Team:Tokyo Tech/Experiment/C6
From 2012.igem.org
Construction of the 3OC6HSL-dependent 3OC12HSL production module
For construction of the 3OC6HSL-dependent 3OC12HSL production module, we firstly constructed a new part Plux-LasI ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K934022 BBa_K934022]). Plux-LasI cell is an engineered E.coli that contains a 3OC6HSL-dependent LasI generator and a constitutive LuxR generator. As a 3OC6HSL-dependent LasI generator, we constructed a new Biobrick part Plux-LasI ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K934022 BBa_K934022]) by combining Plux promoter ([http://partsregistry.org/wiki/index.php?title=Part:BBa_R0062 BBa_R0062]) and LasI ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K081016 BBa_K081016] ). As a constitutive LuxR generator, we used Ptet-LuxR ([http://partsregistry.org/wiki/index.php?title=Part:BBa_S03119 BBa_S03119]). By introducing Plux-LasI ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K934022 BBa_K934022] ) and Ptet-LuxR ([http://partsregistry.org/wiki/index.php?title=Part:BBa_S03119 BBa_S03119]) into E.coli strain JM2.300, we constructed Plux-LasI cell. Then we performed a reporter assay by using Las reporter cell to characterize the function of Plux-LasI ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K934022 BBa_K934022]) . As the negative control of 3OC12HSL production, we prepared 3OC12HSL non-producer cell (ΔP-LasI cell) that contains, in addition to Ptet-LuxR ([http://partsregistry.org/wiki/index.php?title=Part:BBa_S03119 BBa_S03119]), promoterless-LasI ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K081016 BBa_K081016]) instead of Plux-LasI ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K934022 BBa_K934022]) (Fig2-1-3-1-2).
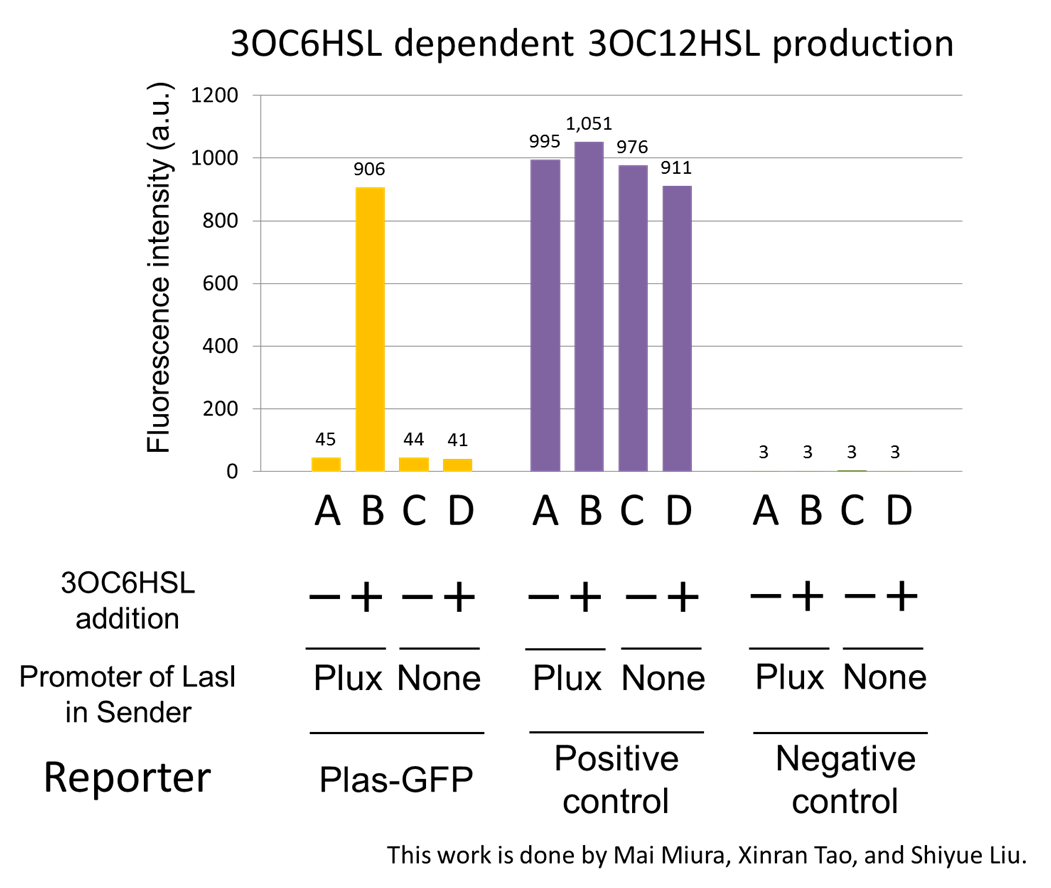
The ⊿P-LasI cell does not produce 3OC12HSL even though 3OC6HSL exist. The supernatants of the cultures of these modules were used as the inducer in the reporter assay (Fig2-1-3-1-3). We prepared four conditions as follow.
A) Culture containing Plux-LasI cell without 3OC6HSL induction
B) Culture containing Plux-LasI cell with 3OC6HSL induction
C) Culture containing ⊿P-LasI cell without 3OC6HSL induction
D) Culture containing ⊿P-LasI cell with 3OC6HSL induction
Using the supernatant of the four culture conditions, we performed the reporter assay.
In the reporter assay, we used a Las reporter strain that contains Ptrc-LasR and Plas-GFP ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K649001 BBa_K649001]). Also, a reporter cell that expresses GFP constitutively and a reporter cell that does not express GFP were used as the positive control and the negative control, respectively.
Fig2-1-3-1-4 shows fluorescence intensities by the reporter cells dependent on different conditions. Only when the supernatant of condition B was used, the fluorescence intensity of the Las reporter cell increased, while the supernatants of other three conditions did not affect. Comparing the results of the condition A and B, it can be said that with the induction of 3OC6HSL to Plux-LasI cell, the fluorescence intensity of the LasI reporter cell increased by 20-folds. This result indicates that Plux-LasI cell produced 3OC12HSL in response to 3OC6HSL induction by the function of Plux-LasI ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K934022 BBa_K934022] ). From this experiment, we confirmed that a new part Plux-LasI ([http://partsregistry.org/wiki/index.php?title=Part:BBa_K934022 BBa_K934022] ) synthesized enough concentration of 3OC12HSL to induce the Las reporter cell. [Protocol]
 "
"