Template:Kyoto/Project/FlowerFairy
From 2012.igem.org
(→Modifying FT gene for Biobrick) |
(→1.EXPRESSION) |
||
| Line 55: | Line 55: | ||
[[File:FTzenN.png|thumb|left|650px|Fig.1-1<br> Necessity for mutation of ''FT'' cDNA. ''FT'' sequence had two cleavage sites of iGEM restriction enzymes.]] | [[File:FTzenN.png|thumb|left|650px|Fig.1-1<br> Necessity for mutation of ''FT'' cDNA. ''FT'' sequence had two cleavage sites of iGEM restriction enzymes.]] | ||
| - | We constructed the plasmid shown in the Fig.1- | + | We constructed the plasmid shown in the Fig.1-2. One was ''FT'' gene with T7 promoter |
[http://partsregistry.org/Part:BBa_I719005 BBa_I719005] and 6His tag, and the other was ''FT'' gene only with T7 promoter. T7 promoter is strong promoter and IPTG switches transcription of T7 RNA polymerase. 6His tag, which is used at later steps, enables us to purify protein from ''E.coli'' with affinity chromatography. | [http://partsregistry.org/Part:BBa_I719005 BBa_I719005] and 6His tag, and the other was ''FT'' gene only with T7 promoter. T7 promoter is strong promoter and IPTG switches transcription of T7 RNA polymerase. 6His tag, which is used at later steps, enables us to purify protein from ''E.coli'' with affinity chromatography. | ||
<br clear="both"> | <br clear="both"> | ||
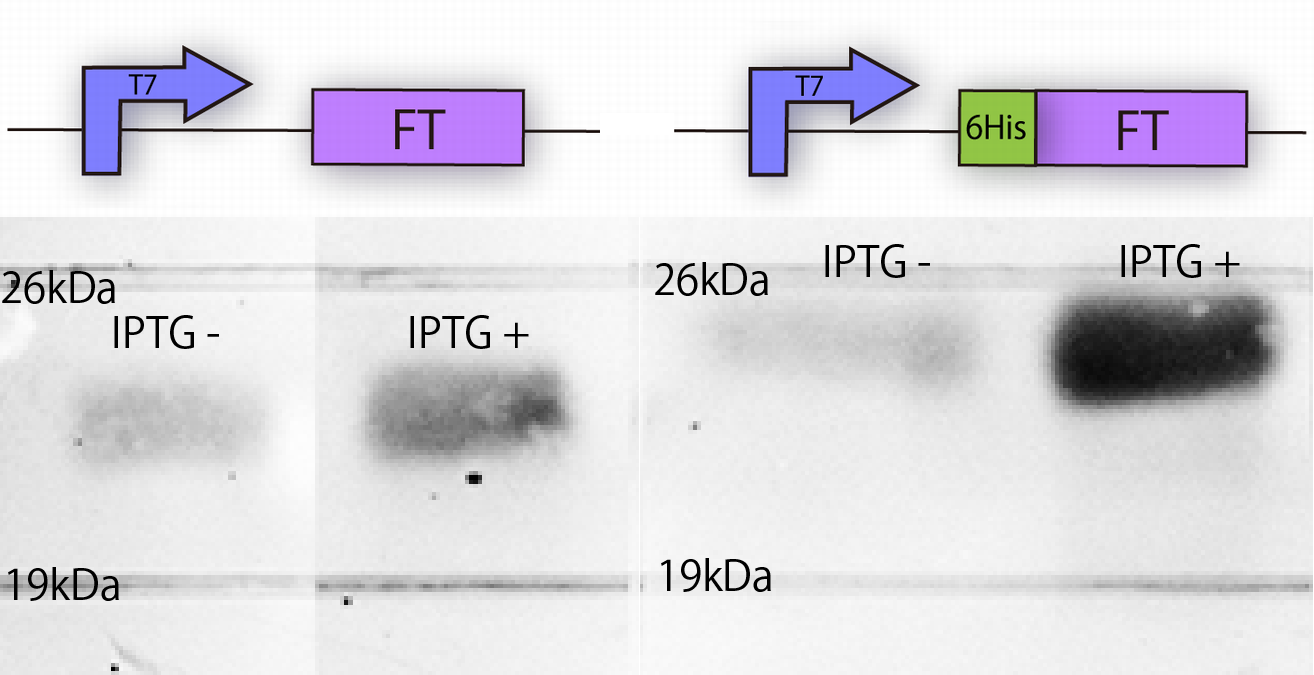
| - | [[File:FTblotting.png|thumb|left|300px|Fig.1- | + | [[File:FTblotting.png|thumb|left|300px|Fig.1-2 ''FT'' construction and the result of Western blotting against FT and 6 His:FT proteins. .]] |
=== Confirming expression of FT === | === Confirming expression of FT === | ||
Revision as of 14:31, 25 October 2012
Contents |
Realizing Flower Fairy in real world
Have you ever seen flower fairies? Probably no (some of you might come across them in your childhood), because they are imaginary creatures existing only in fairy tales. Don’t you think it is happy if you can live with flower fairies? In addition to feeling happiness, their lovely powers to make flowers bloom would be profitable for us, including application to agriculture. That’s why we set our project to realize it with synthetic biology, Flower Fairy E.coli!!
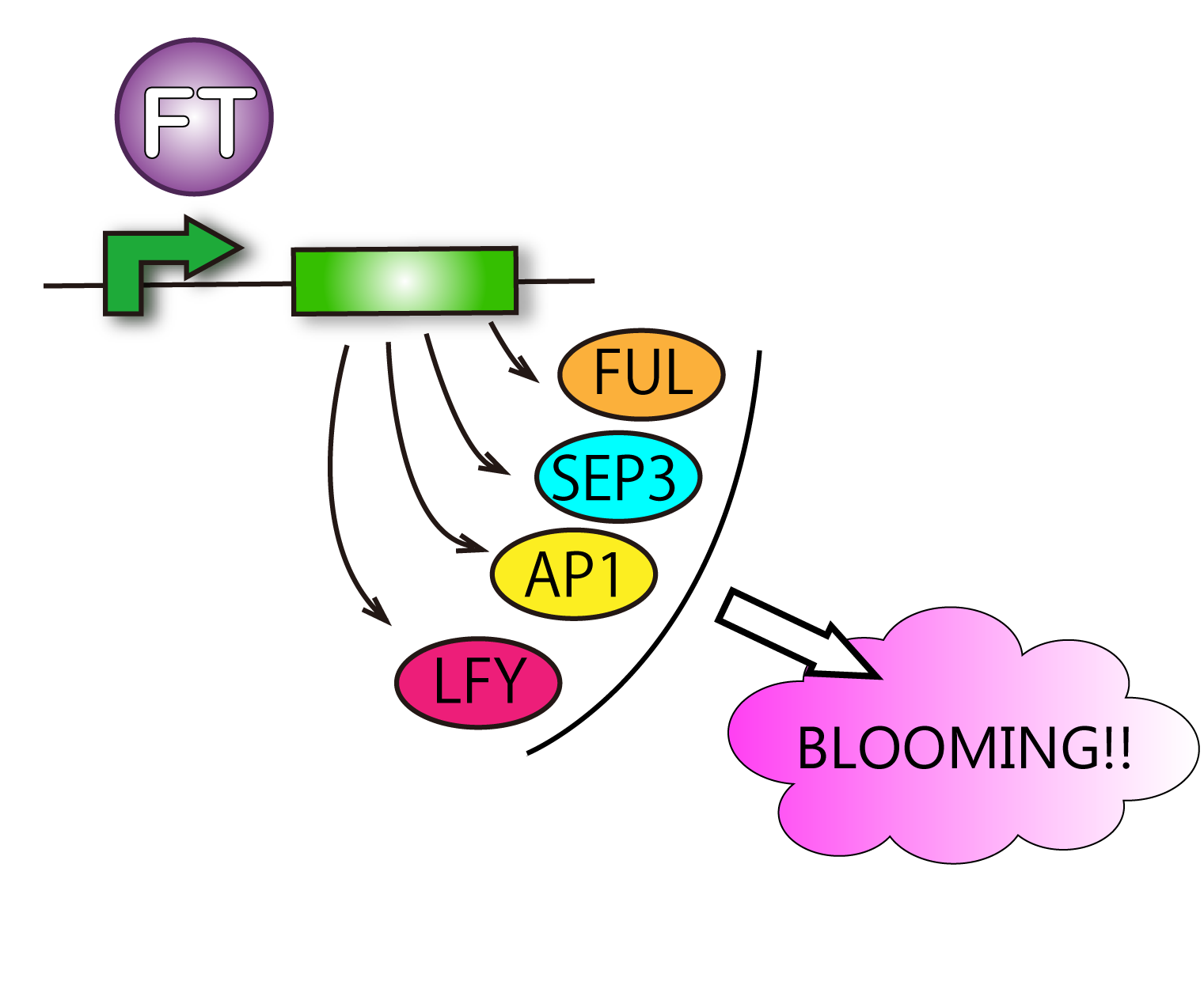
Our goal is to produce E.coli which can make flowers bloom as Flower Fairies. To make it possible, we focus on FT protein, known as Florigen.This protein is a kind of plant hormone. At first, FT protein is produced in leaves. After that, it moves to shoot apex and bloom flowers. Therefore, FT protein is a key to our project.
Our Goal is to Put Flower Fairy E.coli on leaves for flower formation!
When you want to use our Flower Fairy E.coli, there is just one thing you must do to get ready. Only putting them on leaves! When you spread Flower Fairy E.coli with R9 peptide toghether, FT protein is secreted by them and penetrates into cell membranes of plants, and the plants bloom.
We have to go through four steps in order to achieve our goal――Flower Fairy E.coli.
These four steps are composed of “EXPRESSION”,”SECRETION”,
”PENETRATION”, and ”ACTIVATION”
On each step, we have some problems to be solved.
“EXPRESSION”; It is unclear whether ''E.coli'' (prokaryote) could express FT protein, because FT protein is derived from plant cells (eukaryote).
”SECRETION”; After produced, FT protein has to get out of the ''E.coli''.
”PENETRATION”; FT protein has to penetrate into plant cells.
”ACTIVATION”; Even though FT protein could get inside of the cells, it is unknown whether FT in ''E.coli'' can activate shoot apex cells and lead to flower bloming.
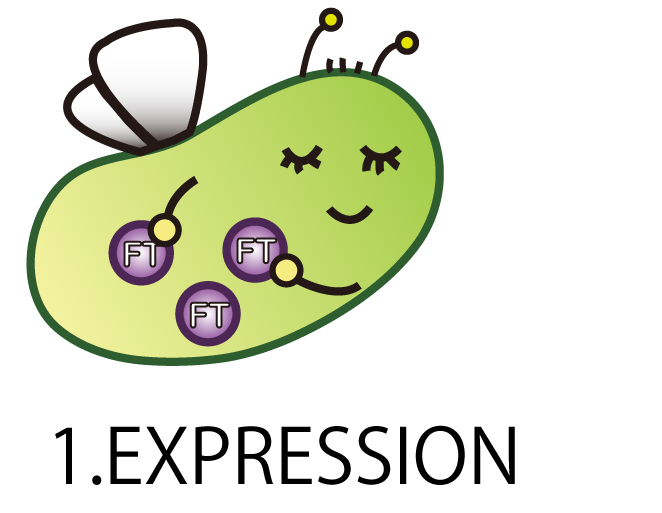
1.EXPRESSION

On the first step; EXPRESSION, We needed to make E.coli produce FT protein.
As a matter of course, E.coli doesn’t have FT gene. Therefore we had to make a new BioBrick part of FT gene and introduce it into E.coli.
Modifying FT gene for Biobrick
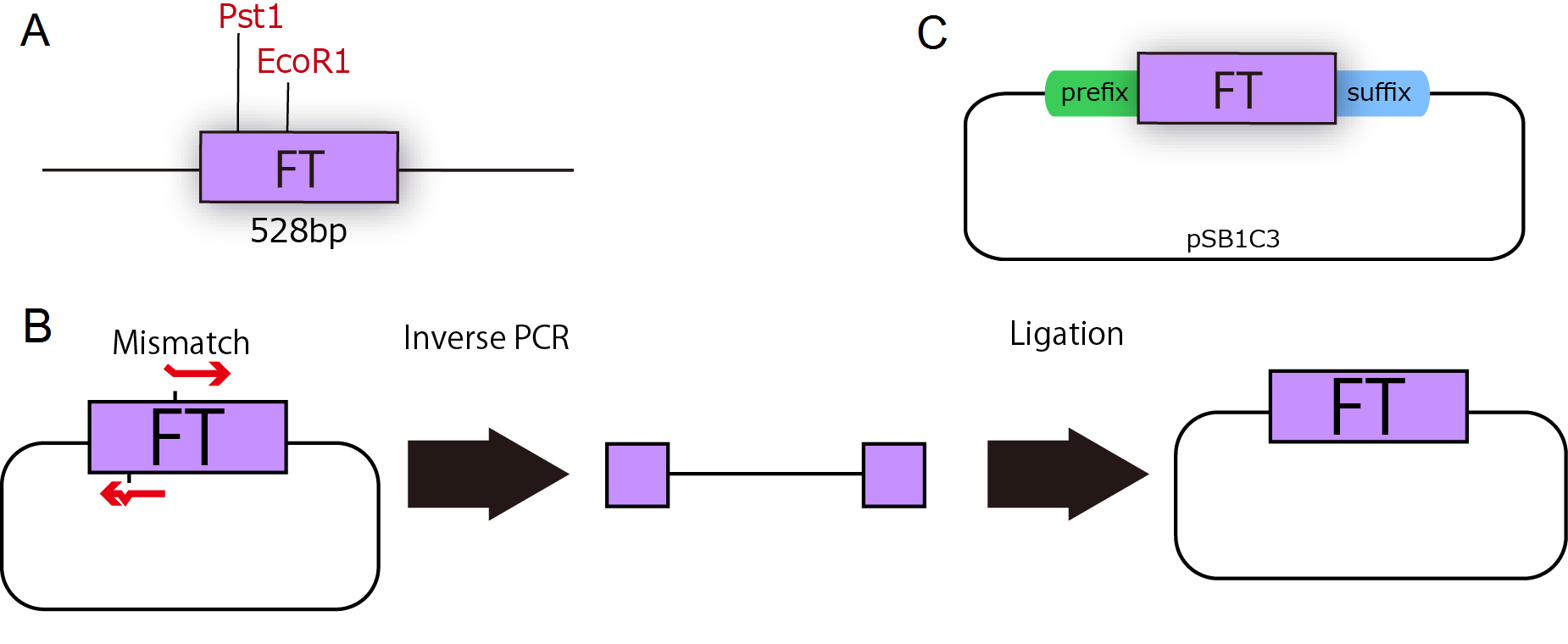
FT gene is derived from Arabidopsis thaliana, a model plant. Professor Araki in Kyoto University kindly gave us FT cDNA in TOPO blunt end 2(Invitrogen). FT sequence had two cleavage sites of iGEM restriction enzymes, EcoR1 and Pst1 (Fig.1-1 A), therefore we modified FT gene sequences by Inverse PCR with primers containing two base mismatches between primer and cDNA(Fig.1-1 B). This PCR enables us to mutate FT gene. As a result, we could get mutated plasmids, which are not cleaved by iGEM restriction enzymes, and by fastening prefix and suffix to FT ,we could make new biobrick parts of FT (Fig.1-1 C).
We constructed the plasmid shown in the Fig.1-2. One was FT gene with T7 promoter
[http://partsregistry.org/Part:BBa_I719005 BBa_I719005] and 6His tag, and the other was FT gene only with T7 promoter. T7 promoter is strong promoter and IPTG switches transcription of T7 RNA polymerase. 6His tag, which is used at later steps, enables us to purify protein from E.coli with affinity chromatography.
Confirming expression of FT
Then we confirmed whether E. coli was correctly expressing FT by Western blotting, because there is possibility that E.coli cannot translate FT or that FT is not stable or is toxic.
In order to confirm the expression of FT protein, we performed Western blotting using anti-FT goat antibody and checked the place of the FT protein band. As a result, FT and 6 His:FT bands were observed at the expected molecular weight region(Fig.1-5).
We succeeded in mutation and expression of FT and 6 His:FT proteins in E.coli
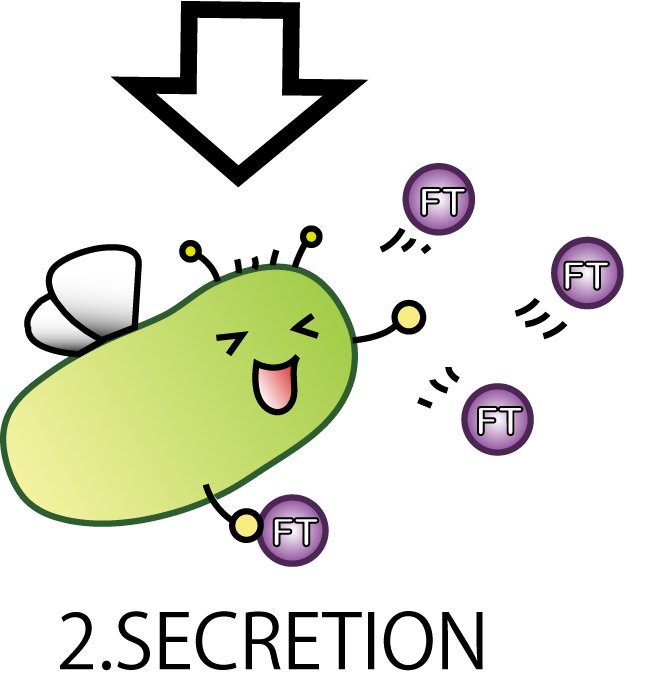
2.SECRETION
On the second step; SECRETION E.coli secretes florigen outside of the cell.
Now our E.coli can produce FT protein, but a big issue still remains: how they can transport proteins to the outside of the cells? Many people might think cell lysis is the best way. But on our project, lysis is not so good. It is because lysis possibly causes all Fairies death. Then, the effect of Flower Fairy E.coli won't continue. So, we adopted a new method by which E.coli can transport FT protein without lysis.
Transport FT protein into periplasm
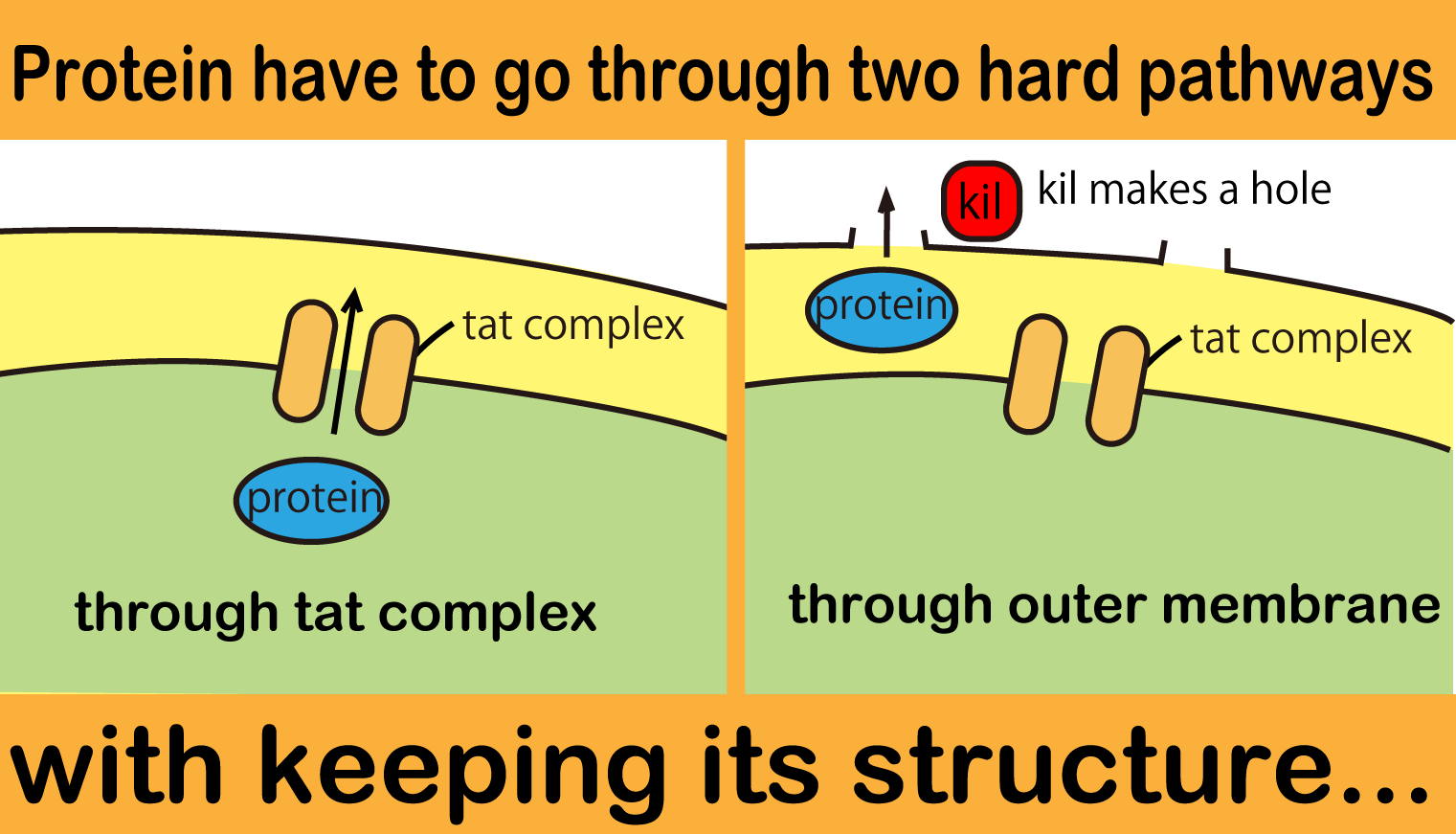
We chose transport system of wild E.coli, called The Twin Arginate Translocation(Tat) pathway. The points that Tat pathway is better than the other secretion system is that E.coli secretes proteins without lysis and keeping proteins' conformation. The mechanism of Tat pathway is that Tat transporter recognizes TorA signal and transports proteins that have TorA signal at N terminal into periplasm.
We tried to combine TorA signal with FT protein. Actually, we searched previous iGEM projects and found some teams submitted TorA signal as iGEM parts (for example, [http://partsregistry.org/Part:BBa_K638402 BBa_K638402]). There were, however, two big problems. One problem is that these parts don’t have RBS, so when iGEMers use these parts, they have to spend additional processing time. The other is that stop codons appear between signal region and target coding sequence when iGEMers assemble these parts and some parts by standard or 3A assembly. So if iGEMers use these parts, TorA-fusion protein is not expressed or only TorA is expressed.
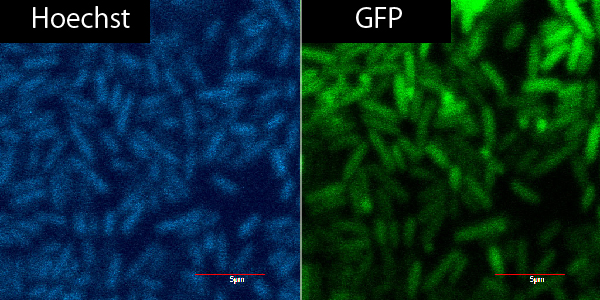
So we made new applicable TorA signal (BBa_). Our part has two advantages. BBa_ contains RBS and doesn’t have stop codon between signal region and target cording sequence. We read the sequence data of Bba_ and confirmed stop codon doesn't appear when it is used in Standard or 3A assembly. Using green fluorescent protein (GFP) as a target protein, we observed the TorA-GFP fusion-expressing cells (Fig.2-1). The TorA-GFP fusion was successfully expressed. This means RBS in our TorA signal worked.
Evaluation of kil protein
Using Tat secretion pathway, FT protein is transported into periplasm.
In previous iGEM competitions, some kinds of parts and devices for protein translocation were already developed. One of the most widely used parts is lysis cassette. This part causes cell lysis and, as a result, makes E.coli scatter materials it includes. This style is, unfortunately, not suitable for our project because of the possibility of accidental all-death. Generally speaking, the concentration of E.coli on a flower is not high so that it can’t be ignored that the possibility of occurring all cell-death. Once all our Fairies disappear, a flower wouldn’t bloom. In addition to that, lysis of E.coli is NOT CUTE.
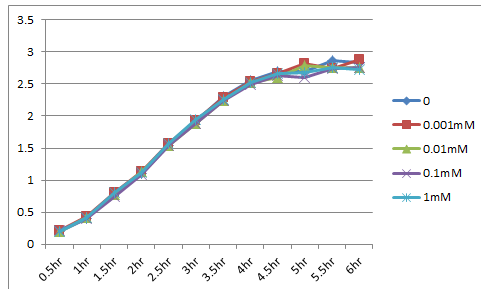
Kil makes holes in outer membrane and we expect that a protein goes through these holes. Larger hole area means larger amount of proteins can go out. On the other hand, the function of outer membrane is essential for E.coli to survive. Overexpression of kil, therefore, causes cell death. For this reason, we must check whether our kil gene is overexpressed or not.
We made the construct, lacp-RBS-kil-double terminator, whose backbone is pSB3C5. After culturing for 18hr at 37℃, we eliminated the supernatant using a centrifuge, and diluted it until OD600=0.1. Then we dispensed it. The dispense volume was 3mL. We added 0/0.001/0.01/0.1/1mM IPTG to each. While culturing again at 37℃, we measured OD600. The table below shows the results. This result indicates that the expression of LacP-RBS-kil-DT(pSB3C5) makes no effect on survival of E.coli.
Construction of Tat cassette
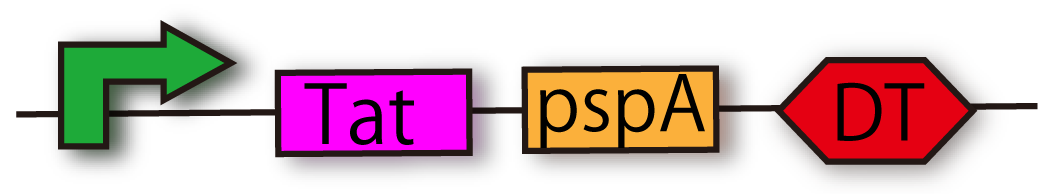
Tat secretion cassette with constitutive promoter [http://partsregistry.org/Part:BBa_K797004 (BBa_K797004)]
Our secretion system is constructed by TatABCD, kil and another gene. Another gene is pspA (phage-shock protein A) gene and E.coli has it originally. This gene is expressed when their inner membrane is damaged. pspA maintains membrane potential and H+ concentration gradient between periplasm and cytoplasm. Our secretion system makes many holes in inner and outer membranes. In other words, E.coli which has our secretion system is under the membrane stress conditions. But by introducing pspA into our Flower Fairy E.coli, the E.coli comes to be able to maintain the vitality, though they have many holes in the membrane.
This cassette allows E.coli to secrete proteins with TorA signal. Wild type Tat protein secretion system is so weak that Kyoto 2012 constructs Tat cassette to reinforce the ability of transportation of Tat system. This part includes TatA, B and C proteins coding region and pspA (phage shock protein A). Tat A, B and C proteins are the main components of Tat complex where proteins with TorA signal go through, and pspA can encourage protein secretion via Tat system. Kyoto 2012 suggests this new way of secretion and provides iGEMers with this cassette regulated by constitutive promoter.
We checked the sequence of TatABCD [http://partsregistry.org/Part:BBa_K797000 (BBa_K797000)] and the sequence of pspA [http://partsregistry.org/Part:BBa_K797001 (BBa_K797001)] individually, and then, we made Tat construction composed of constitutive promoter [http://partsregistry.org/Part:BBa_J23107 (BBa_J23107)], TatABCD [http://partsregistry.org/Part:BBa_K797000 (BBa_K797000)]
,pspA [http://partsregistry.org/Part:BBa_K797001 (BBa_K797001)] and double terminator [http://partsregistry.org/Part:BBa_B0015 (BBa_B0015)]
. This Tat secretion cassette is too long device to sequence, so that we performed electrophoresis of this cassette and confirmed the length of our parts.
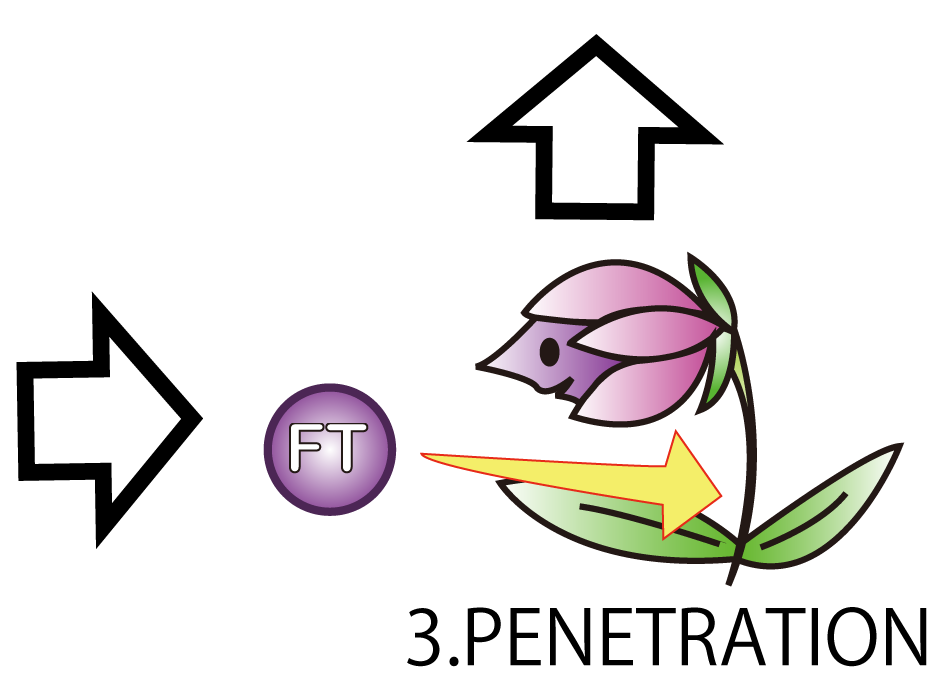
3.PENETRATION

On the third step; PENETRATION. In order to induce flower formation, FT protein from E.coli must enter into plant cells. This is because FT protein upregulates other proteins leading to flower formation in plant cells.
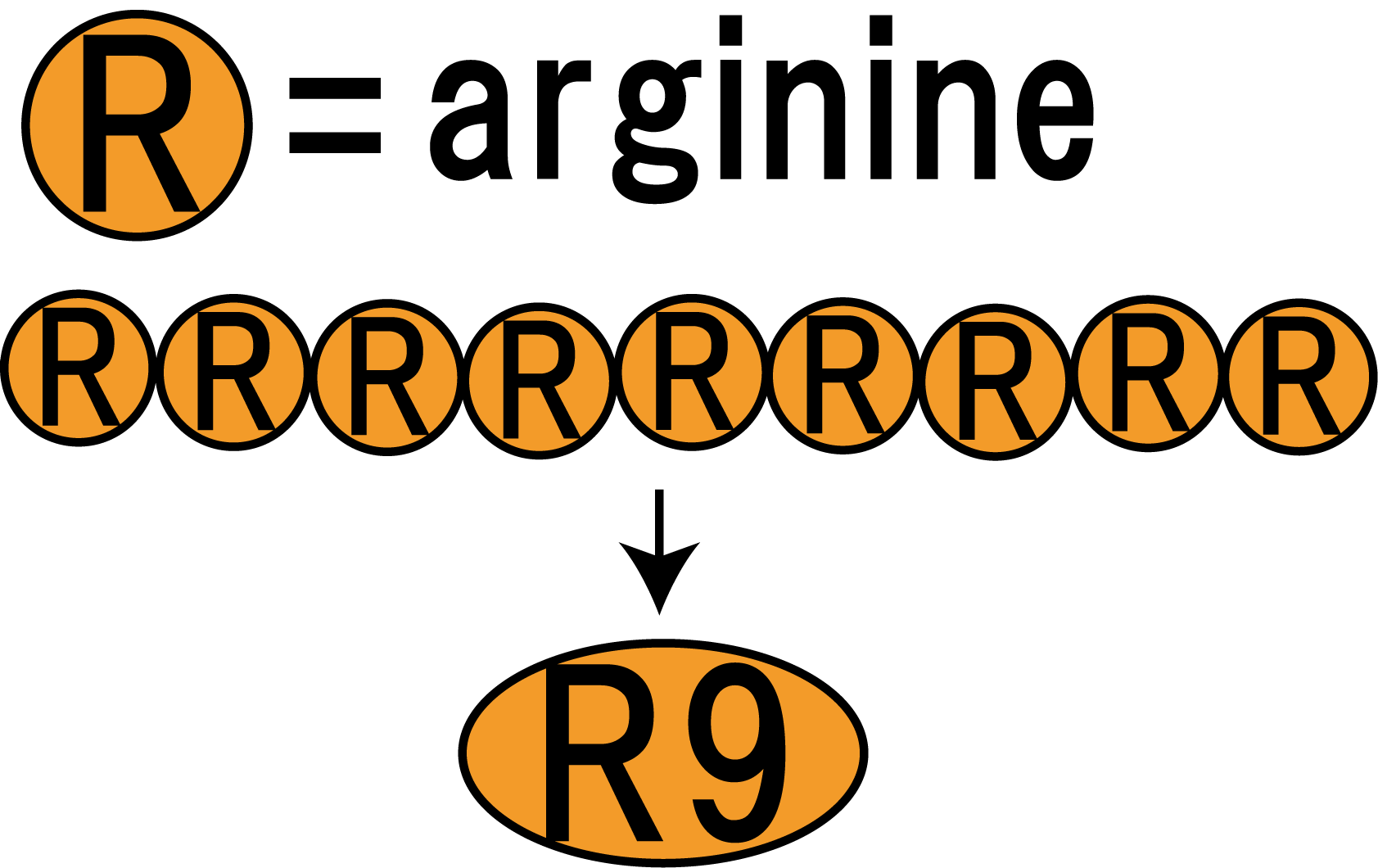
However, normally proteins cannot penetrate cell membranes of plants. Therefore, we needed a method to send FT protein into plant cells. Thanks to the advice from Doctor Washida, we found the method for the cell membrane penetration. A polyarginine ,called R9 peptides, makes FT protein penetrate a cell membrane in this method. This is a type of Cell Penetrating Peptide (CPP).
R9 peptide enables FT protein penetrate membranes by endocytosis
R9 peptide consists of nine arginine residues. (Fig.3-1) It is known through reserch of CPP, arginine-rich peptide tends to penetrate cell memblanes. R9 peptide is suited to our purpose.
This is the mechanism of how R9 peptides work.(Fig3-2)
Firstly, R9 peptides adhere to a cell membrane of a plant because of hydrophobic character.
Secondly, cells response to this stimulus and induce endocytosis.
Finally, FT proteins near R9 are taken in the cell.
We tried to connect R9 with protein for a high degree of penetration efficiency
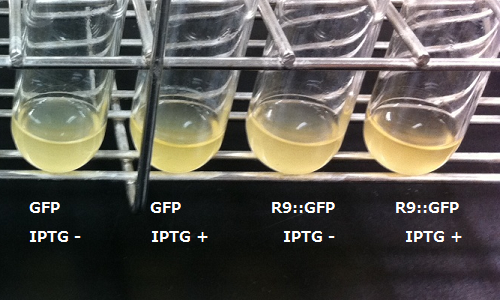
We thought shorting the distance between R9 and protein led to a high degree of penetration efficiency, because R9 induces endocytosis with R9 itself bonded to membranes. Therefore we connected R9 with protein. In order to visualize the function of R9, we tried to prepare R9::GFP fusion protein. However, E.coli expressing the R9::GFP fusion protein was poor in growth.(Fig3-3) Then, we checked R9 effect on R9::GFP fusion protein's expression by Western blotting and RT-PCR. We succeeded in confirming the existence of the mRNA (Fig3-4), but we didn’t find the protein (Fig3-5). These results insists poor translation or quick breakdown of the protein.
We used the existing GFP generator part, [http://partsregistry.org/Part:BBa_I746915 BBa_I746915]. perhaps because of its cytotoxicity of R9 against E.coli.
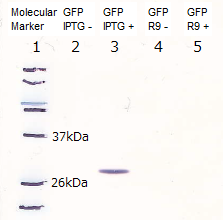
Lane1: Molecular marker
Lane2: GFP([Part:BBa_I746915]) Cell lysate 10µL, not induced
Lane3: GFP([Part:BBa_I746915]) Cell lysate 10µL, IPTG induced
Lane4: R9::GFP Cell lysate 10µL, not induced
Lane5: R9::GFP Cell lysate 10µL, IPTG induced
We prepare R9 and GFP individually to visualize the R9 peptides
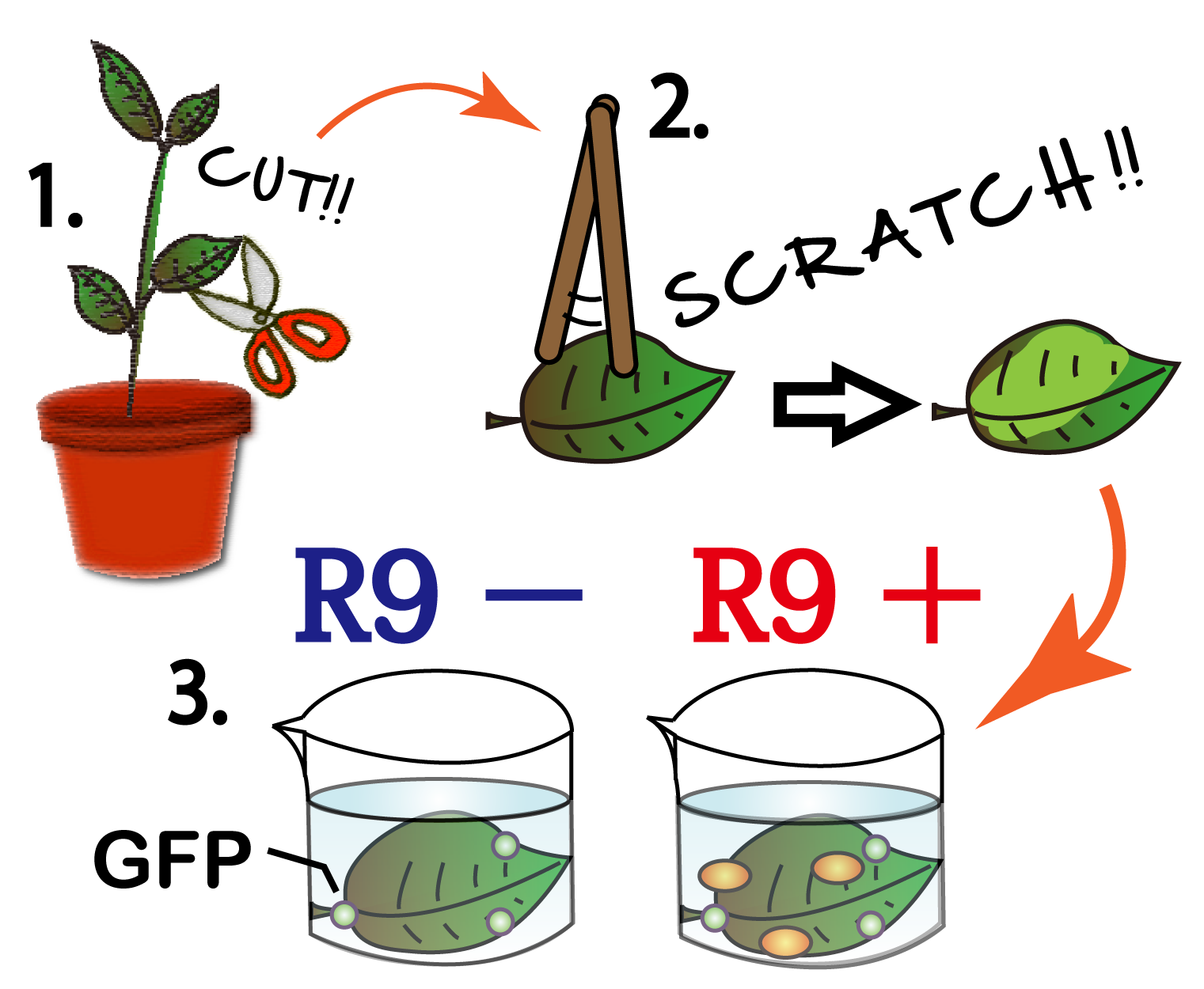
We wondered whether R9 system really work properly and put FT protein into plant cells? Although animal cells often endocytose, there are few examples of plant cell’s endocytosis. Therefore, We need to verify the system. To verify it, we performed followed experiment.(Fig3-6)
First,we scratched the cuticle of Arabidopsis thaliana, a model plant. Second,we soaked them into a solution of only GEP, or GFP and R9 for each. This GFP protein was purified with 6 His tag inserted in our 1st Step,"Expression". After 5 minutes we washed cells by PBS in order to wash GFP and R9 peptide away from leaves. After that, we compared leaves including only GFP, and leaves including R9 and GFP for each.
Then we succeeded in getting the Figure of GFP fluorescence.(Fig3-7)
The control group on the left were soaked in only GFP, and the experimental group on the right were soaked in GFP and R9. These fluorescence indicated that R9 peptide work properly kept GFP in or around plant cells. This Figure strongly suggests that R9 peptide works successfully and penetrates cell membrane with GFP, because this was taken by a confocal microscopy and seen as cross sections.
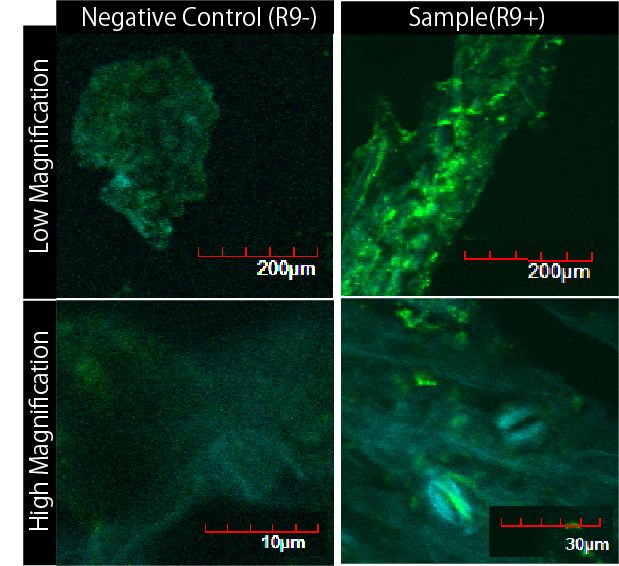
We can make FT protein penetrate cell membrane of plants by R9 peptide function.
4. Activation

On the final step; Activation. We verified whether FT normally worked in plant cells.
FT protein is derived from plant cells and it is capable of post-translational modification. E.coli cannot do post-translational modification, so FT protein derived from Flower Fairy E.coli may not work normally. As a final step, we tried to confirm whether FT protein by our E.coli led to flower formation.
How to Verify FT Function
FT protein increases transcriptive activity of several proteins which lead to flower formation. For that reason it can be said that we have verified FT function when we have found rises of activities of the proteins.
Although such proteins activated by FT are various, we check FRUITFULL(FUL), SEPALLATA3(SEP3) and APETALA 1(AP1). SEPALLATA3(SEP3) and FRUITFULL(FUL). This is because AP1 is the representative protein activated by FT, and SEP3 and FUL are activated in leaves. It is difficult for us to handle cells of tips of stems. So we focused on cells of leaves. Leaves' cells are easy to handle for us. We performed RT-PCR in order to investigate FT protein's function.
We performed RT-PCR to compare mRNA expression of Arabidopsis leaves treated with/without FT.
Two types of samples were prepared, one is treated with FT and the other is treated with GFP as a control.
GFP was used as a control because its molecular weight(27kDa) is relatively similar to that of FT(20kDa.)
Fig.4-2 is the result of RT-PCR.

30mg of leaves of Arabidopsis thaliana before bolting were used for one sample. FT or GFP protein and R9 peptide were diluted in PBS (pH7.4), 50ug/L and 500ug/uL each. Leaves were soaked into FT-R9 or GFP-R9 solution for 5min. and incubated for 16hr. in PBS(pH7.4.) After incubation, leaves were freezed with liquid nitrogen and glinded immediately.
Total RNA was extracted by phenol-chloroform extraction. cDNA was synthesized by reverse transcription and used as templates of RT-PCR. TUBULIN was used for internal control of mRNA expression.
Lane1: TUBULIN (GFP-R9 treated) amplicon 61bp
Lane2: TUBULIN (FT-R9 treated)
Lane3: FUL (GFP-R9 treated) amplicon 132bp
Lane4: FUL (FT-R9 treated)
Lane5: SEP3 (GFP-R9 treated) amplicon 87bp
Lane6: SEP3 (FT-R9 treated)
Lane7: AP1 (GFP-R9 treated) amplicon 958bp
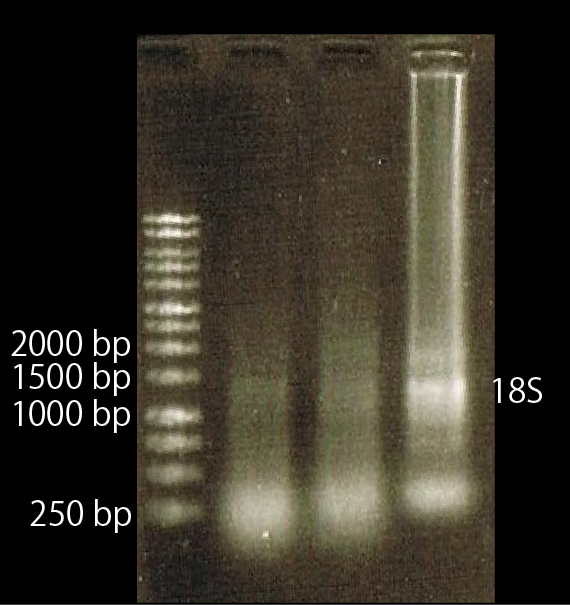
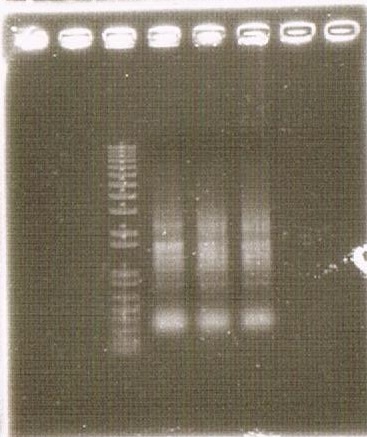
As shown above, we got no correct ampicon band, even of tubulin, which is high expressed gene we used as internal control. One possible reason of this failure is the poor quality of extracted RNA in this experiment. To check this, we compared the total RNA by electrophoresis, shown in fig.4-3.
As shown in the Fig.4-3, RNA samples in this time are degradated.
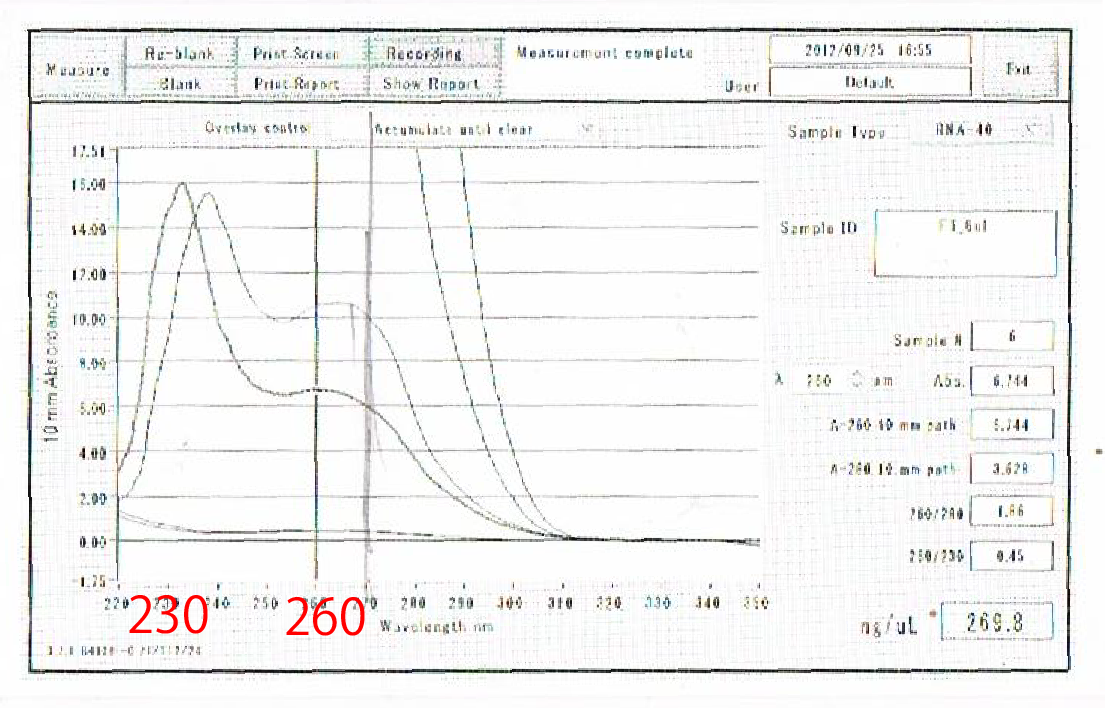
Moreover, the waveforms of them had a law peak at 260nm(Fig.4-4.)
So, it was required to improve the method of RNA extraction.
We improved following things;
1. Total leaf volume was increased.
2. Samples ware freezed with liquid nitrogen and suspended in ISOGEN more rapidly.
3. We centrifuged samples and collected supernatant twice after adding ISOGEN.
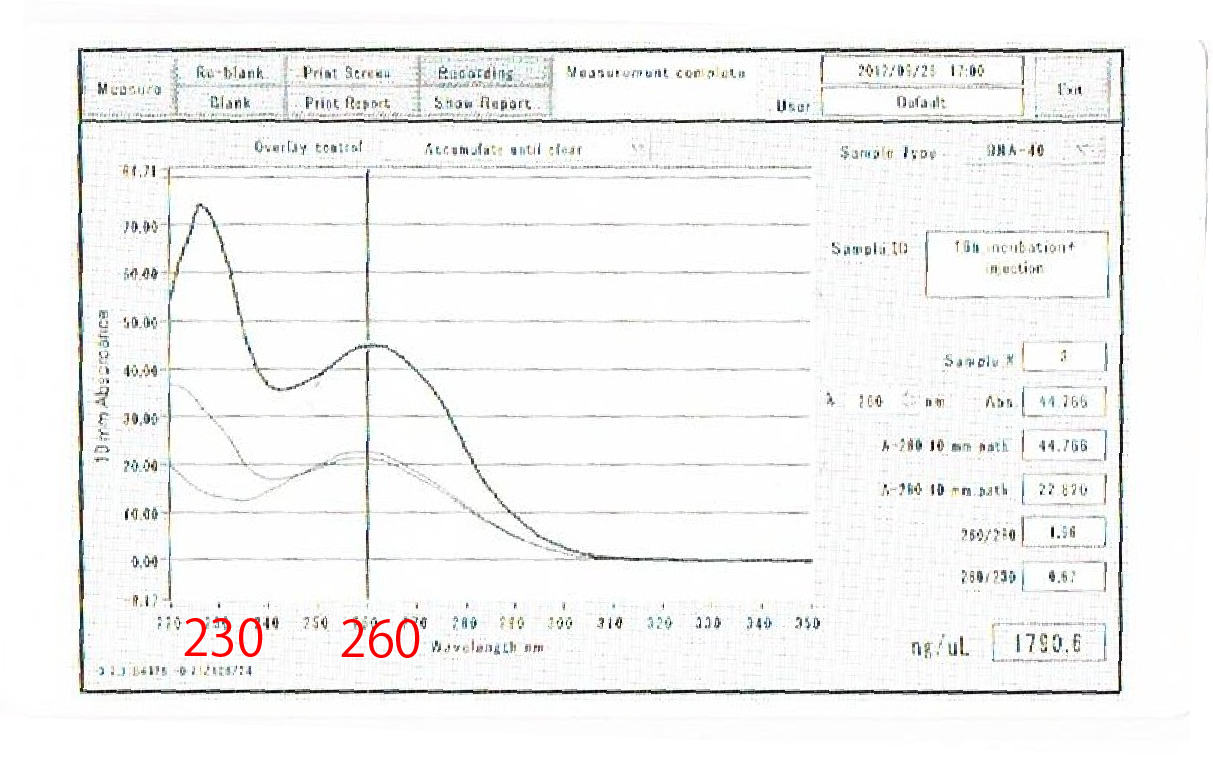
After the improvement, 260nm peak became higher and the degradation is minimized(Fig.4-4, 4-5.)
Then we used better quality of RNA for reverse transcription and retried RT-PCR.
Fig.4-6 shows the result of this RT-PCR.
From these results, we could not confirm the function of FT, upregulation of FUL, SEP3, and AP1.
However, now that we can get good quality of RNA and amplify genes successfully, we are trying to check the function of FT.
Achievement
1.Expression
- Mutate FT sequence
- Standardize FT as an iGEM part
- Confirm expression of FT protein in E.coli
2.Secretion
- Modify TorA signal to be easy to use the signal more
- Construct Tat secretion cassette that contains Constitutive promoter, RBS, TatABCD, pspA, double terminator
- Standardize kil gene
- Multiply TatABC in order to strengthen Tat secretion system--not yet
3.Penetration
- Keep GFP in or around plant cells using R9 peptide
- Introduce FT in plant cells using R9 peptide--not yet
4.Activation
- Get high quality of RNA
- Amplify genes successfully
- Check the function of FT--not yet
Future Works
We noticed only flowering and florigen in this time but there are many other plant hormones. We made translocation pathway from E.coli into plant cells, so we will be able to introduce plant hormones into plant cells if E.coli can make them. It means we can control plant growth in any stage through genetically engineered E.coli. In the future that is not so far, we will be able to meddle in plants' growth――germinating, elongation, flowering, and fructification. We human will finally accomplish a technology that control plants perfectly.
Moreover, R9 peptide functions not only plant cell. R9 peptide works on animal cell similarly. It means that we found a pathway into any kinds of cells. R9 peptide tag enables us to introduce proteins into any cells, so we will be able to control all living cells using this technology.
Biosafety
We cooperated with KAIT-Japan and the mark on the left indicates Biosafety Level of our parts.
[1][http://www.ncbi.nlm.nih.gov/pubmed/15695452 Microsugar Chang et al.(2005) "Cellular internalization of fluorescent proteins via arginine-rich intracellular delivery peptide in plant cells" Plant Cell Physiol, 46(3), 482–488]
[2][http://www.ncbi.nlm.nih.gov/pubmed/16155177 Paula Teper-Bamnolker and Alon Samach1.(2005) "The flowering integrator FT regulates SEPALLATA3 and FRUITFULL accumulation in Arabidopsis leaves" The Plant Cell, 17, 2661–2675]
[3][http://www.ncbi.nlm.nih.gov/pubmed/16099980 Philip A. Wigge et al.(2005) "Integration of spatial and temporal information during floral induction in Arabidopsis" Science, 309(5737), 1056-1059]
[4][http://www.mdpi.com/1424-8247/3/4/961/htm Sara Trabulo et al.(2010) "Cell-penetrating peptides—mechanisms of cellular uptake and generation of delivery systems" Pharmaceuticals, 3, 961-993]
[5][http://www.ncbi.nlm.nih.gov/pubmed/15147914 Unnamalai N, Kang BG, Lee.(2004) "Cationic oligopeptide-mediated delivery of dsRNA for post-transcriptional gene silencing in plant cells" FEBS Lett 21, 566(1-3), 307-10]
[6][http://www.ncbi.nlm.nih.gov/pubmed/22683878 Tracy Palmer and Ben C. Berks.(2012) "The twin-arginine translocation (Tat) protein export pathway" Nat Rev Microbiol, 10(7), 483-96]
[7][http://www.ncbi.nlm.nih.gov/pubmed/14966662 Choi JH, Lee SY.(2004) "Secretory and extracellular production of recombinant proteins using Escherichia coli" Appl Microbiol Biotechnol, 64(5), 625-35]
[8][http://www.ncbi.nlm.nih.gov/pubmed/9042754 Miksch G, Fiedler E, Dobrowolski P, Friehs K.(1997) "The kil gene of the ColE1 plasmid of Escherichia coli controlled by a growth-phase-dependent promoter mediates the secretion of a heterologous periplasmic protein during the stationary phase" Arch Microbiol, 167(2-3), 143-50]
[9][http://www.ncbi.nlm.nih.gov/pubmed/11854367 Seibel BA, Walsh PJ.(2002) "Trimethylamine oxide accumulation in marine animals: relationship to acylglycerol storage" J Exp Biol, 205(Pt 3), 297-306]
[10][http://www.ncbi.nlm.nih.gov/pubmed/11123687 Thomas JD, Daniel RA, Errington J, Robinson C.(2001) "Export of active green fluorescent protein to the periplasm by the twin-arginine translocase (Tat) pathway in Escherichia coli" Mol Microbiol, 39(1), 47-53]
[11][http://www.ncbi.nlm.nih.gov/pubmed/3139642 Suit JL, Luria SE.(1988) "Expression of the kil gene of the ColE1 plasmid in Escherichia coli Kilr mutants causes release of periplasmic enzymes and of colicin without cell death" J Bacteriol, 170(10), 4963-4966]
[12][http://www.ncbi.nlm.nih.gov/pubmed/14702305 DeLisa MP, Lee P, Palmer T, Georgiou G.(2004) "Phage shock protein PspA of Escherichia coli relieves saturation of protein export via the Tat pathway" J Bacteriol, 186(2), 366-373]
[13][http://www.ncbi.nlm.nih.gov/pubmed/16099979 Araki, T et al.(2005) “FD, a bZIP protein mediating signals from the floral pathway integrator FT at the shoot apex” Science 309(5737), 1052–1056]
 "
"