Team:MIT/Results
From 2012.igem.org
| Line 322: | Line 322: | ||
<br> We first implemented the NOT GATE in vitro using DNA instead of RNA strands. | <br> We first implemented the NOT GATE in vitro using DNA instead of RNA strands. | ||
| - | The design of this gate is in figure 1, where a letter with a '*' depicts a complementary domain to the one denoted by the letter alone. | + | The design of this gate is in figure 1, where a letter with a '*' depicts a complementary domain to the one denoted by the letter alone. We arrived to this design after having conceived other 5, trying each time to reduce the number of molecules involved or their complexity. |
| - | To understand the behavior of this NOT GATE it can be useful to consider two extreme cases, that is, no input present and input at high concentration. | + | <br>To understand the behavior of this NOT GATE it can be useful to consider two extreme cases, that is, no input present and input at high concentration. |
When the input is not present the B molecules can bind reversibly with A and reversibly with C. When B start to displace c2 from C, the D molecules will free B that consequently will be able to displace c2 from other C molecules. Finally c2 will irreversibly displace e2 from the readout. Therefore we will see high fluorescence (that is, high level of output with no input) | When the input is not present the B molecules can bind reversibly with A and reversibly with C. When B start to displace c2 from C, the D molecules will free B that consequently will be able to displace c2 from other C molecules. Finally c2 will irreversibly displace e2 from the readout. Therefore we will see high fluorescence (that is, high level of output with no input) | ||
When the input is present in high concentration, B and the input bind irreversibly with A due to the mechanism of the cooperative hybridization( | When the input is present in high concentration, B and the input bind irreversibly with A due to the mechanism of the cooperative hybridization( | ||
Revision as of 15:33, 30 September 2012
RNA Strand Displacement In Vitro
Previously:
In 2011, Lulu Qian and Erik Winfree, researchers at Caltech, published a paper entitled "Scaling Up Digital Circuit Computation with DNA Strand Displacement Cascades." This paper demonstrated how scalable logic circuits based on DNA strand displacement are capable of processes as complicated as the square root function. See our Motivation page (2012.igem.org/Team:MIT/Motivation) for more details.
MIT iGEM 2012:
Before our team attempted to bring the mechanism of strand displacement into an in vivo context, we first decided to assay strand displacement in vitro using RNA. We used 2'-O-methylated RNA strands, which had not been shown to undergo strand displacement in vitro. Before creating our own constructs, we adapted sequences from the Qian/Winfree paper to RNA.
MIT iGEM Foundational Experiment:
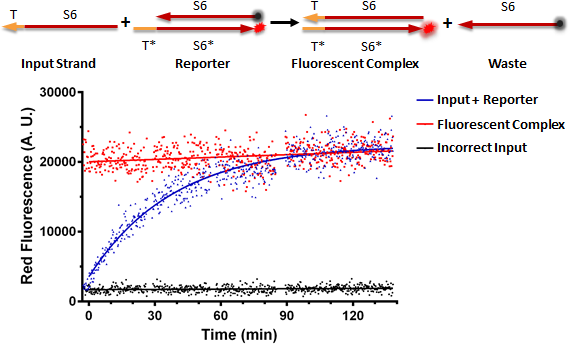
Figure A shows a foundational in vitro RNA strand displacement experiment that was performed on a plate reader. The negative control, in black, is a well that received only an annealed reporter complex. The bottom strand of this complex is the gate strand, T*-S6*, with the 3' end tagged with the ROX fluorophore. The top strand of the complex, the output strand, S6 which is complementary to S6*, is tagged with a quencher, that absorbs fluorescence from the fluorophore. When the two strands of the reporter are annealed, no fluorescence should be observed. The positive control, in red, is the input strand, T-S6, annealed to the gate strand, T*-S6* tagged with ROX. This is what we would expect the product of a strand displacement reaction to behave like. We can see that in the experimental well when the input is present, it can bind to the exposed T* domain of the reporter and displace the output, S6-quencher, strand yielding a fluorescent complex and waste.
Figure A
Nucleic Acid Delivery
To be able to achieve RNA strand displacement logic cascades in vivo, first our team had to ensure that we are capable to delivering nucleic acid to Mammalian Cells. We have achieved the delivery of plasmid DNA, single stranded modified RNA and double stranded modified RNA to mammalian cells through both lipofection and nucleofection.
(1) Delivery of Plasmid DNA to Mammalian Cells
Through the Gateway method, we have assembled many promoter-gene constructs as detailed on our Parts Page. After construction, we deliver the plasmid DNA to Mammalian Cells through the use of transient transfection, lipofection with Lipofectamine 2000 reagent. Figure A shows:
Figure A will go here.
(2) Delivery of 2'-O-Me RNA to Mammalian Cells
Since our modified reporter constructs use 2'-O-Methyl RNA, we must be able to deliver 2'-O-Me RNA into Mammalian Cells.
The movie below shows HEK293 cells expressing constitutive eYFP with a 2'-O-Methyl-RNA strand labeled with ROX (5-carboxy-x-rhodamine) on the 3' end. As time passes, the complex/vesicles are uptaken by the cell, releasing their payload resulting in whole cell fluorescence. Each frame is 5 minutes, movie encompasses 200 minutes in 9 seconds.
Figure B shows time point images taken at t = 0, 2, 3, and 4 hours post-transfection. Images taken at 10X on Zeiss microscope
Figure B

Once we demonstrated ability to deliver 2'-O-Me RNA to mammalian cells, we ran optimization experiments to optimize the ratio of RNA delivered to transfection reagent used. 2'-O-Me RNA to transfection reagent.
15,20,25,30 pmol ratio DATA from FACS .
(3) Inducible Control of Protein Expression
Figure C

Figure D

Delivery of Plasmid DNA which transcribes short RNA Inputs
FF1 Knockdown Data with triplicate data from nathan
In Vivo RNA Strand Displacement
Strategy 1: Lipofectamine 2000 Transfection of RNA version of Reporter from Winfree/QIan 2011 Paper

Images will go here from April 24th experiment - display red fluorescence in all wells, including those that only got reporter or the wrong input - also see red vesicles indicating reporter comes apart inside the vesicles
Strategy 2: Switch Transfection reagent to RNAiMAX
RNAiMAX is supposed to be a better transfection reagent for double stranded RNA
Images will go here from experiment from June 13th onward where we do not see red vesicles, however we still see whole cell red fluorescence
Strategy 3: Tag RNA strand with an Alexa Fluorophore to act as a transfection marker
Strategy 4: Create DNA plasmids driving transcription of RNA inputs, while transfecting RNA Reporter
Strategy 4: Nucleofect RNA reporter, RNA inputs
[Strategy 5]: Redesign RNA Reporter
Not Gate in vitro

Fig1
One of the possible application of the in vivo RNA strand displacement is to sense high and low concentration of specific biomarkers to distinguish, for instance, healthy cells from cancerous cells.
The sensing part of our circuit will translate biomarkers in the form of mRNA in short strand, non coding, RNA. These short strands will be the input of the processing part of our circuit.
To perform correctly the needed information processing we need to 'transform' specific low signals (that is, low concentration of short strands non coding RNA) in high signals that then can be processed by a downstream AND gate. This transformation can be obtained by a NOT gate, where the input and output are the abovementioned short strands RNA.
We first implemented the NOT GATE in vitro using DNA instead of RNA strands. The design of this gate is in figure 1, where a letter with a '*' depicts a complementary domain to the one denoted by the letter alone. We arrived to this design after having conceived other 5, trying each time to reduce the number of molecules involved or their complexity.
To understand the behavior of this NOT GATE it can be useful to consider two extreme cases, that is, no input present and input at high concentration. When the input is not present the B molecules can bind reversibly with A and reversibly with C. When B start to displace c2 from C, the D molecules will free B that consequently will be able to displace c2 from other C molecules. Finally c2 will irreversibly displace e2 from the readout. Therefore we will see high fluorescence (that is, high level of output with no input) When the input is present in high concentration, B and the input bind irreversibly with A due to the mechanism of the cooperative hybridization( Cooperative Hybridization of Oligonucleotides,David Yu Zhang,JACS 2011) , therefore B cannot displace anymore c2 from C. Consequently e2 cannot be displaced from the readout. Therefore we will see no fluorescence (that is, no output with high level of input). In Figure2 the experimental result for the in vitro NOT GATE

Fig 2
The relative concentration of A with respect of input and B is extremely important. Indeed if the concentration of A is too low the cooperative hybridization between A , B and a high concentration of input can be slow, consequently B can displace c2 from C, that is, we would have a high level of output although the input level is high. On the other hand if the concentration of A is too high, even without the presence of input, B will continuously reversibly bind with A. Consequently B will not displace c2 from C and therefore we would not see a high level of output when the level of input is low.
In addition to the relative concentration of the different components another important point is the absolute concentration of them. This is mainly due to how the cooperative hybridization works. Indeed the reactants are three and the products two, consequently at low concentration the reactants are more favorable in the reaction whereas at high concentration the products will be more favorable.
Our strategy consisted first in finding the right concentration to let the cooperative hybridization works and then we tuned the concentration of A to find the right trade of between the interaction of A, input and B when the input is high and the interaction of A and B when the input is low.
 "
"

