Biosynthesis - Fatty Acid
Fatty acids are promising biofuel precursors with a broad and bright future. An enzyme system of nine proteins is responsible for fatty acid biosynthesis in E.coli and is first introduced to iGEM by our team. We choose four crucial enzymes, TesA, FabG, FabZ, and FabI, to enhance the perform of the biosynthesis system.
To identify and to evaluate two outstanding features of the Membrane Magic project: the priority of products to exportation and the refinement of interaction.
To testify the feasibility of the Membrane Magic project in the context of practice.
To justify the universality of the Membrane Magic project
We constructed four enzyme-bear fusion proteins that locate on membrane and assemble orderly.
We gained 50% more fatty acids by solo introduction of membrane anchored TesA.
We successfully increased the yield of fatty acids by 24 fold with the recruitment of Membrane Accelerator.
Background
Fatty acid derivatives are to emerge as prominent fuels in the marketplace. Their high energy density and low water solubility make them promising alternatives to fossil fuel as transportation fuels. Biosynthesis, among all the biofuels production methods, is the one most sustainable, environmental friendly and less controversial.
Fatty acid biosynthesis in E.coli is catalyzed by a series of nine enzymes and the release of free fatty acids is catalyzed by a thioesterase via hydrolysis of acyl-ACP species. We are the first group in iGEM competition to recruit this enzyme system to facilitate the biosynthesis of fatty acid, especially C18 fatty acid.
We put Membrane Accelerator system into practice context to justify its versatility and its efficiency.
Biosynthetic Pathway
Fatty acid biosynthesis in E.coli is carried out by a nine-component enzyme system , FabA, FabB, FabD, FabF, FabG, FabH, FabI, FabZ, and ACP. They orderly cooperate to convert one equivalent of acetyl-CoA and 6-8 equivalents of malonyl-CoA into C14-C18 acyl-ACP species. The cytoplasmic mutant of the periplasmic thioesterase is capable of releasing free fatty acids preventing the fatty acyl yields from being directly harnessed for phospholipid biosynthesis.
 fig.1 shows fatty acid biosynthesis pathways in E.coli, stressing the role of FabG, FabZ, FabI and TesA. Catalytic cycle of the E. coli fatty acid synthesis is initiated when holo-ACP, NADPH and NADH, acetyl-CoA and malonyl-CoA undergo condensation and subsequent reduction to form butyryl-ACP. These reactions are catalyzed by the malonyl-CoA:ACP transacylase FabD, the ketosynthase FabH, the NADPH-dependent ketoreductase FabG, either the dual-function dehydratase/isomerase FabA or the monofunctional dehydratase FabZ, and the NADH-dependent enoyl reductase FabI. Then the butyryl-ACP is extended via 5-7 rounds of analogous reactions to produce a C14 to C18-ACP either fully saturated or monounsaturated. These extension cycles are catalyzed by either the ketosynthase FabB or FabF in collaboration with FabD, FabG, FabZ, and FabI. Finally, the full-length fatty acid is released from the corresponding fatty acyl-ACP via hydrolysis by C16-specific thioesterase,TesA.
FabG, FabI, FabZ play significant roles in the elongation of carbon chain of fatty acyl-ACP. It is TesA that processes fatty acyl-ACP into valuable products. Therefore, we overexpress these four enzymes which together would finally make it possible to efficiently produce fatty acids with longer carbon chains which are much more preferable biofuel precursors.
Design of Experiment
Mechanism beneath our idea suggests the Membrane Magic project enjoys two fundamental privileges: the Priority to Exportation and the Refinement of Interaction.
The Priority to Exportation
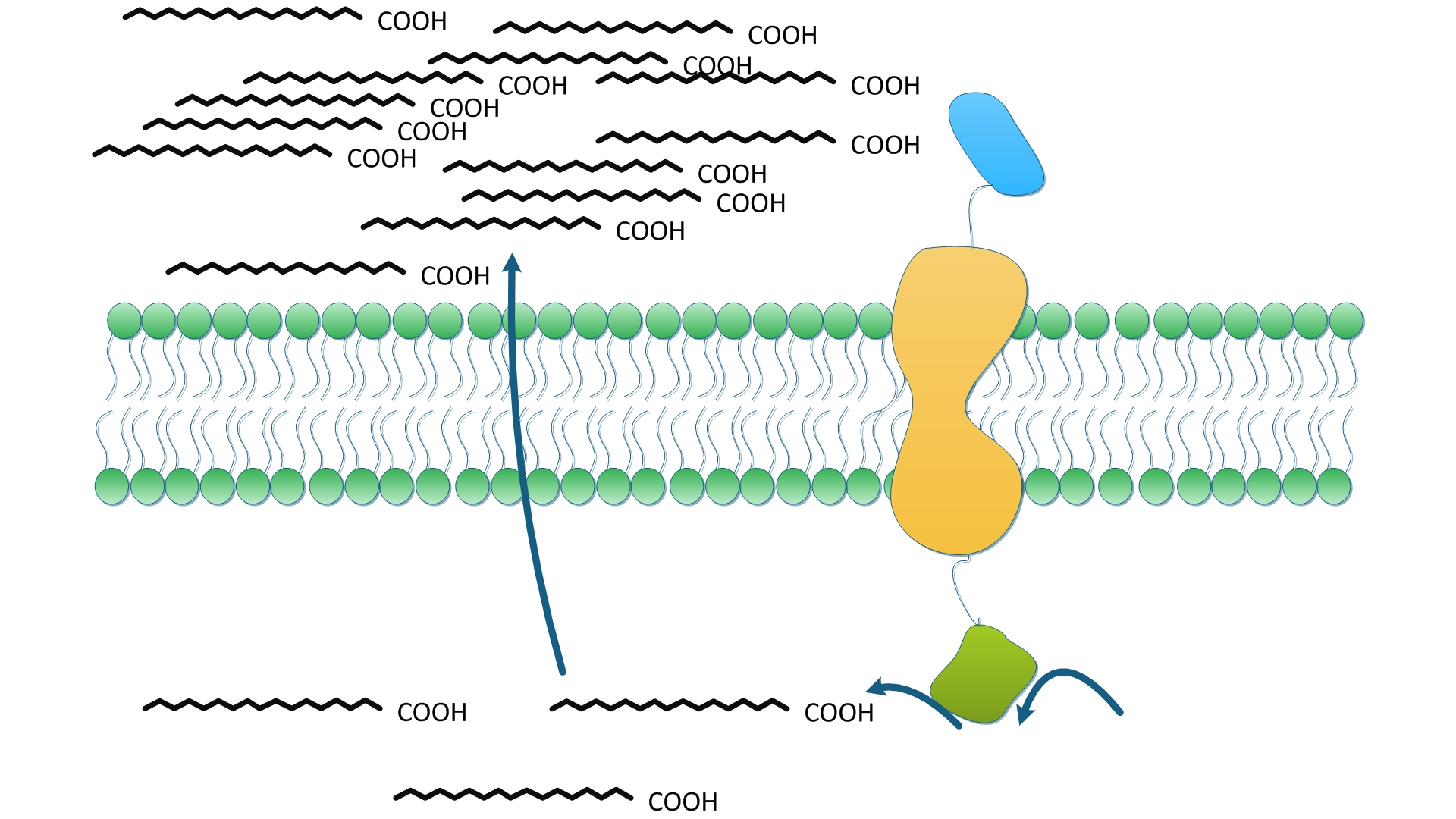 fig2shows how one solo enzyme could serve to facilitate the reaction in term of the priority to exportation. The fatty acid synthesis is terminated by the hydrolysis of fatty acyl-ACP and the release of free fatty acids into cytoplasm. We suppose TesA anchored on membrane could effectively increase the concentration of fatty acids near membrane, which in turn, facilitates the transmembrane transportation of fatty acids. Higher level of fatty acids in the culture medium make it easier to obtain and purify product and more suitable for Industrialized production. On the other hand, TesA removes fatty acyl-ACP from the reaction and thus, according to Le Chatelie principle, shifts the chemical equilibrium to accelerate and to accumulate more fatty acids.
To identify and to evaluate the priority of products to exportation, controlled experiments were designed and conducted. Wild type E.coli and E.coli expressing free-diffuse TesA were used as control groups. TesA was fused with transmembrane protein No.1 as stated in the Construction and the fusion protein was expressed in experimental group.
The Refinement of Interaction
Enzymes fused with membrane anchors will be directed to the membrane as expected right after their translation. Thus, the distribution of enzymes is restricted to the 2D membrane rather than diffusing randomly throughout the cytoplasm. Due to spatial restriction, we expect the receptor-ligand interaction that we employ to form enzymes cluster occurs at a higher frequency comparing to scaffolds in other forms. Moreover, the interaction could be stabilized by phospholipid around transmembrane domain. We believe a tandem of enzymes involved in sequential reactions could be established on the membrane swiftly and orderly.
To testify and to assess the refinement of interaction, controlled experiments were designed and conducted. Four enzymes are selected based on previous study. We express thioesterase, TesA and full complement of reductive enzymes, FabG, FabZ and FabI because moderate overexpression of TesA in gave rise to elevated fatty acid productivity and reductive enzymes could lead to 50% increase in fatty acid turnover. Wild type E.coli and E.coli expressing those four free-diffuse enzymes were used as control groups. TesA was fused with transmembrane protein No.1 as stated in the Construction and FabG, FabZ and FabI with No.2 to No.4 respectively, align with corresponding reactions occurring in sequence. Four fusion proteins were expressed together in experimental group.
Result and Discussion
Overview
Excitingly, the fatty acid biosynthesis was accelerated sharply by recruiting membrane accelerator. The introduce of TesA alone results in 50% increase in fatty acids yield. By gathering FabG, FabI, FabZ and TesA on the membrane, the production of fatty acid was enhanced significantly by 9 fold. The statistical data result strongly supports the hypothesis above that Membrane Magic make it much easier for exportation and interaction.
To demonstrate the priority to exportation and the refinement of interaction, two sets of controlled experiments have been set as following:
WT stands for E.coli transformed with corresponding plasmid(s) without exogenous gene.
F- protein here indicates enzyme diffusing randomly throughout the cytoplasm without modification.
M- protein means enzyme fused with engineered transmembrane domine and localized to E.coli membrane where enzymes aggregate and cooperate.
Assay of the Priority to Exportation
TesA is responsible for the hydrolysis of fatty acyl-ACP, with special affinity to C16- and C18- precursor. As Fig.4 A indicates, 20 hours after induction, E.coli with membrane anchored TesA experienced a 50% increase in C-16, C-18 fatty acids content and total fatty acids (Fig.4 B) in supernatant, compared with E.coli with free TesA.
 fig.4 shows fatty acid content in supernatant of three groups to evaluate the advantages of membrane anchored TesA. Fatty acids yielded from supernatant went up to 0.71mg/(L·OD). The result support that membrane anchored TesA could efficiently transfer fatty acyl-ACP into desirable fatty acids right beneath inner membrane, thus making it much easier for the final products to diffuse into periplasmic space.
 fig.5 shows fatty acid content in sediment of three groups to evaluate the advantages of membrane anchored TesA. Fig.5, on the other hand, shows fatty acids content in sedimentation also went up by 40%, up to 5.02mg/(L·OD). The result is still within our expectation since fatty acyl-ACP has been removed from the reaction system to form fatty acids. As a result, the chemical equilibrium shifts and more fatty acids would be accumulated.
We also witnessed a slight decrease in E.coli expressing free TesA compared with the wildtype, which testifies the statement in a previous study that high levels of TesA inhibits fatty acids synthesis activity but could enhance the activity at low concentrations.
Assay of the Refinement of Interaction
FabG, FabI, FabZ play significant roles in the elongation of carbon chain of fatty acyl-ACP. We tried to decrease the distance between the product by linking enzymes to aggregated membrane proteins in order to accelerate the reaction and increase the turnover per unit time. This would finally make it possible to efficiently produce fatty acids with longer carbon chains which are much more preferable biofuel precursors.
We combined these two strategies together to optimize the productivity of the system we established. Notable increase in both diversity and amount of fatty acids were detected, which lends strong support to our hypothesis.
 fig.6 shows fatty acid content in supernatant of three groups to evaluate the advantages of membrane anchored TesA,FabG,FabZ,FabI. Future Direction
transporter
FatA
脱羧
rare codon
Our study indicates a promising application prospect in biofuel production and further refinement could make it even brighter. FatA is another candidate thioesterasederived from Arabidopsis thaliana. The employment of C18-specific FatA enables the engineered biosynthesis system to produce fatty acids with even longer carbon chain.
|