Team:Kyoto/Secretion/Notebook
From 2012.igem.org
(→PCR) |
|||
| Line 4: | Line 4: | ||
<div class="_kyoto-note"> | <div class="_kyoto-note"> | ||
==February 7== | ==February 7== | ||
| - | + | ====Preculture==== <small>by_???</small><br> | |
We started preculture at 12:10.<br> | We started preculture at 12:10.<br> | ||
| Line 28: | Line 28: | ||
==March 4== | ==March 4== | ||
| - | + | ====Sequence of tatABCD==== | |
{| class="wikitable" style="text-align: right;" | {| class="wikitable" style="text-align: right;" | ||
!Quick Taq||primer-f||promer-r sequence||template||MilliQ||total | !Quick Taq||primer-f||promer-r sequence||template||MilliQ||total | ||
| Line 39: | Line 39: | ||
|25||1||1||1||23||50 | |25||1||1||1||23||50 | ||
|} | |} | ||
| - | + | ====Colony PCR of TMAO==== | |
{| class="wikitable" style="text-align: right;" | {| class="wikitable" style="text-align: right;" | ||
!buffer||dNTPs||NgSO4||primer-f||primerr-r||KOD plus||MilliQ||total | !buffer||dNTPs||NgSO4||primer-f||primerr-r||KOD plus||MilliQ||total | ||
| Line 52: | Line 52: | ||
68℃, 2.5min<br> | 68℃, 2.5min<br> | ||
→25cycles<br><br> | →25cycles<br><br> | ||
| - | + | ====Electrophoresis==== | |
1. 1kb ladder<br> | 1. 1kb ladder<br> | ||
2. tatABCD1<br> | 2. tatABCD1<br> | ||
| Line 58: | Line 58: | ||
4. TMAO<br> | 4. TMAO<br> | ||
5. 1kb ladder<br><br> | 5. 1kb ladder<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!TMAO||EcoR1||BufferH||BSA||MilliQ||total | !TMAO||EcoR1||BufferH||BSA||MilliQ||total | ||
| Line 70: | Line 70: | ||
|} | |} | ||
at 37℃ for 1 hour<br><br> | at 37℃ for 1 hour<br><br> | ||
| - | + | ====Electrophoresis====<br> | |
| - | + | ====Transformation==== | |
{|class="wikitable" | {|class="wikitable" | ||
!pSB1C3||competent cell(made at 2/8)||total | !pSB1C3||competent cell(made at 2/8)||total | ||
| Line 79: | Line 79: | ||
==March 5== | ==March 5== | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!pSB1C3(Xba1, Spe1)||Pst1||BufferH||BSA||MilliQ||total | !pSB1C3(Xba1, Spe1)||Pst1||BufferH||BSA||MilliQ||total | ||
| Line 91: | Line 91: | ||
at 37℃ for 1 hour<br> | at 37℃ for 1 hour<br> | ||
→Then we did ethanol precipitation<br><br> | →Then we did ethanol precipitation<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Kil(EcoR1, Spe1)||pSB1C3(EcoR1)||Ligation High||total | !Kil(EcoR1, Spe1)||pSB1C3(EcoR1)||Ligation High||total | ||
| Line 98: | Line 98: | ||
|} | |} | ||
at 16℃ for 1 hour<br><br> | at 16℃ for 1 hour<br><br> | ||
| - | + | ====Transformation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Kil||competent cell||total | !Kil||competent cell||total | ||
| Line 105: | Line 105: | ||
|} | |} | ||
We used commercially available competent cells in this time.<br><br> | We used commercially available competent cells in this time.<br><br> | ||
| - | + | ====PCR==== | |
TMAO<br> | TMAO<br> | ||
{| class="wikitable" | {| class="wikitable" | ||
| Line 112: | Line 112: | ||
|5||5||3||1.5||1.5||1||1||32||50 | |5||5||3||1.5||1.5||1||1||32||50 | ||
|} | |} | ||
| - | + | ====Electrophoresis==== | |
(31)<br><br> | (31)<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
! ||Lacp||pSB3C5||EcoR1||Pst1||BufferH||BSA||MilliQ||total | ! ||Lacp||pSB3C5||EcoR1||Pst1||BufferH||BSA||MilliQ||total | ||
| Line 125: | Line 125: | ||
1→Ethanol precipitation 45.4µg/mL<br> | 1→Ethanol precipitation 45.4µg/mL<br> | ||
2→Gel extraction 38.7µg/mL<br><br> | 2→Gel extraction 38.7µg/mL<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!LacP||pSB3C5||Ligation High||total | !LacP||pSB3C5||Ligation High||total | ||
| Line 132: | Line 132: | ||
|} | |} | ||
at 4℃ for overnight<br><br> | at 4℃ for overnight<br><br> | ||
| - | + | ====Transformation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Lacp+pSB3C5||competent cell||total | !Lacp+pSB3C5||competent cell||total | ||
| Line 142: | Line 142: | ||
on ice for 2 mins.<br> | on ice for 2 mins.<br> | ||
After we incubate with 200µL of SOC culture for 1 hour, we did plating on LB culture with CP<br><br> | After we incubate with 200µL of SOC culture for 1 hour, we did plating on LB culture with CP<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!GFP Plasmid||EcoR1||Spe1||Buffer2||BSA||MilliQ||total | !GFP Plasmid||EcoR1||Spe1||Buffer2||BSA||MilliQ||total | ||
| Line 149: | Line 149: | ||
|} | |} | ||
at 37℃ for 2 hours | at 37℃ for 2 hours | ||
| - | + | ====Electrophoresis==== | |
1. Ladder 2µL<br> | 1. Ladder 2µL<br> | ||
2. GFP Plasmid (already restricted) 2µL + Loading Dye 2µL + MilliQ 8µL<br> | 2. GFP Plasmid (already restricted) 2µL + Loading Dye 2µL + MilliQ 8µL<br> | ||
3. Ladder 2µL<br> | 3. Ladder 2µL<br> | ||
| - | + | ====PCR==== | |
torA signal and pspA | torA signal and pspA | ||
pspAはコロニーPCR<br> | pspAはコロニーPCR<br> | ||
| Line 165: | Line 165: | ||
60℃, 30sec<br> | 60℃, 30sec<br> | ||
68℃, 30sec<br> | 68℃, 30sec<br> | ||
| - | + | ====electrophoresis==== | |
1. 100bp Ladder<br> | 1. 100bp Ladder<br> | ||
2. torA signal (272bp)<br> | 2. torA signal (272bp)<br> | ||
| Line 172: | Line 172: | ||
==March 6== | ==March 6== | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!GFP||EcoR1||Spe1||BufferM||BSA||MilliQ||total | !GFP||EcoR1||Spe1||BufferM||BSA||MilliQ||total | ||
| Line 181: | Line 181: | ||
We did incubate at 37℃ for 1.5hours.<br> | We did incubate at 37℃ for 1.5hours.<br> | ||
And we did gel extraction on 3/7 and get 40.0μg/mL GFP.<br><br> | And we did gel extraction on 3/7 and get 40.0μg/mL GFP.<br><br> | ||
| - | + | ====PCR==== | |
{| class="wikitable" | {| class="wikitable" | ||
!buffer||dNTPs||MgSO4||Primer-f||Primer-r||template||KOD plus neo||MilliQ||total | !buffer||dNTPs||MgSO4||Primer-f||Primer-r||template||KOD plus neo||MilliQ||total | ||
| Line 192: | Line 192: | ||
68℃ (torA 10sec / pspA 30sec) 25 cycles<br> | 68℃ (torA 10sec / pspA 30sec) 25 cycles<br> | ||
We used product of PCR on 3/5 of torA and pspA as template.<br><br> | We used product of PCR on 3/5 of torA and pspA as template.<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Kil||EcoR1||Spe1||BufferM||BSA||MilliQ||total | !Kil||EcoR1||Spe1||BufferM||BSA||MilliQ||total | ||
| Line 205: | Line 205: | ||
at 37℃ for 2 hours<br> | at 37℃ for 2 hours<br> | ||
→ purification 37.7ng/μL<br><br> | → purification 37.7ng/μL<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Kil||pSB1C3||Ligation High Ver.2||total | !Kil||pSB1C3||Ligation High Ver.2||total | ||
| Line 216: | Line 216: | ||
==March 7== | ==March 7== | ||
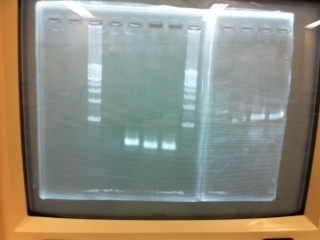
| - | + | ====Electrophoresis====[[File:Electrophoresis0307.JPG|400px|thumb|right]]<br> | |
1. 1kb ladder 2µL<br> | 1. 1kb ladder 2µL<br> | ||
2. pspA (PCR product)2.5µL + Loading Dye 0.5µL<br> | 2. pspA (PCR product)2.5µL + Loading Dye 0.5µL<br> | ||
| Line 222: | Line 222: | ||
4. 1kb ladder 2µL<br> | 4. 1kb ladder 2µL<br> | ||
*The GFP was gel extracted on 3/6 and concentrated by Vacuum in 150µg/mL<br><br> | *The GFP was gel extracted on 3/6 and concentrated by Vacuum in 150µg/mL<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!VectorDNA||GFP||Ligation High Ver.2||total | !VectorDNA||GFP||Ligation High Ver.2||total | ||
| Line 228: | Line 228: | ||
|5||15||10||30 | |5||15||10||30 | ||
|} | |} | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!torA||EcoR1||Spe1||bufferM||BSA||MilliQ||total | !torA||EcoR1||Spe1||bufferM||BSA||MilliQ||total | ||
| Line 240: | Line 240: | ||
|} | |} | ||
at 37℃ for 1.5 hours<br><br> | at 37℃ for 1.5 hours<br><br> | ||
| - | + | ====Purification==== | |
torA→31.8ng/µL<br> | torA→31.8ng/µL<br> | ||
pspA→49.3ng/µL<br><br> | pspA→49.3ng/µL<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!torA||pSB1C3||Ligation High Ver.2||total | !torA||pSB1C3||Ligation High Ver.2||total | ||
| Line 261: | Line 261: | ||
==March 8== | ==March 8== | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!pSB4K5||EcoR1||Spe1||BufferM||BSA||MilliQ||total | !pSB4K5||EcoR1||Spe1||BufferM||BSA||MilliQ||total | ||
| Line 270: | Line 270: | ||
at 37℃ for 1 hour.<br> | at 37℃ for 1 hour.<br> | ||
→Purification : 36.6ng/µL<br><br> | →Purification : 36.6ng/µL<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Kil||pSB4K5||Ligation High Ver.2||total | !Kil||pSB4K5||Ligation High Ver.2||total | ||
| Line 279: | Line 279: | ||
*Kil→37.7ng/µL×10µL=377ng=879fmol<br> | *Kil→37.7ng/µL×10µL=377ng=879fmol<br> | ||
*pSB4K5→36.6ng/µL×1µL=36.6ng=86fmol<br><br> | *pSB4K5→36.6ng/µL×1µL=36.6ng=86fmol<br><br> | ||
| - | + | ====Liquid culture==== | |
Lacp + pSB3C5 -1, 2<br><br> | Lacp + pSB3C5 -1, 2<br><br> | ||
| - | + | ====Transformation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!torA||pspA||competent cell||total | !torA||pspA||competent cell||total | ||
| Line 292: | Line 292: | ||
==March 9== | ==March 9== | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!tatABCD||Xba1||Pst1||BufferM||BSA||MilliQ||total | !tatABCD||Xba1||Pst1||BufferM||BSA||MilliQ||total | ||
| Line 300: | Line 300: | ||
at 37℃ for 1hour<br> | at 37℃ for 1hour<br> | ||
→purification : 75.0μg/mL (1.11 260/280 , 0.81 260/230)<br><br> | →purification : 75.0μg/mL (1.11 260/280 , 0.81 260/230)<br><br> | ||
| - | + | ====Miniprep==== | |
lacP + pSB3C5 1 80.5μg/mL (1.78 260/280 , 2.00 260/230)<br> | lacP + pSB3C5 1 80.5μg/mL (1.78 260/280 , 2.00 260/230)<br> | ||
lacP + pSB3C5 2 107.2μg/mL (1.83 260/280 , 1.90 260/230)<br><br> | lacP + pSB3C5 2 107.2μg/mL (1.83 260/280 , 1.90 260/230)<br><br> | ||
| - | + | ====Colony PCR==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Quick Taq||VF||VR||MilliQ||total | !Quick Taq||VF||VR||MilliQ||total | ||
| Line 314: | Line 314: | ||
68℃ 6sec<br> | 68℃ 6sec<br> | ||
25cycles<br><br> | 25cycles<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!tatABCD||constP J23107||Ligation High Ver.2||total | !tatABCD||constP J23107||Ligation High Ver.2||total | ||
| Line 322: | Line 322: | ||
tatABCD : 227fmol<br> | tatABCD : 227fmol<br> | ||
constP J23107 : 21fmol<br><br> | constP J23107 : 21fmol<br><br> | ||
| - | + | ====Transformation==== | |
{| class="wikitable" | {| class="wikitable" | ||
! ||pspA||torA||Kil||competent cell | ! ||pspA||torA||Kil||competent cell | ||
| Line 332: | Line 332: | ||
|3||0||0||1||10 | |3||0||0||1||10 | ||
|} | |} | ||
| - | + | ====Miniprep==== | |
4mL of plusgrow which had been cultured for overnight.<br> | 4mL of plusgrow which had been cultured for overnight.<br> | ||
pSB4K5 : 80.5μg/mL<br> | pSB4K5 : 80.5μg/mL<br> | ||
==March 10== | ==March 10== | ||
| - | + | ====Screening PCR==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Quick Taq||VF||VR||MilliQ||total | !Quick Taq||VF||VR||MilliQ||total | ||
| Line 351: | Line 351: | ||
1, 100bp ladder<br> | 1, 100bp ladder<br> | ||
2,3,4, torA signal<br><br> | 2,3,4, torA signal<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!LacP-pSB3C5||Spe1||Pst1||BufferM||BSA||MilliQ||total | !LacP-pSB3C5||Spe1||Pst1||BufferM||BSA||MilliQ||total | ||
| Line 366: | Line 366: | ||
for 2.5 hours at 37℃<br> | for 2.5 hours at 37℃<br> | ||
→purification 91.8ng/μL<br><br> | →purification 91.8ng/μL<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!torA||Lacp-pSB3C5||Ligation High Ver.2||total | !torA||Lacp-pSB3C5||Ligation High Ver.2||total | ||
| Line 377: | Line 377: | ||
==March 11== | ==March 11== | ||
| - | + | ====Miniprep==== | |
We used 3μL of plus grow that we had cultured for overnight.<br> | We used 3μL of plus grow that we had cultured for overnight.<br> | ||
torA : 62.8ng/μL<br><br> | torA : 62.8ng/μL<br><br> | ||
| - | + | ====Screening PCR==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Quick Taq||VF||VR||MilliQ||total | !Quick Taq||VF||VR||MilliQ||total | ||
| Line 386: | Line 386: | ||
|25||1||1||23||50 | |25||1||1||23||50 | ||
|} | |} | ||
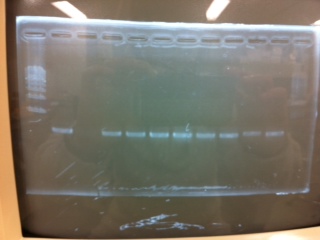
| - | + | ====Electrophoresis==== | |
[[File:Electrophoresis031101.JPG|400px|thumb|right]] | [[File:Electrophoresis031101.JPG|400px|thumb|right]] | ||
1. 1kb ladder<br> | 1. 1kb ladder<br> | ||
| Line 402: | Line 402: | ||
==March 12== | ==March 12== | ||
| - | + | ====Transformation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!DT(1ng/μL)||DT(0.1ng/μL)||Kil||lacP-torA||MilliQ||competent cell||total | !DT(1ng/μL)||DT(0.1ng/μL)||Kil||lacP-torA||MilliQ||competent cell||total | ||
| Line 416: | Line 416: | ||
|0||0||0||0||1||20||21 | |0||0||0||0||1||20||21 | ||
|} | |} | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!pspA||EcoR1||Spe1||BufferM||BSA||MilliQ||total | !pspA||EcoR1||Spe1||BufferM||BSA||MilliQ||total | ||
| Line 424: | Line 424: | ||
at 37℃ for 4 hours<br> | at 37℃ for 4 hours<br> | ||
→ We did purification and got 48.3ng/μL pspA.<br><br> | → We did purification and got 48.3ng/μL pspA.<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
! |pspA||pSB1C3||MilliQ||Ligation High Ver.2||total | ! |pspA||pSB1C3||MilliQ||Ligation High Ver.2||total | ||
| Line 436: | Line 436: | ||
==March 13== | ==March 13== | ||
| - | + | ====Miniprep==== | |
pSB1C3 74.2µg/mL 1.65 (260/280) 1.31 (260/230) | pSB1C3 74.2µg/mL 1.65 (260/280) 1.31 (260/230) | ||
==March 14== | ==March 14== | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!pSB1C3||EcoR1||Spe1||BufferM||BSA||MilliQ||total | !pSB1C3||EcoR1||Spe1||BufferM||BSA||MilliQ||total | ||
| Line 446: | Line 446: | ||
|} | |} | ||
We did gel extraction and got 47.2ng/µL pSB1C3 but we did not cut out RFP.<br><br> | We did gel extraction and got 47.2ng/µL pSB1C3 but we did not cut out RFP.<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!torA||pSB1C3||Ligation High Ver.2||total | !torA||pSB1C3||Ligation High Ver.2||total | ||
| Line 459: | Line 459: | ||
*torA : 0.707pmol<br> | *torA : 0.707pmol<br> | ||
*pSB1C3 : 0.068pmol<br><br> | *pSB1C3 : 0.068pmol<br><br> | ||
| - | + | ====Liquid culture==== | |
We cultured Lacp-pSB3C5 1,2,3,4,5 (CP tolerance)on culture with Amp.<br> | We cultured Lacp-pSB3C5 1,2,3,4,5 (CP tolerance)on culture with Amp.<br> | ||
→Only 4 which did not be cultured succeeded. | →Only 4 which did not be cultured succeeded. | ||
==March 15== | ==March 15== | ||
| - | + | ====Liquid culture==== | |
We cultured Lacp-pSB3C5 6,7,8,9,10 on culture with ampicillin.<br> | We cultured Lacp-pSB3C5 6,7,8,9,10 on culture with ampicillin.<br> | ||
→6,8,9,10 were succeeded.<br><br> | →6,8,9,10 were succeeded.<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!GFP||EcoR1||Spe1||Buffer2||BSA||MilliQ||total | !GFP||EcoR1||Spe1||Buffer2||BSA||MilliQ||total | ||
| Line 479: | Line 479: | ||
|} | |} | ||
for 2 hours at 37℃.<br><br> | for 2 hours at 37℃.<br><br> | ||
| - | + | ====Miniprep==== | |
pspA (pSB1C3) 40.5ng/µL<br><br> | pspA (pSB1C3) 40.5ng/µL<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!pspA||DT||Ligation High Ver.2||total | !pspA||DT||Ligation High Ver.2||total | ||
| Line 489: | Line 489: | ||
*pspA : 385fmol<br> | *pspA : 385fmol<br> | ||
*DT : 36fmol<br><br> | *DT : 36fmol<br><br> | ||
| - | + | ====Transformation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!J23107-tatABCD||DT (0.1ng/µL)||DT (0.01ng/µL)||pspA-DT||competent cells on 3/15||total | !J23107-tatABCD||DT (0.1ng/µL)||DT (0.01ng/µL)||pspA-DT||competent cells on 3/15||total | ||
| Line 501: | Line 501: | ||
|0||0||0||2||20||22 | |0||0||0||2||20||22 | ||
|} | |} | ||
| - | + | ====Screening PCR==== | |
Kil, pspA and torA<br> | Kil, pspA and torA<br> | ||
{| class="wikitable" | {| class="wikitable" | ||
| Line 510: | Line 510: | ||
==March 16== | ==March 16== | ||
| - | + | ====Miniprep==== | |
torA (pSB1C3) 68.8ng/µL<br> | torA (pSB1C3) 68.8ng/µL<br> | ||
Kil (pSB4K5) 92.7ng/µL<br> | Kil (pSB4K5) 92.7ng/µL<br> | ||
torA was red for some reason. We do not know why.<br><br> | torA was red for some reason. We do not know why.<br><br> | ||
| - | + | ====Colony PCR==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Quick Taq||Primer-r||Primer-f||MilliQ||total | !Quick Taq||Primer-r||Primer-f||MilliQ||total | ||
| Line 525: | Line 525: | ||
68℃, 6sec<br> | 68℃, 6sec<br> | ||
→25cycles<br><br> | →25cycles<br><br> | ||
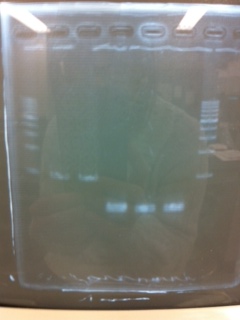
| - | + | ====Electrophoresis==== | |
[[File:Electrophoresis0316.JPG|400px|thumb|right]] | [[File:Electrophoresis0316.JPG|400px|thumb|right]] | ||
The results were shown as photograph in the right.<br><br> | The results were shown as photograph in the right.<br><br> | ||
| - | + | ====Checking Transformation Efficiency==== | |
competent cells that were made on March 15.<br> | competent cells that were made on March 15.<br> | ||
DNA : 0.02ng → 668 colonies Transformation Efficiency : 3.3×10^7<br> | DNA : 0.02ng → 668 colonies Transformation Efficiency : 3.3×10^7<br> | ||
DNA : 0.2ng → 1739 colonies Transformation Efficiency : 8.7×10^6<br><br> | DNA : 0.2ng → 1739 colonies Transformation Efficiency : 8.7×10^6<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!pSB1C3||EcoR1||Spe1||BSA||BufferM||BufferH||MilliQ||total | !pSB1C3||EcoR1||Spe1||BSA||BufferM||BufferH||MilliQ||total | ||
| Line 560: | Line 560: | ||
for overnight at 37℃.<br> | for overnight at 37℃.<br> | ||
We did this to confirm.<br><br> | We did this to confirm.<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!torA||pSB1C3||Ligation High Ver.2||total | !torA||pSB1C3||Ligation High Ver.2||total | ||
| Line 576: | Line 576: | ||
==March 17== | ==March 17== | ||
| - | + | ====Miniprep==== | |
J23107-tatABCD 72.7ng/µL<br> | J23107-tatABCD 72.7ng/µL<br> | ||
pspA-DT 50.5ng/µL<br><br> | pspA-DT 50.5ng/µL<br><br> | ||
| - | + | ====Checking the Insert==== | |
{| class="wikitable" | {| class="wikitable" | ||
!J21037-tatABCD||EcoR1||Pst1||BSA||BufferH||MilliQ||total | !J21037-tatABCD||EcoR1||Pst1||BSA||BufferH||MilliQ||total | ||
| Line 600: | Line 600: | ||
==March 19== | ==March 19== | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!DT||EcoR1||Xba1||BSA||BufferM||MilliQ||total | !DT||EcoR1||Xba1||BSA||BufferM||MilliQ||total | ||
| Line 607: | Line 607: | ||
|} | |} | ||
We did Gel extraction and got 17.2ng/µL of DT.<br><br> | We did Gel extraction and got 17.2ng/µL of DT.<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Kil||DT||Ligation High Ver.2||total | !Kil||DT||Ligation High Ver.2||total | ||
| Line 614: | Line 614: | ||
|} | |} | ||
We did this for an hour at 16℃.<br><br> | We did this for an hour at 16℃.<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!GFP||EcoR1||Spe1||BSA||BufferM||MilliQ||total | !GFP||EcoR1||Spe1||BSA||BufferM||MilliQ||total | ||
| Line 621: | Line 621: | ||
|} | |} | ||
We did this for 4 hours at 37℃<br><br> | We did this for 4 hours at 37℃<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!pspA||DT||Ligation High Ver.2||total | !pspA||DT||Ligation High Ver.2||total | ||
| Line 637: | Line 637: | ||
*pspA (4µL)→339fmol<br> | *pspA (4µL)→339fmol<br> | ||
*pSB1C3→34fmol<br><br> | *pSB1C3→34fmol<br><br> | ||
| - | + | ====Transformation==== | |
pspA-DT, pspA (pSB1C3), torA (pSB1C3) and GFP-DT | pspA-DT, pspA (pSB1C3), torA (pSB1C3) and GFP-DT | ||
==March 20== | ==March 20== | ||
| - | + | ====Screaning PCR==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Quick Taq||Primer-R||Primer-F||MilliQ||total | !Quick Taq||Primer-R||Primer-F||MilliQ||total | ||
| Line 659: | Line 659: | ||
*pspA→○<br> | *pspA→○<br> | ||
*pspA-DT→×<br><br> | *pspA-DT→×<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!torA||EcoR1||Spe1||BSA||BufferM||MilliQ||total | !torA||EcoR1||Spe1||BSA||BufferM||MilliQ||total | ||
| Line 675: | Line 675: | ||
==March 21== | ==March 21== | ||
| - | + | ====Miniprep==== | |
GFP-DT-1 : 77.7ng/µL<br> | GFP-DT-1 : 77.7ng/µL<br> | ||
GFP-DT-2 : 67.6ng/µL<br><br> | GFP-DT-2 : 67.6ng/µL<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!pSB1C3||EcoR1||Spe1||BSA||BufferM||MilliQ||total | !pSB1C3||EcoR1||Spe1||BSA||BufferM||MilliQ||total | ||
| Line 697: | Line 697: | ||
|} | |} | ||
We did this for 3 hours at 37℃, and then we did Purification. We got 30.7ng/µL GFP-DT.<br><br> | We did this for 3 hours at 37℃, and then we did Purification. We got 30.7ng/µL GFP-DT.<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
! ||torA||pSB1C3||pspA||DT||GFP-DT||Ligation High Ver.2||total | ! ||torA||pSB1C3||pspA||DT||GFP-DT||Ligation High Ver.2||total | ||
| Line 712: | Line 712: | ||
==March 22== | ==March 22== | ||
| - | + | ====PCR==== | |
We did PCR to amplify torA_signal that was product of PCR at March 5 with redesigned primers.<br> | We did PCR to amplify torA_signal that was product of PCR at March 5 with redesigned primers.<br> | ||
{| class="wikitable" | {| class="wikitable" | ||
| Line 725: | Line 725: | ||
→30cycles<br> | →30cycles<br> | ||
→Purification 110.7ng/µL<br><br> | →Purification 110.7ng/µL<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!torA||EcoR1||Spe1||BSA||BufferM||MilliQ||total | !torA||EcoR1||Spe1||BSA||BufferM||MilliQ||total | ||
| Line 731: | Line 731: | ||
|10||0.2||0.2||0.3||3||16.3||30 | |10||0.2||0.2||0.3||3||16.3||30 | ||
|} | |} | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
! ||torA||pSSB1C3||pspA||DT||GFP-DT||Ligation high||total | ! ||torA||pSSB1C3||pspA||DT||GFP-DT||Ligation high||total | ||
| Line 749: | Line 749: | ||
==March 23== | ==March 23== | ||
| - | + | ====Screening PCR==== | |
torA (pSB1C3), torA-GFP-DT and pspA-DT<br> | torA (pSB1C3), torA-GFP-DT and pspA-DT<br> | ||
{| class="wikitable" | {| class="wikitable" | ||
| Line 757: | Line 757: | ||
|} | |} | ||
E.coli in liquid culture that had pspA(pSB1C3) also expressed RFP.<br><br> | E.coli in liquid culture that had pspA(pSB1C3) also expressed RFP.<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!pspA||EcoR1||Spe1||BufferM||BSA||MilliQ||total | !pspA||EcoR1||Spe1||BufferM||BSA||MilliQ||total | ||
| Line 764: | Line 764: | ||
|} | |} | ||
And then we did ethanol precipitation<br><br> | And then we did ethanol precipitation<br><br> | ||
| - | + | ====Ethanol precipitation==== | |
pspA 11.5ng/µL.<br><br> | pspA 11.5ng/µL.<br><br> | ||
| - | + | ====Miniprep==== | |
Lacp+pSB3C5-8 77.9µg/mL<br> | Lacp+pSB3C5-8 77.9µg/mL<br> | ||
Lacp+pSB3C5-10 69.0µg/mL<br> | Lacp+pSB3C5-10 69.0µg/mL<br> | ||
Kil+DT-4 58.0µg/mL<br> | Kil+DT-4 58.0µg/mL<br> | ||
Kil+DT-7 15.2µg/mL<br><br> | Kil+DT-7 15.2µg/mL<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Lacp+pSB3C5-8||Spe1||Pst1||Buffer2||BSA||MilliQ||total | !Lacp+pSB3C5-8||Spe1||Pst1||Buffer2||BSA||MilliQ||total | ||
| Line 784: | Line 784: | ||
at 37℃ for 1.5 hours<br> | at 37℃ for 1.5 hours<br> | ||
And then we did Gel extraction.<br><br> | And then we did Gel extraction.<br><br> | ||
| - | + | ====Gel Extraction==== | |
Lacp+pSB3C5-8 40.4µg/mL<br> | Lacp+pSB3C5-8 40.4µg/mL<br> | ||
Kil+DT-4 26.8µg/mL<br> | Kil+DT-4 26.8µg/mL<br> | ||
==March 26== | ==March 26== | ||
| - | + | ====Miniprep==== | |
torA(pSB1C3) 18.5[ng/µL]<br> | torA(pSB1C3) 18.5[ng/µL]<br> | ||
torA-GFP-DT 20.0[ng/µL]<br><br> | torA-GFP-DT 20.0[ng/µL]<br><br> | ||
| - | + | ====Restriction==== | |
{| class="wikitable" | {| class="wikitable" | ||
!torA(pSB1C3)||EcoR1||Pst1||BufferH||BSA||MilliQ||total | !torA(pSB1C3)||EcoR1||Pst1||BufferH||BSA||MilliQ||total | ||
| Line 810: | Line 810: | ||
|} | |} | ||
We did Gel extraction and then got ??? 28.7[ng/µL]<br><br> | We did Gel extraction and then got ??? 28.7[ng/µL]<br><br> | ||
| - | + | ====Ligation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Lacp (pSB3C5)||torA-GFP-DT||Ligation High Ver.2||total | !Lacp (pSB3C5)||torA-GFP-DT||Ligation High Ver.2||total | ||
| Line 837: | Line 837: | ||
|} | |} | ||
for 2 hours at 16℃<br><br> | for 2 hours at 16℃<br><br> | ||
| - | + | ====Transformation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Lacp-Kil-DT||competent cell||total | !Lacp-Kil-DT||competent cell||total | ||
| Line 845: | Line 845: | ||
==March 27== | ==March 27== | ||
| - | + | ====Miniprep==== | |
We retryed miniprep of torA(pSB1C3).<br> | We retryed miniprep of torA(pSB1C3).<br> | ||
We got torA and its concentration was 39.3[ng/µL].<br><br> | We got torA and its concentration was 39.3[ng/µL].<br><br> | ||
| - | + | ====Transformation==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Name||Well||Sample||Competent Cells||Total||Plate||Colony | !Name||Well||Sample||Competent Cells||Total||Plate||Colony | ||
| Line 855: | Line 855: | ||
|} | |} | ||
We added 100[µL] of culture medium before we started culturing the E.coli.<br><br> | We added 100[µL] of culture medium before we started culturing the E.coli.<br><br> | ||
| - | + | ====Screening PCR==== | |
{| class="wikitable" | {| class="wikitable" | ||
!Quick Taq||VF2||VR||MilliQ||Total | !Quick Taq||VF2||VR||MilliQ||Total | ||
| Line 865: | Line 865: | ||
E.coli that had pspA(pSB1C3) did not make any colony.<br> | E.coli that had pspA(pSB1C3) did not make any colony.<br> | ||
Lacp-Kil-DT 1,2,3,4,5,6,7,8 was failed.<br><br> | Lacp-Kil-DT 1,2,3,4,5,6,7,8 was failed.<br><br> | ||
| - | + | ====Liquid culture==== | |
Lacp-torA-GFP-DT<br> | Lacp-torA-GFP-DT<br> | ||
</div> | </div> | ||
{{Kyoto/footer}} | {{Kyoto/footer}} | ||
Revision as of 16:31, 9 September 2012
Secretion Notebook
February 7
====Preculture==== by_???
We started preculture at 12:10.
February 8
March 1
March 2
March 3
PCR
| template | buffer | dNTPs | MgSO4 | VF | VR | KOD plus | MilliQ | total | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 1 | 5 | 5 | 3 | 1.5 | 1.5 | 1 | 32 | 50 |
| 2 | 2 | 5 | 5 | 3 | 1.5 | 1.5 | 1 | 31 | 50 |
94℃, 2min
98℃, 10sec
50℃, 30sec
68℃, 1min
→30cycles
Miniprep
March 4
Sequence of tatABCD
| Quick Taq | primer-f | promer-r sequence | template | MilliQ | total |
|---|---|---|---|---|---|
| 25 | 1 | 1 | 1 | 23 | 50 |
| Quick Taq | primer-f sequence | primer-r | template | MilliQ | total |
|---|---|---|---|---|---|
| 25 | 1 | 1 | 1 | 23 | 50 |
Colony PCR of TMAO
| buffer | dNTPs | NgSO4 | primer-f | primerr-r | KOD plus | MilliQ | total |
|---|---|---|---|---|---|---|---|
| 5 | 5 | 4 | 1.5 | 1.5 | 1 | 32 | 50 |
→ethanol precipitation
94℃, 2min
94℃, 30sec
55℃, 30sec
68℃, 2.5min
→25cycles
Electrophoresis
1. 1kb ladder
2. tatABCD1
3. tatABCD2
4. TMAO
5. 1kb ladder
Restriction
| TMAO | EcoR1 | BufferH | BSA | MilliQ | total |
|---|---|---|---|---|---|
| 10 | 0.2 | 3 | 0.3 | 16.5 | 30 |
| TMAO | Xba1 | Pst1 | BudderM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.2 | 0.2 | 3 | 0.3 | 16.3 | 30 |
at 37℃ for 1 hour
====Electrophoresis====
Transformation
| pSB1C3 | competent cell(made at 2/8) | total |
|---|---|---|
| 5 | 100 | 105 |
March 5
Restriction
| pSB1C3(Xba1, Spe1) | Pst1 | BufferH | BSA | MilliQ | total |
|---|---|---|---|---|---|
| 10 | 0.2 | 2 | 0.2 | 7.6 | 20 |
| pSB1C3(Xba1, Spe1) | EcoR1 | BufferH | BSA | MilliQ | total |
| 10 | 0.2 | 2 | 0.2 | 7.6 | 20 |
at 37℃ for 1 hour
→Then we did ethanol precipitation
Ligation
| Kil(EcoR1, Spe1) | pSB1C3(EcoR1) | Ligation High | total |
|---|---|---|---|
| 5 | 1 | 3 | 9 |
at 16℃ for 1 hour
Transformation
| Kil | competent cell | total |
|---|---|---|
| 1 | 10 | 11 |
We used commercially available competent cells in this time.
PCR
TMAO
| buffer | dNTPs | MgSO4 | Primer-f | Primer-r | Template | KOD plus | MilliQ | total |
|---|---|---|---|---|---|---|---|---|
| 5 | 5 | 3 | 1.5 | 1.5 | 1 | 1 | 32 | 50 |
Electrophoresis
(31)
Restriction
| Lacp | pSB3C5 | EcoR1 | Pst1 | BufferH | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|---|
| 1 | 20 | 0 | 0.5 | 0.5 | 3 | 0.5 | 5.5 | 30 |
| 2 | 0 | 20 | 0.5 | 0.5 | 3 | 0.5 | 5.5 | 30 |
at 37℃ for 1 hour
1→Ethanol precipitation 45.4µg/mL
2→Gel extraction 38.7µg/mL
Ligation
| LacP | pSB3C5 | Ligation High | total |
|---|---|---|---|
| 10 | 2 | 6 | 18 |
at 4℃ for overnight
Transformation
| Lacp+pSB3C5 | competent cell | total |
|---|---|---|
| 1 | 10 | 11 |
on ice for 30 mins.
heat shock at 42℃ for 60secs
on ice for 2 mins.
After we incubate with 200µL of SOC culture for 1 hour, we did plating on LB culture with CP
Restriction
| GFP Plasmid | EcoR1 | Spe1 | Buffer2 | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.5 | 0.5 | 3 | 0.5 | 15.5 | 30 |
at 37℃ for 2 hours
Electrophoresis
1. Ladder 2µL
2. GFP Plasmid (already restricted) 2µL + Loading Dye 2µL + MilliQ 8µL
3. Ladder 2µL
PCR
torA signal and pspA
pspAはコロニーPCR
| Buffer | dNTPs | MgSO4 | Primer-f | Primer-r | template(TMAO) | KOD plus | MilliQ | total |
|---|---|---|---|---|---|---|---|---|
| 2.5 | 2.5 | 1.5 | 0.75 | 0.75 | 0.5 | 0.5 | 16 | 25 |
94℃, 2min
98℃, 10sec
60℃, 30sec
68℃, 30sec
electrophoresis
1. 100bp Ladder
2. torA signal (272bp)
3. pspA (969bp)
4. 100bp Ladder
March 6
Restriction
| GFP | EcoR1 | Spe1 | BufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 12 | 0.5 | 0.5 | 3 | 0.5 | 13.5 | 30 |
We did incubate at 37℃ for 1.5hours.
And we did gel extraction on 3/7 and get 40.0μg/mL GFP.
PCR
| buffer | dNTPs | MgSO4 | Primer-f | Primer-r | template | KOD plus neo | MilliQ | total |
|---|---|---|---|---|---|---|---|---|
| 5 | 5 | 3 | 1.5 | 1.5 | 0.5 | 1 | 32.5 | 50 |
94℃ 2min
98℃ 10sec
60℃ 30sec
68℃ (torA 10sec / pspA 30sec) 25 cycles
We used product of PCR on 3/5 of torA and pspA as template.
Restriction
| Kil | EcoR1 | Spe1 | BufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.3 | 0.3 | 3 | 0.3 | 16.1 | 30 |
| DT | EcoR1 | Xba1 | Buffer2 | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.5 | 0.5 | 3 | 0.5 | 15.5 | 30 |
at 37℃ for 2 hours
→ purification 37.7ng/μL
Ligation
| Kil | pSB1C3 | Ligation High Ver.2 | total |
|---|---|---|---|
| 4 | 2 | 3 | 9 |
- Kil : 350fmol
- pSB1C3 : 29fmol
at 16℃ for overnight
March 7
====Electrophoresis====1. 1kb ladder 2µL
2. pspA (PCR product)2.5µL + Loading Dye 0.5µL
3. GFP 30µL + Loading Dye 6µL
4. 1kb ladder 2µL
- The GFP was gel extracted on 3/6 and concentrated by Vacuum in 150µg/mL
Ligation
| VectorDNA | GFP | Ligation High Ver.2 | total |
|---|---|---|---|
| 5 | 15 | 10 | 30 |
Restriction
| torA | EcoR1 | Spe1 | bufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.3 | 0.3 | 3 | 0.3 | 16.1 | 30 |
| pspA | EcoR1 | Spe1 | bufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 5 | 0.3 | 0.3 | 3 | 0.3 | 21.1 | 30 |
at 37℃ for 1.5 hours
Purification
torA→31.8ng/µL
pspA→49.3ng/µL
Ligation
| torA | pSB1C3 | Ligation High Ver.2 | total |
|---|---|---|---|
| 3 | 3 | 3 | 9 |
| pspA | pSB1C3 | Ligation High Ver.2 | total |
|---|---|---|---|
| 4 | 2 | 3 | 9 |
at 4℃, for overnight
- torA→31.8ng/µL×3µL=95.4ng=0.529pmol
- pSB1C3→19.4ng/µL×3µL=58.2ng=0.042pmol
- pspA→49.3ng/µL×4µL=197.2ng=0.308pmol
- pSB1C3→19.4ng/µL×2µL=38.8ng=0.029pmol
March 8
Restriction
| pSB4K5 | EcoR1 | Spe1 | BufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 20 | 0.2 | 0.2 | 3 | 0.2 | 6.4 | 30 |
at 37℃ for 1 hour.
→Purification : 36.6ng/µL
Ligation
| Kil | pSB4K5 | Ligation High Ver.2 | total |
|---|---|---|---|
| 10 | 1 | 5 | 16 |
at 4℃ for overnight
- Kil→37.7ng/µL×10µL=377ng=879fmol
- pSB4K5→36.6ng/µL×1µL=36.6ng=86fmol
Liquid culture
Lacp + pSB3C5 -1, 2
Transformation
| torA | pspA | competent cell | total |
|---|---|---|---|
| 1 | 0 | 10 | 11 |
| 0 | 1 | 10 | 11 |
We use commercially available competent cells in this time.
March 9
Restriction
| tatABCD | Xba1 | Pst1 | BufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.2 | 0.2 | 3 | 0.3 | 16.3 | 30 |
at 37℃ for 1hour
→purification : 75.0μg/mL (1.11 260/280 , 0.81 260/230)
Miniprep
lacP + pSB3C5 1 80.5μg/mL (1.78 260/280 , 2.00 260/230)
lacP + pSB3C5 2 107.2μg/mL (1.83 260/280 , 1.90 260/230)
Colony PCR
| Quick Taq | VF | VR | MilliQ | total |
|---|---|---|---|---|
| 25 | 1 | 1 | 23 | 50 |
94℃ 2min
94℃ 30sec
55℃ 30sec
68℃ 6sec
25cycles
Ligation
| tatABCD | constP J23107 | Ligation High Ver.2 | total |
|---|---|---|---|
| 5 | 1 | 3 | 9 |
tatABCD : 227fmol
constP J23107 : 21fmol
Transformation
| pspA | torA | Kil | competent cell | |
|---|---|---|---|---|
| 1 | 1 | 0 | 0 | 10 |
| 2 | 0 | 1 | 0 | 10 |
| 3 | 0 | 0 | 1 | 10 |
Miniprep
4mL of plusgrow which had been cultured for overnight.
pSB4K5 : 80.5μg/mL
March 10
Screening PCR
| Quick Taq | VF | VR | MilliQ | total |
|---|---|---|---|---|
| 25 | 1 | 1 | 23 | 50 |
Then we did electrophoresis to confirm.
1.1kb ladder
2.kil (649bp)
3,4,5, pspA (969bp)
6,1kb ladder
1, 100bp ladder
2,3,4, torA signal
Restriction
| LacP-pSB3C5 | Spe1 | Pst1 | BufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.2 | 0.2 | 2 | 0.2 | 7.4 | 20 |
for 2.5 hours at 37℃
→purification 29.0ng/μL
| torA | Xba1 | Pst1 | BufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.2 | 0.2 | 2 | 0.2 | 7.4 | 20 |
for 2.5 hours at 37℃
→purification 91.8ng/μL
Ligation
| torA | Lacp-pSB3C5 | Ligation High Ver.2 | total |
|---|---|---|---|
| 4 | 2 | 3 | 9 |
torA : 929fmol
Lacp-pSB3C5 : 284fmol
for overnight at 4℃
March 11
Miniprep
We used 3μL of plus grow that we had cultured for overnight.
torA : 62.8ng/μL
Screening PCR
| Quick Taq | VF | VR | MilliQ | total |
|---|---|---|---|---|
| 25 | 1 | 1 | 23 | 50 |
Electrophoresis
1. 1kb ladder
2〜11. pspA (969bp)
12. 1kb ladder
The results were shown as photograph in the right.
It seemed that there were shorter sample than expected sample, so we did electrophoresis with pspA which was product of PCR and pspA which had already cut with EcoR1 and Spe1.
1.1kb ladder
2. pspA (PCR product)
3, pspA (Eco, Spe)
4〜6, pspA (colony PCR)
7,1kb ladder
The results were shown as photograph in the right.
March 12
Transformation
| DT(1ng/μL) | DT(0.1ng/μL) | Kil | lacP-torA | MilliQ | competent cell | total |
|---|---|---|---|---|---|---|
| 1 | 0 | 0 | 0 | 0 | 20 | 21 |
| 0 | 1 | 0 | 0 | 0 | 20 | 21 |
| 0 | 0 | 5 | 0 | 0 | 50 | 51 |
| 0 | 0 | 0 | 5 | 0 | 50 | 51 |
| 0 | 0 | 0 | 0 | 1 | 20 | 21 |
Restriction
| pspA | EcoR1 | Spe1 | BufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.5 | 0.5 | 3 | 0.3 | 15.7 | 30 |
at 37℃ for 4 hours
→ We did purification and got 48.3ng/μL pspA.
Ligation
| pspA | pSB1C3 | MilliQ | Ligation High Ver.2 | total |
|---|---|---|---|---|
| 4 | 2 | 0 | 3 | 9 |
| 2 | 2 | 0 | 2 | 6 |
| 0 | 2 | 2 | 2 | 6 |
March 13
Miniprep
pSB1C3 74.2µg/mL 1.65 (260/280) 1.31 (260/230)
March 14
Restriction
| pSB1C3 | EcoR1 | Spe1 | BufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 20 | 0.2 | 0.2 | 4 | 0.4 | 15.2 | 40 |
We did gel extraction and got 47.2ng/µL pSB1C3 but we did not cut out RFP.
Ligation
| torA | pSB1C3 | Ligation High Ver.2 | total |
|---|---|---|---|
| 4 | 2 | 3 | 9 |
| MilliQ | pSB1C3 | Ligation High Ver.2 | total |
| 4 | 2 | 3 | 9 |
at 16℃, for 1 hour
- torA : 0.707pmol
- pSB1C3 : 0.068pmol
Liquid culture
We cultured Lacp-pSB3C5 1,2,3,4,5 (CP tolerance)on culture with Amp.
→Only 4 which did not be cultured succeeded.
March 15
Liquid culture
We cultured Lacp-pSB3C5 6,7,8,9,10 on culture with ampicillin.
→6,8,9,10 were succeeded.
Restriction
| GFP | EcoR1 | Spe1 | Buffer2 | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.5 | 0.5 | 3 | 0.5 | 15.5 | 30 |
| DT | EcoR1 | Xba1 | Buffer2 | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.5 | 0.5 | 3 | 0.5 | 15.5 | 30 |
for 2 hours at 37℃.
Miniprep
pspA (pSB1C3) 40.5ng/µL
Ligation
| pspA | DT | Ligation High Ver.2 | total |
|---|---|---|---|
| 5 | 1.5 | 3 | 9.5 |
- pspA : 385fmol
- DT : 36fmol
Transformation
| J23107-tatABCD | DT (0.1ng/µL) | DT (0.01ng/µL) | pspA-DT | competent cells on 3/15 | total |
|---|---|---|---|---|---|
| 2 | 0 | 0 | 0 | 20 | 22 |
| 0 | 2 | 0 | 0 | 20 | 22 |
| 0 | 0 | 2 | 0 | 20 | 22 |
| 0 | 0 | 0 | 2 | 20 | 22 |
Screening PCR
Kil, pspA and torA
| Quick Taq | Primer-r | Primer-f | MilliQ | total |
|---|---|---|---|---|
| 25 | 1 | 1 | 23 | 50 |
March 16
Miniprep
torA (pSB1C3) 68.8ng/µL
Kil (pSB4K5) 92.7ng/µL
torA was red for some reason. We do not know why.
Colony PCR
| Quick Taq | Primer-r | Primer-f | MilliQ | total |
|---|---|---|---|---|
| 25 | 1 | 1 | 23 | 50 |
94℃, 2min
94℃, 30sec
55℃, 30sec
68℃, 6sec
→25cycles
Electrophoresis
The results were shown as photograph in the right.
Checking Transformation Efficiency
competent cells that were made on March 15.
DNA : 0.02ng → 668 colonies Transformation Efficiency : 3.3×10^7
DNA : 0.2ng → 1739 colonies Transformation Efficiency : 8.7×10^6
Restriction
| pSB1C3 | EcoR1 | Spe1 | BSA | BufferM | BufferH | MilliQ | total |
|---|---|---|---|---|---|---|---|
| 20 | 0.2 | 0.2 | 0.3 | 3 | 0 | 6.3 | 30 |
| 5 | 0.2 | 0 | 0.2 | 0 | 2 | 12.6 | 20 |
at 37℃, for 2 hours.
We did gel extraction for product with EcoR1, Spe1. We got 46.1ng/µL pSB1C3.
| Kil(pSB4K5) | EcoR1 | Pst1 | BSA | BufferH | MilliQ | total |
|---|---|---|---|---|---|---|
| 5 | 0.2 | 0.2 | 0.2 | 2 | 12.4 | 20 |
| 5 | 0.2 | 0 | 0.2 | 2 | 12.6 | 20 |
for overnight at 37℃.
We did this to confirm.
| pspA (pSB1C3) | EcoR1 | Pst1 | BSA | BufferH | MilliQ | total |
|---|---|---|---|---|---|---|
| 5 | 0.2 | 0.2 | 0.2 | 2 | 12.4 | 20 |
| 5 | 0.2 | 0 | 0.2 | 2 | 12.6 | 20 |
for overnight at 37℃.
We did this to confirm.
Ligation
| torA | pSB1C3 | Ligation High Ver.2 | total |
|---|---|---|---|
| 4 | 2 | 3 | 9 |
| MilliQ | pSB1C3 | Ligation High Ver.2 | total |
|---|---|---|---|
| 4 | 2 | 3 | 9 |
at 16℃, for overnight
- torA→767fmol
- pSB1C3→68fmol
March 17
Miniprep
J23107-tatABCD 72.7ng/µL
pspA-DT 50.5ng/µL
Checking the Insert
| J21037-tatABCD | EcoR1 | Pst1 | BSA | BufferH | MilliQ | total |
|---|---|---|---|---|---|---|
| 5 | 0.2 | 0.2 | 0.2 | 2 | 12.4 | 20 |
| 5 | 0.2 | 0 | 0.2 | 2 | 12.6 | 20 |
Success.
| pspA-DT | EcoR1 | Pst1 | BSA | BufferH | MilliQ | total |
|---|---|---|---|---|---|---|
| 5 | 0.2 | 0.2 | 0.2 | 2 | 12.4 | 20 |
| 5 | 0.2 | 0 | 0.2 | 2 | 12.6 | 20 |
Failed.
March 19
Restriction
| DT | EcoR1 | Xba1 | BSA | BufferM | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.2 | 0.2 | 0.3 | 3 | 16.3 | 30 |
We did Gel extraction and got 17.2ng/µL of DT.
Ligation
| Kil | DT | Ligation High Ver.2 | total |
|---|---|---|---|
| 10 | 2 | 6 | 18 |
We did this for an hour at 16℃.
Restriction
| GFP | EcoR1 | Spe1 | BSA | BufferM | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.5 | 0.5 | 0.5 | 3 | 15.5 | 30 |
We did this for 4 hours at 37℃
Ligation
| pspA | DT | Ligation High Ver.2 | total |
|---|---|---|---|
| 5 | 5 | 5 | 15 |
| pspA | pSB1C3 | Ligation High Ver.2 | total |
|---|---|---|---|
| 4 | 1 | 3 | 8 |
We did these for an hour at 16℃.
- pspA (5µL)→377fmol
- DT→39fmol
- pspA (4µL)→339fmol
- pSB1C3→34fmol
Transformation
pspA-DT, pspA (pSB1C3), torA (pSB1C3) and GFP-DT
March 20
Screaning PCR
| Quick Taq | Primer-R | Primer-F | MilliQ | total |
|---|---|---|---|---|
| 25 | 1 | 1 | 23 | 50 |
- pspA→○
- pspA-DT→○
- GFP-DT→○
- torA→×
- Kil-DT 6 of 8 sumples→○
| Quick Taq | VR | VF | MilliQ | total |
|---|---|---|---|---|
| 25 | 1 | 1 | 23 | 50 |
- pspA→○
- pspA-DT→×
Restriction
| torA | EcoR1 | Spe1 | BSA | BufferM | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.3 | 0.3 | 0.3 | 3 | 16.1 | 30 |
| DT | EcoR1 | Xba1 | BSA | BufferM | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.2 | 0.2 | 0.3 | 3 | 16.3 | 30 |
We did this for overnight at 37℃. And we did purification.
torA 34.2ng/µL
DT 28.0ng/µL
March 21
Miniprep
GFP-DT-1 : 77.7ng/µL
GFP-DT-2 : 67.6ng/µL
Restriction
| pSB1C3 | EcoR1 | Spe1 | BSA | BufferM | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.2 | 0.2 | 0.3 | 3 | 16.3 | 30 |
| 5 | 0.2 | 0 | 0.2 | 2 | 12.6 | 20 |
We did this for 3 hours at 37℃, and then we did gel extraction. We got 25.1ng/µL pSB1C3
| GFP-DT | EcoR1 | Pst1 | Xba1 | BSA | BufferM | MilliQ | total |
|---|---|---|---|---|---|---|---|
| 5 | 0.2 | 0.2 | 0 | 0.2 | 2 | 12.4 | 20 |
| 5 | 0.2 | 0 | 0 | 0.2 | 2 | 12.6 | 20 |
| 10 | 0.2 | 0 | 0.2 | 0.3 | 3 | 16.3 | 30 |
We did this for 3 hours at 37℃, and then we did Purification. We got 30.7ng/µL GFP-DT.
Ligation
| torA | pSB1C3 | pspA | DT | GFP-DT | Ligation High Ver.2 | total | |
|---|---|---|---|---|---|---|---|
| 1 | 4 | 1 | 0 | 0 | 0 | 3 | 8 |
| 2 | 0 | 1 | 7 | 0 | 0 | 4 | 12 |
| 3 | 0 | 0 | 5 | 3 | 0 | 4 | 12 |
| 4 | 3 | 0 | 0 | 0 | 5 | 4 | 12 |
We did this for an hour at16℃.
March 22
PCR
We did PCR to amplify torA_signal that was product of PCR at March 5 with redesigned primers.
| Buffer | dNTPs | MgSO4 | Primer-F | Primer-R | Template | MilliQ | KOD plus neo | total |
|---|---|---|---|---|---|---|---|---|
| 5 | 5 | 3 | 1.5 | 1.5 | 0.5 | 32.5 | 1 | 50 |
94℃, 2min
98℃, 10sec
60℃, 30sec
68℃, 10sec
→30cycles
→Purification 110.7ng/µL
Restriction
| torA | EcoR1 | Spe1 | BSA | BufferM | MilliQ | total |
|---|---|---|---|---|---|---|
| 10 | 0.2 | 0.2 | 0.3 | 3 | 16.3 | 30 |
Ligation
| torA | pSSB1C3 | pspA | DT | GFP-DT | Ligation high | total | |
|---|---|---|---|---|---|---|---|
| 1 | 4 | 3 | 0 | 0 | 0 | 4 | 11 |
| 2 | 3 | 0 | 0 | 0 | 3 | 3 | 9 |
| 3 | 0 | 0 | 5 | 5 | 0 | 5 | 15 |
- torA (4µL)→512fmol
- pSB1C3→54fmol
- torA (3µL)→384fmol
- GFP-DT→36fmol
- pspA→377fmol
- DT→65fmol
March 23
Screening PCR
torA (pSB1C3), torA-GFP-DT and pspA-DT
| Quick Taq | VF | VR | MilliQ | total |
|---|---|---|---|---|
| 25 | 1 | 1 | 23 | 50 |
E.coli in liquid culture that had pspA(pSB1C3) also expressed RFP.
Restriction
| pspA | EcoR1 | Spe1 | BufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 5 | 0.3 | 0.3 | 3 | 0.3 | 21.1 | 30 |
And then we did ethanol precipitation
Ethanol precipitation
pspA 11.5ng/µL.
Miniprep
Lacp+pSB3C5-8 77.9µg/mL
Lacp+pSB3C5-10 69.0µg/mL
Kil+DT-4 58.0µg/mL
Kil+DT-7 15.2µg/mL
Restriction
| Lacp+pSB3C5-8 | Spe1 | Pst1 | Buffer2 | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 20 | 0.5 | 0.5 | 3 | 0.5 | 5.5 | 30 |
| Kil+DT-4 | Xba1 | Pst1 | Buffer2 | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 20 | 0.5 | 0.5 | 3 | 0.5 | 5.5 | 30 |
at 37℃ for 1.5 hours
And then we did Gel extraction.
Gel Extraction
Lacp+pSB3C5-8 40.4µg/mL
Kil+DT-4 26.8µg/mL
March 26
Miniprep
torA(pSB1C3) 18.5[ng/µL]
torA-GFP-DT 20.0[ng/µL]
Restriction
| torA(pSB1C3) | EcoR1 | Pst1 | BufferH | BSA | MilliQ | total |
|---|---|---|---|---|---|---|
| 5 | 0.2 | 0.2 | 2 | 0.2 | 12.4 | 20 |
| 5 | 0.2 | 0 | 2 | 0.2 | 12.6 | 20 |
| torA-GFP-DT | EcoR1 | Xba1 | Pst1 | BufferH | BufferM | BSA | MilliQ | total |
|---|---|---|---|---|---|---|---|---|
| 5 | 0.2 | 0 | 0.2 | 2 | 0 | 0.2 | 12.4 | 20 |
| 5 | 0.2 | 0 | 0 | 2 | 0 | 0.2 | 12.6 | 20 |
| 20 | 0 | 0.2 | 0.2 | 0 | 3 | 0.3 | 6.3 | 30 |
We did Gel extraction and then got ??? 28.7[ng/µL]
Ligation
| Lacp (pSB3C5) | torA-GFP-DT | Ligation High Ver.2 | total |
|---|---|---|---|
| 1 | 5 | 3 | 9 |
- Lacp : 22fmol
- torA-GFP-DT : 197fmol
| pspA | pSB1C3 | Ligation High Ver.2 | total |
|---|---|---|---|
| 10 | 1 | 5 | 16 |
| pspA | DT | Ligation High Ver.2 | total |
|---|---|---|---|
| 10 | 1 | 5 | 16 |
- pspA : 180fmol
- pSB1C3 : 18fmol
- DT : 16fmol
| LacP(pSB3C5) | Kil-DT | Ligation High Ver.2 | total |
|---|---|---|---|
| 1 | 5 | 3 | 9 |
for 2 hours at 16℃
Transformation
| Lacp-Kil-DT | competent cell | total |
|---|---|---|
| 1 | 10 | 11 |
March 27
Miniprep
We retryed miniprep of torA(pSB1C3).
We got torA and its concentration was 39.3[ng/µL].
Transformation
| Name | Well | Sample | Competent Cells | Total | Plate | Colony |
|---|---|---|---|---|---|---|
| BBa_K117004 | 14J(2011 plate2) | 5 | 20 | ? | ? | ? |
We added 100[µL] of culture medium before we started culturing the E.coli.
Screening PCR
| Quick Taq | VF2 | VR | MilliQ | Total |
|---|---|---|---|---|
| 25 | 1 | 1 | 23 | 50 |
Lacp-torA-GFP-DT 1, 3, 5, 6, 8, 9 was successful.
pspA-DT was failed.
E.coli that had pspA(pSB1C3) did not make any colony.
Lacp-Kil-DT 1,2,3,4,5,6,7,8 was failed.
Liquid culture
Lacp-torA-GFP-DT
 "
"