Team:Penn/Notebook/Biofilms
From 2012.igem.org
(Difference between revisions)
(Created page with "==6/6/12== *Set up equipment *Autoclaved 2x 1L Sterile ddH<sub>2</sub>O *Autoclaved 500 mL LB Broth for scale up of luxS (Plate 3 Well 2H, pSB1A2), plsr (Plate 2 Well 14H in pSB1...") |
|||
| Line 20: | Line 20: | ||
*[plsr]=112 ng/uL | *[plsr]=112 ng/uL | ||
*[luxS]=114 ng/uL | *[luxS]=114 ng/uL | ||
| + | ==6/11/12-6/16/12== | ||
| + | *Dry lab work | ||
| + | ==6/18/12== | ||
| + | *Obtained & resuspended primers for eGFP & plsr | ||
| + | ==6/19/12== | ||
| + | *performed PCR w/ NEB standard taq kit w/ primers for eGFP & plsr | ||
| + | *Tested 2 annealing temperatures, 55C and 50C | ||
| + | *Ran gel w/ PCR product (2% agarose), saw clear eGFP bands, but possible .1kb obscured by loading dye. | ||
| + | ==6/20/12== | ||
| + | *Performed qiagen PCR purification & nanodropped (it like it's hot) | ||
| + | ==6/21/12== | ||
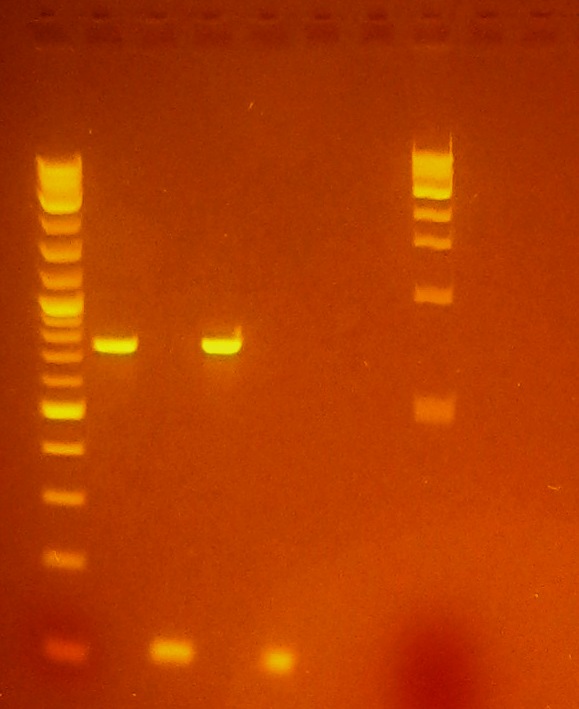
| + | *Ran gel of yesterday's PCR purification | ||
| + | [[File:PCR Purified eGFP & plsr.JPG]] | ||
| + | *Obtained & transformed pET-26b | ||
Revision as of 17:51, 21 June 2012
Contents |
6/6/12
- Set up equipment
- Autoclaved 2x 1L Sterile ddH2O
- Autoclaved 500 mL LB Broth for scale up of luxS (Plate 3 Well 2H, pSB1A2), plsr (Plate 2 Well 14H in pSB1A2 resistance to Amp)
- Added iGEM logo to wiki
6/7/12
- Resuspended available biobricks luxS & plsr (resuspended in 10uL ddH2O)
- Resuspended DNA transformed into DH5α (40uL DH5α+2uL DNA) & plated on LB plates w/ 15 uL of 100 mg/mL Amp spread on surface
- Requested lsrR & lsrK from iGEM HQ
6/8/12
- iGEM Wiki now split into two separate Notebooks
- Plenty of colonies, too many to count(TMTC) grew from plsr & luxS transformation from 6/7/12
- Selected one colony each
- Grew in 10 mL LB+Amp (100 ug/mL)
- Contacted researchers for V. harveyi bioassay & lysostaphin, V. harveyi must be purchased through ATCC ($300), lysostaphin obtained thru MTA.
6/9/12
- ~2mL of LB evaporated during incubation
- Cells spun down @ 5000 rpm for 10 min
- Miniprepped 4x Lysis into 1 column
- [plsr]=112 ng/uL
- [luxS]=114 ng/uL
6/11/12-6/16/12
- Dry lab work
6/18/12
- Obtained & resuspended primers for eGFP & plsr
6/19/12
- performed PCR w/ NEB standard taq kit w/ primers for eGFP & plsr
- Tested 2 annealing temperatures, 55C and 50C
- Ran gel w/ PCR product (2% agarose), saw clear eGFP bands, but possible .1kb obscured by loading dye.
6/20/12
- Performed qiagen PCR purification & nanodropped (it like it's hot)
6/21/12
- Ran gel of yesterday's PCR purification
- Obtained & transformed pET-26b
 "
"