Team:Cambridge/Lab book/Week 6
From 2012.igem.org
(→Monday) |
|||
| (37 intermediate revisions not shown) | |||
| Line 8: | Line 8: | ||
!align="center"|[[Team:Cambridge/Lab_book/Week_6|6]] | !align="center"|[[Team:Cambridge/Lab_book/Week_6|6]] | ||
!align="center"|[[Team:Cambridge/Lab_book/Week_7|7]] | !align="center"|[[Team:Cambridge/Lab_book/Week_7|7]] | ||
| + | !align="center"|[[Team:Cambridge/Lab_book/Week_8|8]] | ||
| + | !align="center"|[[Team:Cambridge/Lab_book/Week_9|9]] | ||
| + | !align="center"|[[Team:Cambridge/Lab_book/Week_10|10]] | ||
| + | !align="center"|[[Team:Cambridge/Lab_book/Week_11|11]] | ||
| + | !align="center"|[[Team:Cambridge/Lab_book/Week_12|12]] | ||
| + | !align="center"|[[Team:Cambridge/Lab_book/Week_13|13]] | ||
| + | !align="center"|[[Team:Cambridge/Lab_book/Week_14|14]] | ||
|} | |} | ||
| - | ===Monday=== | + | ===Monday (30/07/12)=== |
| - | '''[[Team:Cambridge/Protocols/PCRProtocol|PCR of vectors and mOrange]]''' | + | '''Ratiometrica & Ribosense: [[Team:Cambridge/Protocols/PCRProtocol|PCR of vectors and mOrange]]''' |
---- | ---- | ||
| + | |||
| + | [[File:mOrangegel.jpg|250px|thumb|mOrange PCR run at multiple temperatures, every two lanes increacing the temperature of the annealing step by 2 °C. No lanes worked.]] | ||
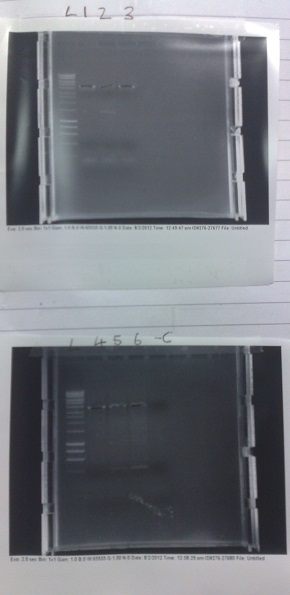
[[File:vectorgel1.jpg|250px|thumb|Top: Fusion, lux containing vector gel. Lanes 2-4, Vector DNA. Lane 5, +ve control. Lane 6, -ve control. Bottom: Vector gel. Lanes 1-3, Fluorescent contruct vector DNA. Lanes 4-6, Riboswitch construct vector DNA.]] | [[File:vectorgel1.jpg|250px|thumb|Top: Fusion, lux containing vector gel. Lanes 2-4, Vector DNA. Lane 5, +ve control. Lane 6, -ve control. Bottom: Vector gel. Lanes 1-3, Fluorescent contruct vector DNA. Lanes 4-6, Riboswitch construct vector DNA.]] | ||
| Line 28: | Line 37: | ||
*Remaining stray product had a slightly tricky secondary structure at the 3' end. It will be run at a series of annealing temperatures in a PCR machine capable of a temperature gradient. | *Remaining stray product had a slightly tricky secondary structure at the 3' end. It will be run at a series of annealing temperatures in a PCR machine capable of a temperature gradient. | ||
| - | *mOrange PCR run at many different temperatures, from 62 °C to 76 °C. | + | *mOrange PCR run at many different temperatures, from 62 °C to 76 °C. However this doesn't seem to solve our problem (refer to Gel photos). It seems likely that it is a primer design problem. |
| - | '''[[Team:Cambridge/Protocols/GelElectrophoresis|Separation of vector and mOrange DNA]]''' | + | '''Ratiometrica: [[Team:Cambridge/Protocols/GelElectrophoresis|Separation of vector and mOrange DNA]]''' |
---- | ---- | ||
| Line 38: | Line 47: | ||
*Realized correct lux vector template was not added during PCR preparation, consequently no amplification occured. Still appears to be a primer smear. | *Realized correct lux vector template was not added during PCR preparation, consequently no amplification occured. Still appears to be a primer smear. | ||
| - | *Fluorescent | + | *Fluorescent vector backbone produced several bands. Appears to be due to mis-priming during PCR. Either changing the primers or raising the annealing temperature should solve this problem, but may mean that we have to do this PCR separately. |
*Riboswitch vector also failed. However, the extraction from the previous PCR run may have worked. We will try producing a functional plasmid with this extraction before running this PCR again. | *Riboswitch vector also failed. However, the extraction from the previous PCR run may have worked. We will try producing a functional plasmid with this extraction before running this PCR again. | ||
| - | '''[[Team:Cambridge/Protocols/Gibsonassembly|Construction of riboswitch plasmid with Gibson Assembly]]''' | + | '''Ribosense: [[Team:Cambridge/Protocols/Gibsonassembly|Construction of riboswitch plasmid with Gibson Assembly]]''' |
---- | ---- | ||
| Line 50: | Line 59: | ||
*DNA from lanes 22,23 and 24 fused with riboswitch DNA produced two weeks ago to produce riboswitch construct. This has replacement of the first 8 codons of lac I with the 8 codons native to the gene downstream of the riboswitch. | *DNA from lanes 22,23 and 24 fused with riboswitch DNA produced two weeks ago to produce riboswitch construct. This has replacement of the first 8 codons of lac I with the 8 codons native to the gene downstream of the riboswitch. | ||
| - | '''[[Team:Cambridge/Protocols/TransformationofB.subtilis|Transformation of ''Bacillus'' with riboswitch construct]]''' | + | '''Ribosense: [[Team:Cambridge/Protocols/TransformationofB.subtilis|Transformation of ''Bacillus'' with riboswitch construct]]''' |
---- | ---- | ||
| Line 56: | Line 65: | ||
*Plasmids made by Gibson transformed into ''bacillus'' cells made two weeks ago and transformants plated out on 5μg/ml chloramphenicol plates. | *Plasmids made by Gibson transformed into ''bacillus'' cells made two weeks ago and transformants plated out on 5μg/ml chloramphenicol plates. | ||
| - | ===Tuesday=== | + | ===Tuesday (31/07/12)=== |
| - | '''New biobricks''' | + | '''Existing biobricks: New biobricks''' |
| + | |||
| + | ---- | ||
*E.coli containing biobricks ordered were plated out on kanomycin plates (50μg/ml). | *E.coli containing biobricks ordered were plated out on kanomycin plates (50μg/ml). | ||
| - | '''[[Team:Cambridge/Protocols/TransformationofB.subtilis|Transformation of ''Bacillus'' with riboswitch construct]]''' | + | '''Ribosense: [[Team:Cambridge/Protocols/TransformationofB.subtilis|Transformation of ''Bacillus'' with riboswitch construct]]''' |
---- | ---- | ||
| Line 68: | Line 79: | ||
*Plasmids made by Gibson transformed into ''bacillus'' cells made two weeks ago and transformants plated out on 5μg/ml chloramphenicol plates. | *Plasmids made by Gibson transformed into ''bacillus'' cells made two weeks ago and transformants plated out on 5μg/ml chloramphenicol plates. | ||
| - | '''[[Team:Cambridge/Protocols/TransformationofE.coli|Transformation of E.coli with riboswitch construct]]''' | + | '''Ribosense: [[Team:Cambridge/Protocols/TransformationofE.coli|Transformation of TOP10 E.coli with riboswitch construct]]''' |
---- | ---- | ||
| Line 74: | Line 85: | ||
*Plasmids made by Gibson transformed into TOP10 e.coli cells and transformants plated out on 100μg/ml ampicillin plates. | *Plasmids made by Gibson transformed into TOP10 e.coli cells and transformants plated out on 100μg/ml ampicillin plates. | ||
| - | + | '''Ratiometrica & Ribosense: [[Team:Cambridge/Protocols/PCRProtocol|PCR of vector backbone]]''' | |
| - | '''Mg2+ Riboswitch''' | + | ---- |
| + | |||
| + | *3rd attempt at vector backbone PCR; owing to the mispriming yesterday we decided to try touch down PCR. | ||
| + | |||
| + | *PCR Programme: | ||
| + | |||
| + | *Results: very hazy bands for the Ratiometrica-Lux vector at approximately the right size (Gel #1 lanes 2-4); working positive control (Gel #1 lane 6); Bands of the wrong size for the Ratiometrica-Flu vector (Gel #2 lanes 2-3); no bands for the Mg RS vector (Gel #2 lanes 5-7) | ||
| + | |||
| + | ===Wednesday (01/08/12)=== | ||
| + | |||
| + | '''Ribosense: Mg2+ Riboswitch''' | ||
---- | ---- | ||
| Line 84: | Line 105: | ||
*Colonies also grown up in 10ml of medium A for use with plate reader later. | *Colonies also grown up in 10ml of medium A for use with plate reader later. | ||
| - | '''[[Team:Cambridge/Protocols/PCRProtocol|PCR of | + | ===Thursday (02/08/12)=== |
| + | |||
| + | '''Ratiometrica-Flu: [[Team:Cambridge/Protocols/PCRProtocol|PCR of split fluorescent construct]]''' | ||
---- | ---- | ||
| - | + | [[File:Flu split vector.jpg|250px|right|thumb|Fluorescent construct gels. Top: Lanes 2-4: Fragment A - expected size 4500bp. Bottom: Lanes 2-4: Fragment B - expected size 3000bp. Lane 5: negative control. All lanes worked well.]] | |
| - | * | + | *Our present theory is that the vectors that we are trying to amplify is too large for the PCR to effectively take place. Given this, we are using split primers to try to reduce the size of the fragments (kindly provided by [[Team:Cambridge/Special_thanks|P.J.Steiner]]). |
| - | :* | + | *New primer sequences: |
| + | |||
| + | :*Forward: tgaagtgttcgacaatataaatgtg|CGAAACGATCCTCATCCTGT | ||
| + | |||
| + | :*Reverse: ACAGGATGAGGATCGTTTCG|cacatttatattgtcgaacacttca | ||
| + | |||
| + | *PCR Programme | ||
| + | |||
| + | *After gel electrophorsis, it was found that the PCR had worked perfectly. Given we now have all the fragments for the fluorescent construct, we will try to make it with Gibson ASAP. | ||
| + | |||
| + | ===Friday (03/08/12)=== | ||
| + | |||
| + | '''Ratiometrica-Flu: [[Team:Cambridge/Protocols/Gibsonassembly|Construction of fluorescent plasmid with Gibson Assembly]]''' | ||
| + | |||
| + | ---- | ||
| + | |||
| + | *'''Isothermal reaction buffer''' *5 remade with correct composition. Needed to order NAD+, so used some master mix from Haseloff lab. | ||
| + | |||
| + | *Various Gibson tail tagged DNA fragments produced over last few weeks assembled together using Gibson protocol: | ||
| + | |||
| + | :*pJS130 plasmid fragment A | ||
| + | |||
| + | :*pJS130 plasmid fragment B | ||
| + | |||
| + | :*CFP (E0020) | ||
| + | |||
| + | :*Terminator (B0015) | ||
| + | |||
| + | :*pVEG + RBS (K143053) | ||
| + | |||
| + | :*YFP (E0030) | ||
| + | |||
| + | '''Ratiometrica-Flu: [[Team:Cambridge/Protocols/TransformationofE.coli|Transformation of TOP10 e.coli with Gibson products]]''' | ||
| + | |||
| + | ---- | ||
| + | |||
| + | *3 tubes of TOP10 transformed with Gibson products. Cells plated out on 100μg/ml ampicillin plates. | ||
| + | |||
| + | *No positive control added. If no cells grow, will add positive control to all future experiments. | ||
| + | |||
| + | *Results: no growth (04/08/12) | ||
| + | |||
| + | '''Ribosense: [[Team:Cambridge/Protocols/PCRProtocol|PCR of split Mg2+ vector]]''' | ||
| + | |||
| + | ---- | ||
| + | |||
| + | [[File:split Mg2+ 1.jpg|right|250px|thumb|results of split magnesium vector PCR. None of the products worked]] | ||
| + | |||
| + | *PCR of magnesium riboswitch vector repeated with primers to split plasmid. | ||
| + | |||
| + | *Lanes 2 + 3: Fragment A (center - cut site (promotor side)) | ||
| + | |||
| + | *Lanes 4 + 5: Fragment B (without 8 codon substitution) (cut site (lac I side) - center) | ||
| + | |||
| + | *Lanes 6 + 7: Fragment B (with 8 codon substitution) (cut stie (lac I side) - center) | ||
| + | |||
| + | *Gels run, found PCR was unsuccessful. | ||
| + | |||
| + | '''Ratiometrica-Lux: [[Team:Cambridge/Protocols/PCRProtocol|PCR of mOrange]]''' | ||
| + | |||
| + | ---- | ||
| + | |||
| + | *with the forward primer correctly labelled and the single-base deletion in the reversed primer fixed, we were able to obtain the mOrange fragment | ||
| + | |||
| + | *PCR Programme: | ||
| + | |||
| + | *Result: bands of the correct size (Lanes 7-8) | ||
| + | |||
| + | '''Existing biobricks: [[Team:Cambridge/Protocols/PCRProtocol|PCR of RFP and Mg promoter]]''' | ||
| + | |||
| + | ---- | ||
| + | |||
| + | *First attempt at PCR of the RFP and Mg promoter from the registry. | ||
| + | |||
| + | *PCR Programme: | ||
| + | |||
| + | *Result: No bands for either (RFP lanes 3-4; Mg promoter lanes 5-6); very likely a problem with the mastermix. | ||
| + | |||
| + | ===Sunday (05/08/12)=== | ||
| + | |||
| + | '''Ribosense: [[Team:Cambridge/Protocols/PCRProtocol|PCR of Mg Riboswitch vector fragments]]''' | ||
| + | |||
| + | ---- | ||
| + | |||
| + | *2nd attempt since the PCR on Friday did not work, possibly due to mastermix problems | ||
| + | |||
| + | *PCR Programme: | ||
| + | |||
| + | *Results: Fragment A was successfully PCR-ed (lanes 3-4); Fragment B did not come out, with or without the 8 codons (lanes 5-6 (-8); lanes 7-8 (+8)) | ||
| + | |||
| + | '''Existing biobricks: [[Team:Cambridge/Protocols/PCRProtocol|PCR of RFP and Mg promoter]] | ||
| + | |||
| + | ---- | ||
| - | + | *2nd attempt since the PCR on Friday did not work, possibly due to mastermix problems | |
| - | + | *PCR Programme: | |
| - | + | *Results: RFP was successfully PCR-ed (lanes 3-4); there are no bands for Mg promoter (lanes 5-6), likely to be due to sequence inconsistency of the registry DNA | |
{{Template:Team:Cambridge/CAM_2012_TEMPLATE_FOOT}} | {{Template:Team:Cambridge/CAM_2012_TEMPLATE_FOOT}} | ||
Latest revision as of 15:22, 14 September 2012
| Week: | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 |
|---|
Contents |
Monday (30/07/12)
Ratiometrica & Ribosense: PCR of vectors and mOrange
- Not all products were obtained during Friday's PCR. Most of these missing products were large vector backbones. They are being run again, with a much longer extension time of 300s. If that fails, primers will be ordered to split the vectors into manageable chunks, and the PCR reattempted when they arrive.
- PCR cycle x35:
- 15s Denaturing at 95 C
- 45s Annealing at 60 C
- 300s Extension at 72 C
- Remaining stray product had a slightly tricky secondary structure at the 3' end. It will be run at a series of annealing temperatures in a PCR machine capable of a temperature gradient.
- mOrange PCR run at many different temperatures, from 62 °C to 76 °C. However this doesn't seem to solve our problem (refer to Gel photos). It seems likely that it is a primer design problem.
Ratiometrica: Separation of vector and mOrange DNA
- Positive control produced no band. No primer smear - primers may not be in mix for some reason.
- Realized correct lux vector template was not added during PCR preparation, consequently no amplification occured. Still appears to be a primer smear.
- Fluorescent vector backbone produced several bands. Appears to be due to mis-priming during PCR. Either changing the primers or raising the annealing temperature should solve this problem, but may mean that we have to do this PCR separately.
- Riboswitch vector also failed. However, the extraction from the previous PCR run may have worked. We will try producing a functional plasmid with this extraction before running this PCR again.
Ribosense: Construction of riboswitch plasmid with Gibson Assembly
- DNA from lanes 27+28, 27+29, 27+30 from gels run on Friday fused together with Gibson assembly to produce riboswitch construct. This does not have replacement of the first 8 codons of lac I with the 8 codons native to the gene downstream of the riboswitch.
- DNA from lanes 22,23 and 24 fused with riboswitch DNA produced two weeks ago to produce riboswitch construct. This has replacement of the first 8 codons of lac I with the 8 codons native to the gene downstream of the riboswitch.
Ribosense: Transformation of Bacillus with riboswitch construct
- Plasmids made by Gibson transformed into bacillus cells made two weeks ago and transformants plated out on 5μg/ml chloramphenicol plates.
Tuesday (31/07/12)
Existing biobricks: New biobricks
- E.coli containing biobricks ordered were plated out on kanomycin plates (50μg/ml).
Ribosense: Transformation of Bacillus with riboswitch construct
- Plasmids made by Gibson transformed into bacillus cells made two weeks ago and transformants plated out on 5μg/ml chloramphenicol plates.
Ribosense: Transformation of TOP10 E.coli with riboswitch construct
- Plasmids made by Gibson transformed into TOP10 e.coli cells and transformants plated out on 100μg/ml ampicillin plates.
Ratiometrica & Ribosense: PCR of vector backbone
- 3rd attempt at vector backbone PCR; owing to the mispriming yesterday we decided to try touch down PCR.
- PCR Programme:
- Results: very hazy bands for the Ratiometrica-Lux vector at approximately the right size (Gel #1 lanes 2-4); working positive control (Gel #1 lane 6); Bands of the wrong size for the Ratiometrica-Flu vector (Gel #2 lanes 2-3); no bands for the Mg RS vector (Gel #2 lanes 5-7)
Wednesday (01/08/12)
Ribosense: Mg2+ Riboswitch
- Successful colonies produced from transformations two days ago streaked out onto chloramphenicol (5 μg/ml) containing plates.
- Colonies also grown up in 10ml of medium A for use with plate reader later.
Thursday (02/08/12)
Ratiometrica-Flu: PCR of split fluorescent construct
- Our present theory is that the vectors that we are trying to amplify is too large for the PCR to effectively take place. Given this, we are using split primers to try to reduce the size of the fragments (kindly provided by P.J.Steiner).
- New primer sequences:
- Forward: tgaagtgttcgacaatataaatgtg|CGAAACGATCCTCATCCTGT
- Reverse: ACAGGATGAGGATCGTTTCG|cacatttatattgtcgaacacttca
- PCR Programme
- After gel electrophorsis, it was found that the PCR had worked perfectly. Given we now have all the fragments for the fluorescent construct, we will try to make it with Gibson ASAP.
Friday (03/08/12)
Ratiometrica-Flu: Construction of fluorescent plasmid with Gibson Assembly
- Isothermal reaction buffer *5 remade with correct composition. Needed to order NAD+, so used some master mix from Haseloff lab.
- Various Gibson tail tagged DNA fragments produced over last few weeks assembled together using Gibson protocol:
- pJS130 plasmid fragment A
- pJS130 plasmid fragment B
- CFP (E0020)
- Terminator (B0015)
- pVEG + RBS (K143053)
- YFP (E0030)
Ratiometrica-Flu: Transformation of TOP10 e.coli with Gibson products
- 3 tubes of TOP10 transformed with Gibson products. Cells plated out on 100μg/ml ampicillin plates.
- No positive control added. If no cells grow, will add positive control to all future experiments.
- Results: no growth (04/08/12)
Ribosense: PCR of split Mg2+ vector
- PCR of magnesium riboswitch vector repeated with primers to split plasmid.
- Lanes 2 + 3: Fragment A (center - cut site (promotor side))
- Lanes 4 + 5: Fragment B (without 8 codon substitution) (cut site (lac I side) - center)
- Lanes 6 + 7: Fragment B (with 8 codon substitution) (cut stie (lac I side) - center)
- Gels run, found PCR was unsuccessful.
Ratiometrica-Lux: PCR of mOrange
- with the forward primer correctly labelled and the single-base deletion in the reversed primer fixed, we were able to obtain the mOrange fragment
- PCR Programme:
- Result: bands of the correct size (Lanes 7-8)
Existing biobricks: PCR of RFP and Mg promoter
- First attempt at PCR of the RFP and Mg promoter from the registry.
- PCR Programme:
- Result: No bands for either (RFP lanes 3-4; Mg promoter lanes 5-6); very likely a problem with the mastermix.
Sunday (05/08/12)
Ribosense: PCR of Mg Riboswitch vector fragments
- 2nd attempt since the PCR on Friday did not work, possibly due to mastermix problems
- PCR Programme:
- Results: Fragment A was successfully PCR-ed (lanes 3-4); Fragment B did not come out, with or without the 8 codons (lanes 5-6 (-8); lanes 7-8 (+8))
Existing biobricks: PCR of RFP and Mg promoter
- 2nd attempt since the PCR on Friday did not work, possibly due to mastermix problems
- PCR Programme:
- Results: RFP was successfully PCR-ed (lanes 3-4); there are no bands for Mg promoter (lanes 5-6), likely to be due to sequence inconsistency of the registry DNA
 "
"