Team:Bielefeld-Germany/Labjournal/week19
From 2012.igem.org
Contents |
Week 19 (09/03 - 09/09/12)
Monday September 3rd
- Team Cultivation & Purification:
- Made one half of the SDS-Pages of the flask cultivation from 08/30.
- Work on fermentation of B.pumilus laccase (08/31) was continued and we performed the cell disruption by high-pressure homogeniser in 3 cycles. Then it was purificated by using a Talon column. The product was given to the activity team for analysis.
- Fermentation of E.coli KRX with His-tagged GFP for the cellulose binding team --> Settings: fermenter: Braun, autoinduction medium, 3 L, 37 °C, stirrer on cascade to hold a pO2 of 50 %, airflow: 2 NF/m. This time we cultivated only for 9 hours and then decreased the temperature at 20°C for a better folding of the protein for 1 hour. We had some problems with cascade at the end of the fermentation.
- Cells of today's fermentation of His-tagged GFP were disrupted via high-pressure homogeniser and laccase was purified by using the Talon column. The flow-through fluoresced very strong, so that not that much of the protein had bound to the column. Conclusion: The column had to be reloaded.
- Fermentation of E.coli KRX with laccase from B.pumilus. --> Settings: fermenter: Braun, autoinduction medium, 3 L, 37 °C, stirrer on cascade to hold a pO2 of 50 %, airflow: 2 NF/m, 10 hours. We only cultivated 10 hours, because we saw that a great amount of our cells already died.
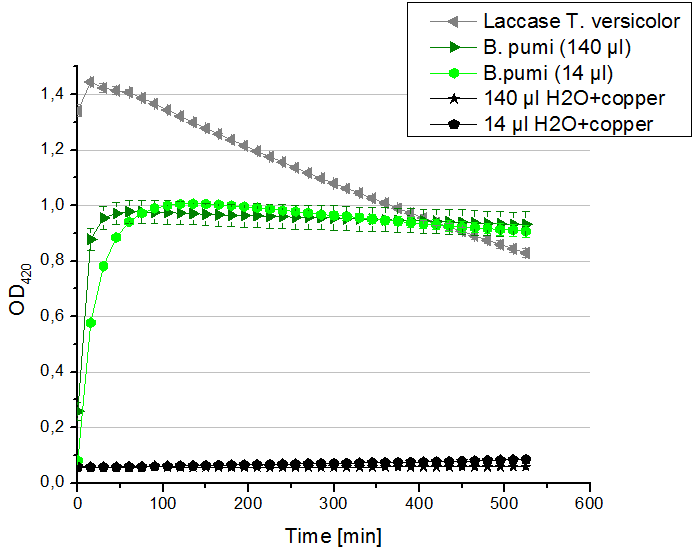
- Team Activity Tests: Today our hard-working Team Cultivation offered us some laccases from B. pumi. After fermentation and TALON purification the laccase was still hanging around in imidazole buffer, so we rebuffered it into nanopure water. We incubated the sample with copper and considered to measure the protein concentration before running the activity tests. Using the protocol for determining the protein concentration via Bradford (standards were 1 mg/ml, 1.5 mg/ml and 2 mg/ml BSA) we got a final B. pumi laccase concentration of 0.22 mg/ml. With this information we designed the following activity test different as usual. As a positive control we took 140 µl of our bought laccase from T. versicolor with a concentration of 0,021 mg/ml. Accordingly to this concentration we applied 14 µl of the B. pumi laccase to get approximately the same amount of laccase into each well. Additionally we tested 140 µl of the B. pumi laccase to make sure that we will definitely see any activity. After measuring the samples the whole night we got our results in the morning. It turned out, that our store bought laccase was rapidly active as seen before in our activity tests. The B. pumi laccase was also active. Using 140 µl of it led to the maximal state of oxidation of ABTS after 45 minutes. With the reduced amount of 14 µl of laccase this optimum was reached after 90 minutes. For our following activity tests we want to include the protein concentration again.
Tuesday September 4th
- We signed in for our tracks today! Our first choice is the 'Environment' track since it is obvious that we want to clean up drinking water and therefore help our environment. The second choice is the 'New Application' track. We are hoping our filter system can be applied in wastewater treatment plants and establishes a new area of water purification.
Wednesday September 5th
Thursday September 6th
Friday September 7th
Saturday September 8th
Sunday September 9th
Sunday
| 55px | | | | | | | | | | |
 "
"