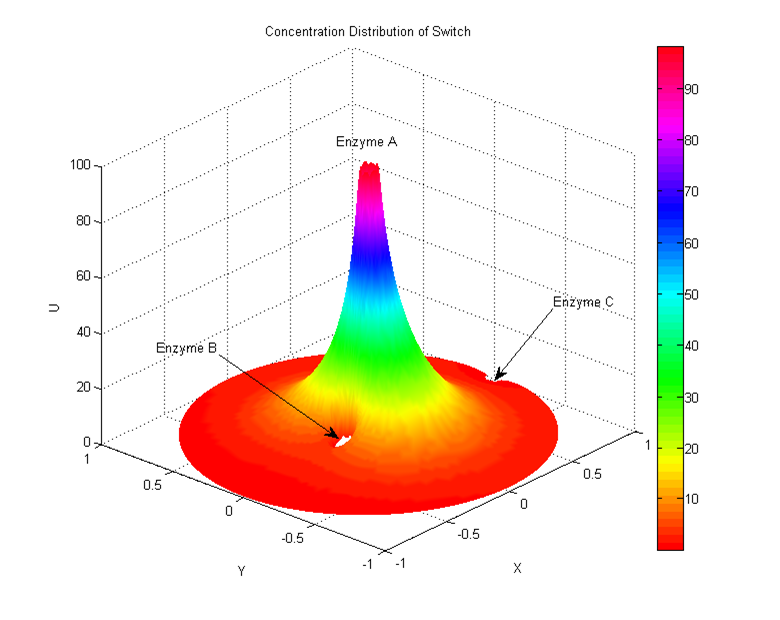
Enzymes in the enzyme cluster AB are adjacent to each other, and have a tendency to produce more products that enzyme C. Basic simulation shows that the concentration of substrate α distributes like this: | Enzymes in the enzyme cluster AB are adjacent to each other, and have a tendency to produce more products that enzyme C. Basic simulation shows that the concentration of substrate α distributes like this: | ||
| - | [[Image:12SJTU_Modelling7.png|thumb|600px|center|''Fig.7'']] | + | [[Image:12SJTU_Modelling7.png|thumb|600px|center|''Fig.7'' Distribution of substrate α concentration]] |
To illustrate the diffusion behavior and the reaction progress, we assume that enzyme A locates on the center and produce α as the rate of (v_max [A])/(K_m+[A]), and enzyme B and enzyme C occupies the region of Ф1 and Ф2. In the beginning, enzyme B and C share different substrate concentration due to their distance to enzyme A. And enzyme B may produces much more products. Then we give the system a switch signal which can shorten the distance between enzyme A and C. The products of enzyme C rise because it gets more substrate than before. The percentage of these two products | To illustrate the diffusion behavior and the reaction progress, we assume that enzyme A locates on the center and produce α as the rate of (v_max [A])/(K_m+[A]), and enzyme B and enzyme C occupies the region of Ф1 and Ф2. In the beginning, enzyme B and C share different substrate concentration due to their distance to enzyme A. And enzyme B may produces much more products. Then we give the system a switch signal which can shorten the distance between enzyme A and C. The products of enzyme C rise because it gets more substrate than before. The percentage of these two products | ||
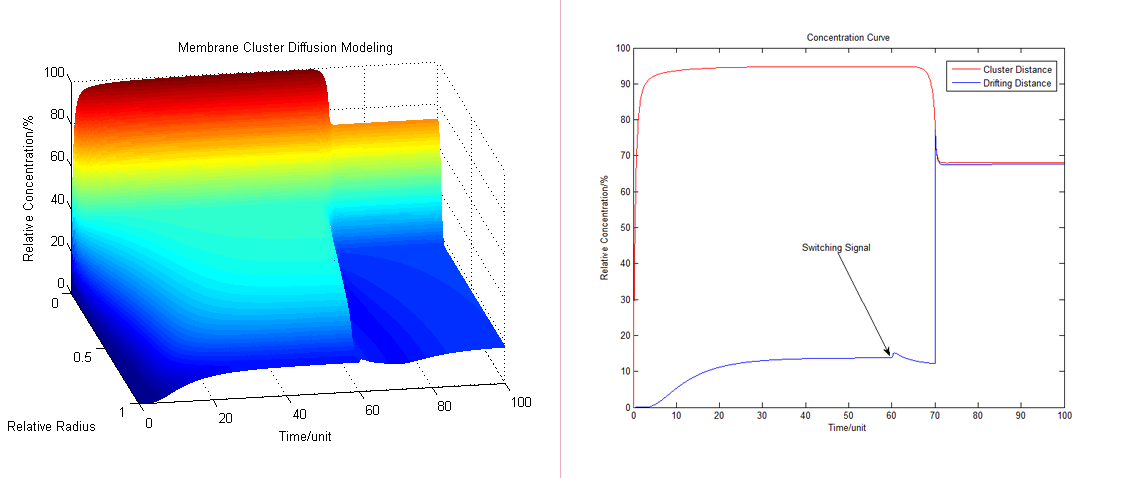
The whole process in membrane switch can be simulated as following: | The whole process in membrane switch can be simulated as following: | ||
| - | [[Image:12SJTU_Switching12.png|thumb| | + | [[Image:12SJTU_Switching12.png|thumb|700px|center|''Fig.8'' Dynamic modeling of substrate α concentration before and after signaling ]] |
| - | [[Image:12SJTU_Switching34.png|thumb| | + | At the beginning of all, the enzyme B and C locates on the membrane, A & B are linked together to make a cluster. With the time goes by the concentration of α quickly reaches a peak, and diffuses towards the outside. After producing for some time, the ratio of different products reaches a stable state. At time=60, the system receives a signal. Then enzyme approaches the cluster at a very fast speed and consumes more substrates. (Fig. 8) |
| + | |||
| + | [[Image:12SJTU_Switching34.png|thumb|700px|center|''Fig.9'' Integral and Ratio of product amount]] | ||
| + | |||
| + | We call the product of Enzyme B as the major product. To show the equilibrium state after a long period of inducement, we assume that the signal comes at time=100. We can easily see that the major products product amount changes from about 70% of all products to 50%. In other words, the minor product amount has increased by 67%. (Fig. 9) | ||
==Discussion== | ==Discussion== | ||
| + | In our model we assume that the reaction & substrate consumption abilities of B and C are the same. However in reality, the Km of different enzymes are not the same. Considering Michaelis-Menten equation, if the substrate concentration is 10 or more times the Km, we can ignore the effects of different abilities of enzymes, just as the normal biochemical conditions. | ||
| + | |||
| + | Amazingly our experiment results exactly agree with the modeling ratio, which changes from around 70% to 50%, ignoring deviations. These results strongly indicates the mechanism of steering process. | ||
{{Template:12SJTU_footer}} | {{Template:12SJTU_footer}} | ||
Revision as of 10:30, 26 September 2012
| ||
|
 "
"