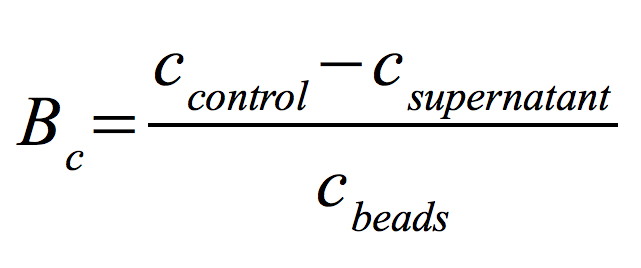Team:Bielefeld-Germany/Results/immo
From 2012.igem.org
(→Immobilization Strategy and Optimization of CPC-beads:) |
(→Immobilization Strategy and Optimization of CPC-beads:) |
||
| Line 25: | Line 25: | ||
In order to figure out the amount of protein bound to the beads after several incubation period of times, the protein concentration in the supernatant was measured using Roti®-Nanoquant. After comparing the results, it was noticeable that after 10 hours, the maximum amount of bound proteins to beads could be achieved (see Fig. 1). This remained relatively constant over the remaining incubation period, although an incubation time of at least 18 hours was recommended in all protocols. Therefore, another experiment was carried out to test the protein binding within the first 10 hours. The results showed that 6 hours were actually enough to reach a maximum binding (see Fig. 2) | In order to figure out the amount of protein bound to the beads after several incubation period of times, the protein concentration in the supernatant was measured using Roti®-Nanoquant. After comparing the results, it was noticeable that after 10 hours, the maximum amount of bound proteins to beads could be achieved (see Fig. 1). This remained relatively constant over the remaining incubation period, although an incubation time of at least 18 hours was recommended in all protocols. Therefore, another experiment was carried out to test the protein binding within the first 10 hours. The results showed that 6 hours were actually enough to reach a maximum binding (see Fig. 2) | ||
| - | [[File:Bielefeld2012_ | + | [[File:Bielefeld2012_ timealt.jpg|500px|left|thumb|Fig. 1: The mass of bound TVEL0 to beads (µg TVEL0/g beads) over different incubation periods of time. The highest capacity is achieved after an incubation period of 24 h.]] |
<br style="clear: both" /> | <br style="clear: both" /> | ||
Revision as of 15:37, 26 October 2012

Contents |
First Approach
The initial step was to find out a convenient method of immobilization using commercially acquired laccases from Trametes versicolor (named TVEL0) as a standard. The first alternative was the use of silica dioxide beads from the lab. Different bead concentrations were used (ratio 1:500, 1:1000 and 1:1500) and different buffers (HBSS buffer, recrystallization buffer and Britton-Robinson buffer). However, no significant activity could be detected. Hence, it was agreed to try out CPC-(controlled pore carrier) silica beads, to which laccases covalently bind, especially that some papers provide protocols and activity tests which prove the efficiency of these beads.
Immobilization Strategy and Optimization of CPC-beads:
In order to identify the best conditions for TVEL0, different bead concentrations: 0.01, 0.02, 0.04, 0.06, and 0.08 g.mL-1, as well as different incubation time periods: 18 h and 36 h were examined. Britton-Robinson buffer (pH 5) was used, since TVEL0 showed the highest activity in pH 5. The beads were first immersed in 2.5 % glutaraldehyde and incubated for two hours under light vacuum in order to allow as much beads’ surface area to be coated with aldehyde groups, which crosslink the laccases to the beads. After that, the beads were washed 3 times with Britton-Robinson Buffer and then immersed in 1mg.mL-1 laccases from TVEL0 and placed on a rotator at 4 °C for 18 h and 36 h respectively. These time periods were selected according to different protocols describing this immobilization method. After incubation, the supernatants were gathered to be tested for laccase activity. The beads were then washed with Britton-Robinson Buffer, then with 0.5 M NaCl solution in order to wash away all noncovalently bound laccases and again twice with the same buffer. Subsequently, the beads were immersed in 2.5 mg/mL glycine for 18 h at 4 °C. Finally, the beads were rinsed again with with Britton-Robinson Buffer, then with 0.5 M NaCl solution and again twice with the same buffer.
In order to figure out the amount of protein bound to the beads after several incubation period of times, the protein concentration in the supernatant was measured using Roti®-Nanoquant. After comparing the results, it was noticeable that after 10 hours, the maximum amount of bound proteins to beads could be achieved (see Fig. 1). This remained relatively constant over the remaining incubation period, although an incubation time of at least 18 hours was recommended in all protocols. Therefore, another experiment was carried out to test the protein binding within the first 10 hours. The results showed that 6 hours were actually enough to reach a maximum binding (see Fig. 2)
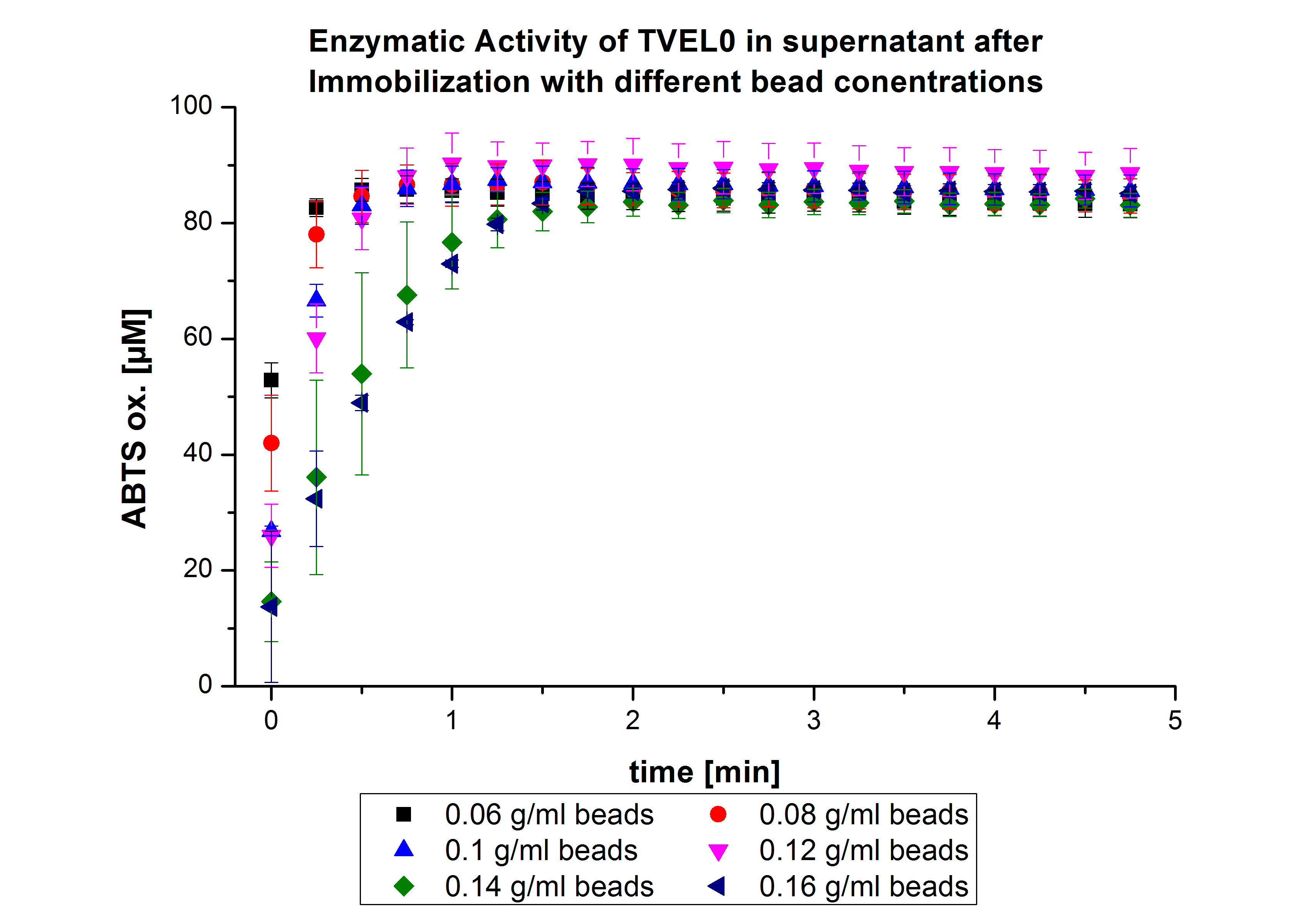
The activity of the nonbound laccases present in the supernatant was measured using ABTS as substrateOn the other hand, the activity also decreased upon increasing the concentration of the beads without reaching a saturation level (see Fig. 1). Therefore, another experiment was carried out with higher bead-concentrations.
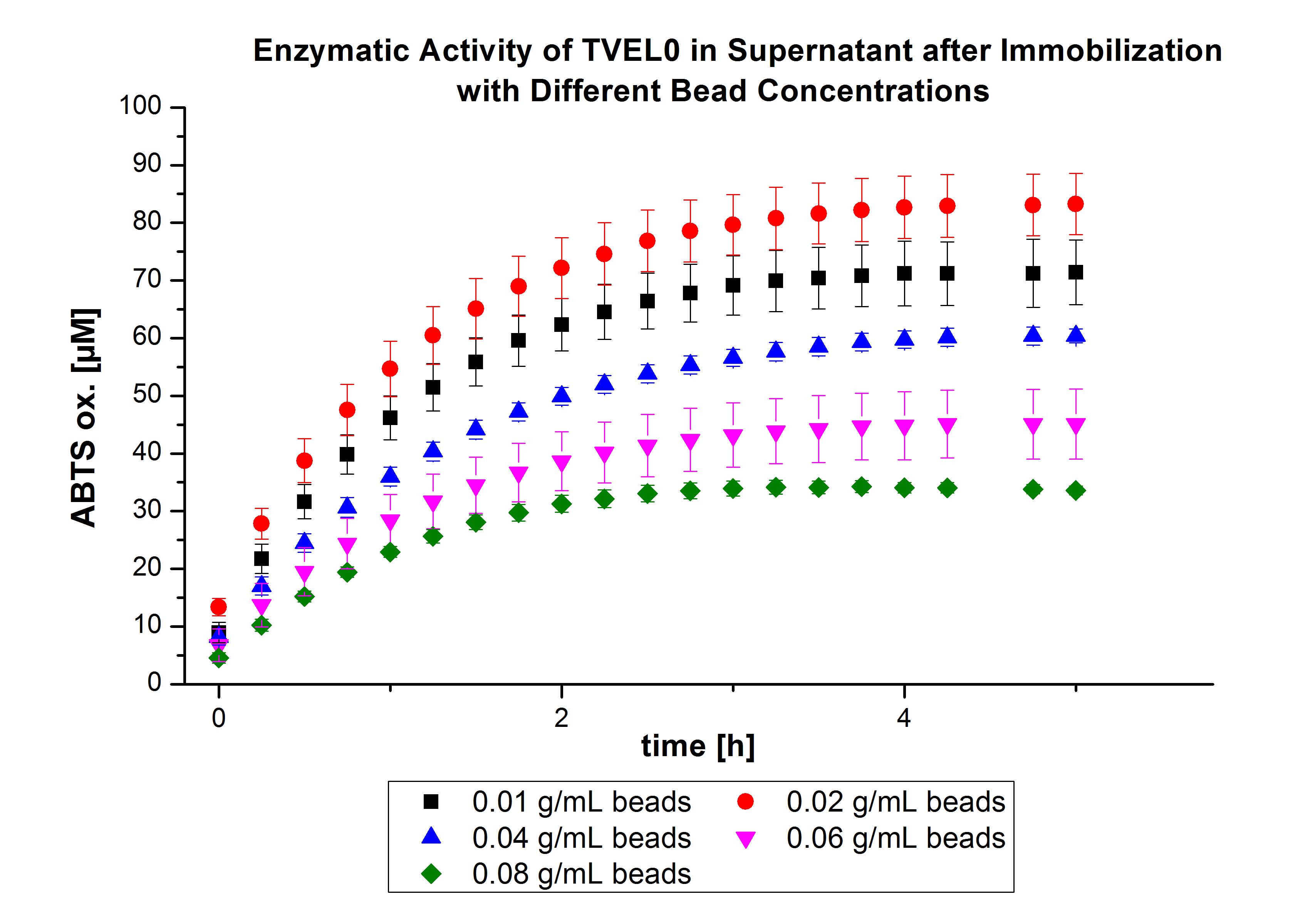
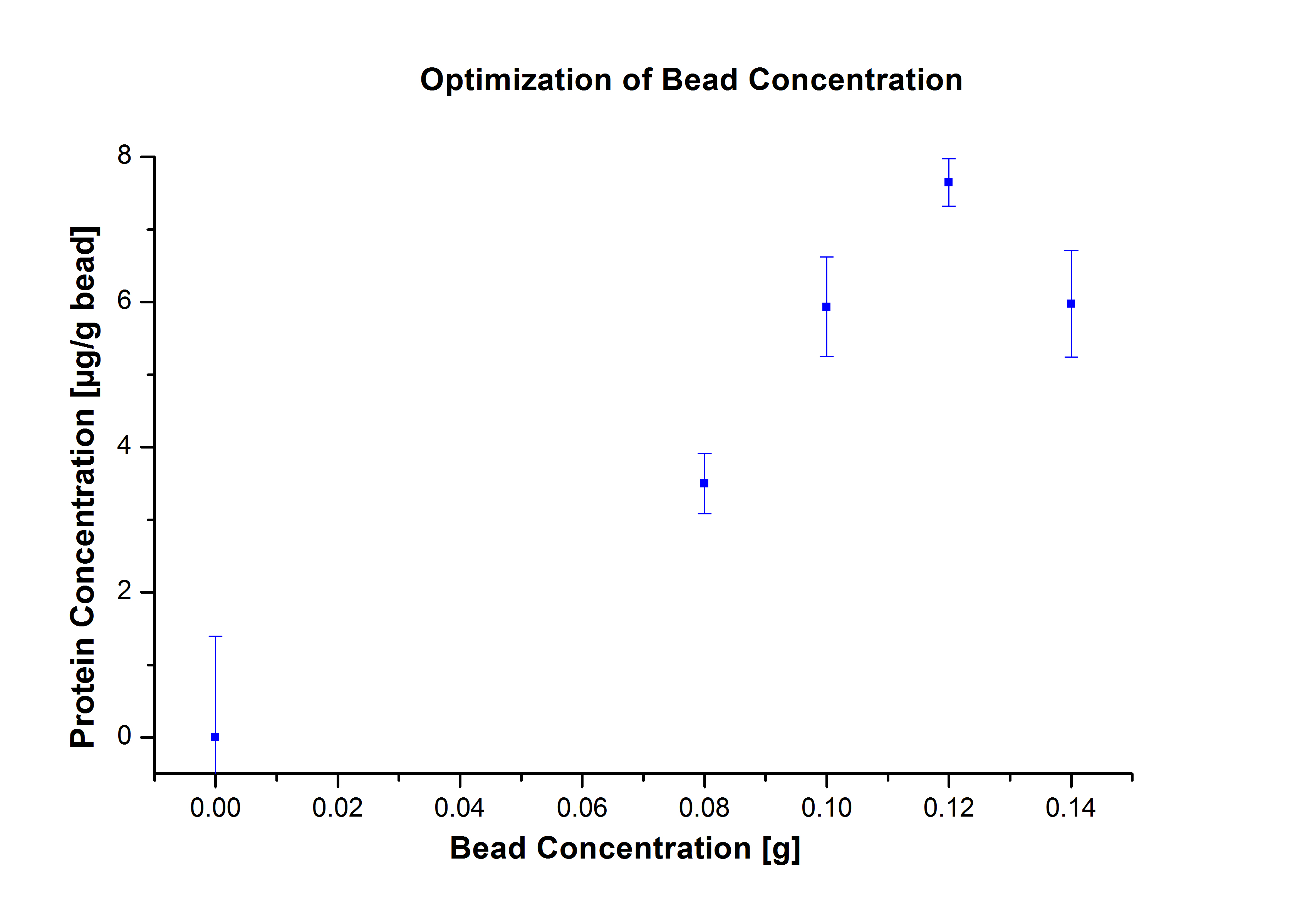
The same experiment was carried out again but with the following bead concentrations: 0.06, 0.08, 0.1, 0.12, 0.14 and 0.16 g mL-1. Based on the results of the previous experiment, the samples were incubated with 1 mg mL-1 TVEL0 for 36 h. The results showed no significant change in the laccase activity upon increasing the concentration of beads above 0.06 g ml-1 (see Fig. 2), a saturation level has been achieved.
However, as expected, it was not possible to measure the activity of laccases bound to beads using the Tecan. The beads were probably disturbing the Tecan’s laser, and centrifugation wasn´t an option because it would have simply taken too long and wouldn’t stop the reaction at a precise point of time. Therefore, multi-well membrane-bottom filter plates was considered to be a solution. These plates work in a similar way to the regular plates used for the Tecan but furthermore they contain a membrane that sieves the liquid through the filter. Thus the beads are separated and the ABTS-Buffer solution can be analyzed at 420 nm for oxidized ABTS.
The ability of the “multi-well membrane-bottom filter plates” to measure the activity of laccases bound to beads was tested with TVEL0 laccases. The results were promising (for further Information see labjournal. Subsequently, after receiving the laccases purified from E. coli BL21 (DE3) (named ECOL), the same procedures were followed to optimize the best conditions. However, the activity was measured over a longer period of time (35 min). The results indicated a similar behavior of ECOL to TVEL0 (see Fig. 3). Consequently, the same approach was followed for the immobilization of the next laccases.
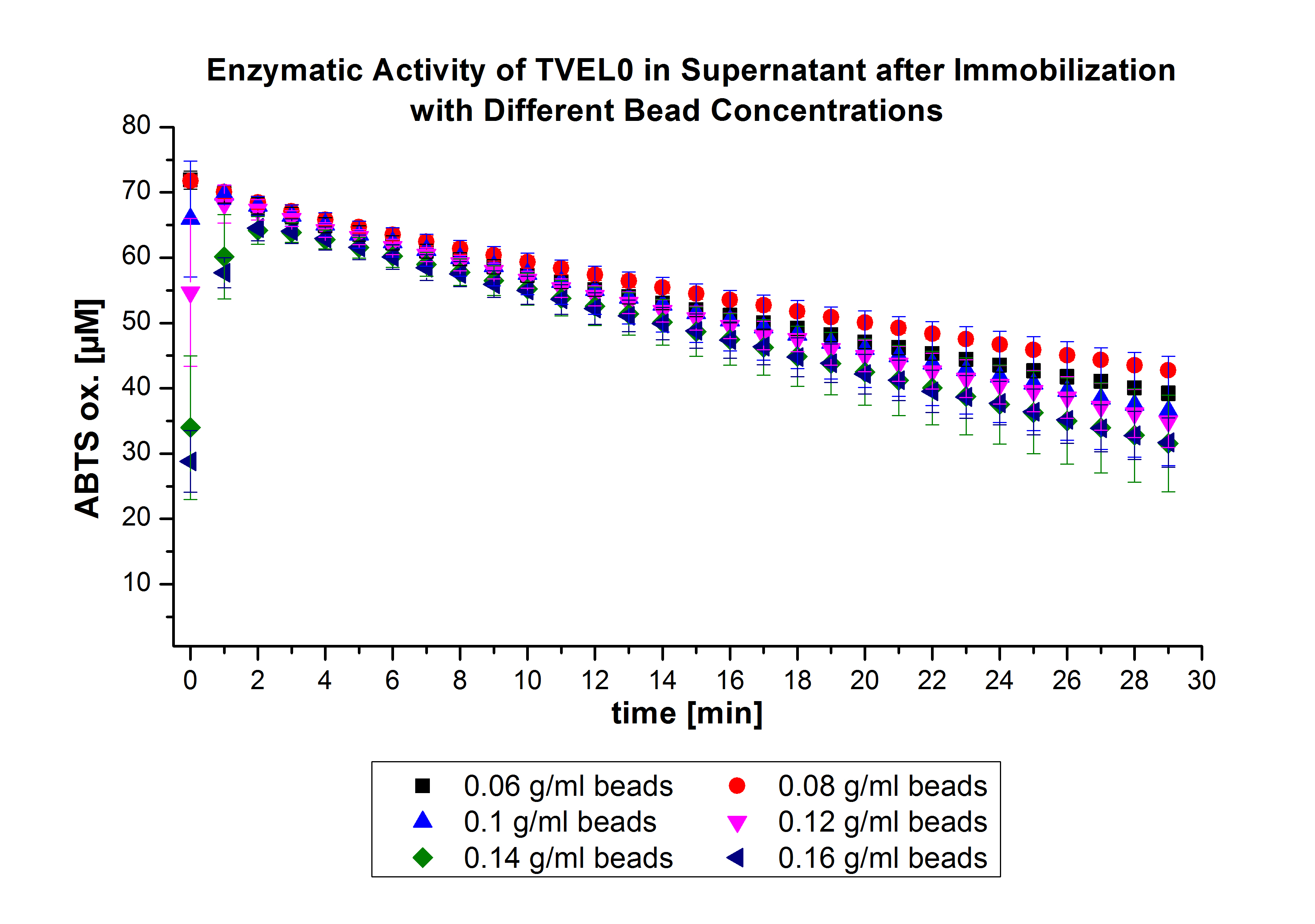
On the other hand, TVEL0 bound to beads showed a prompt activity in the first minutes vis-à-vis a slow ascending activity of bound E.coli. Due to the rapid reaction of TVEL0, it was only possible to detect the reverse reaction (Fig. 4).
Immobilization of purified ECOL and BPUL and Activity Tests
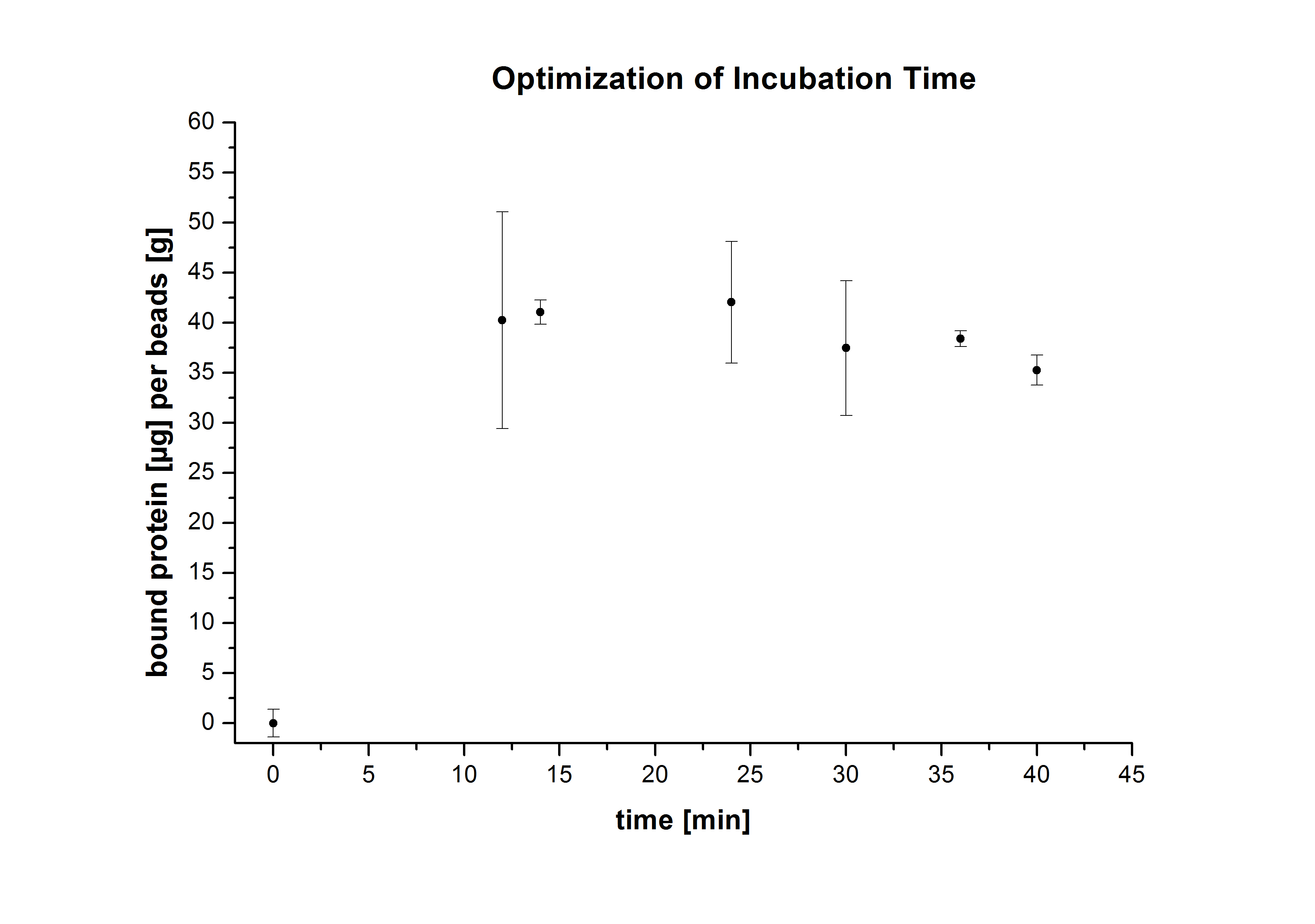
After optimizing the immobilization conditions for TVEL0 and ECOL, and proving their ability to bind to beads and preserve part of their activity, it was interesting to find out the percentage of laccases that bind to the beads, as well as the activity preserved by these laccases. Again, different bead concentrations were used: 0.08, 0.1, 0.12 and 0.14 g/ml, and the incubation time period was varied between 12 and 40 hours. The protein concentration was measured using Roti®-Nanoquant. In order to determine the optimal ratio of CPC-beads to protein, the binding capacity (Bc) was determined as follows:
The results were consistent with the activity tests performed previously. The most convenient bead concentration was 0.12 g/ml (see Fig.5) and the optimal incubation period was 24 h (see Fig. 6).
Accordingly, an immobilization experiment was carried out using TVEL0, ECOL and purified laccases from Bacillus pumilus DSM 27 ( ATCC7061) (named BPUL) with a bead concentration of 0.12 g and over an incubation period of 24 h. The concentration of laccases in the supernatant after incubation was measured using Roti®-Nanoquant. The percentage of laccases in the supernatant relative to the original concentration is presented in Fig. 7. The results show that only 0.2 % of ECOL laccases were still present in the supernatant, whereas 75 % of BPUL remained in the supernatant. This illustrate that almost all ECOL were bound to the beads. On the contrary, only 25 % of BPUL laccases were able to bind.

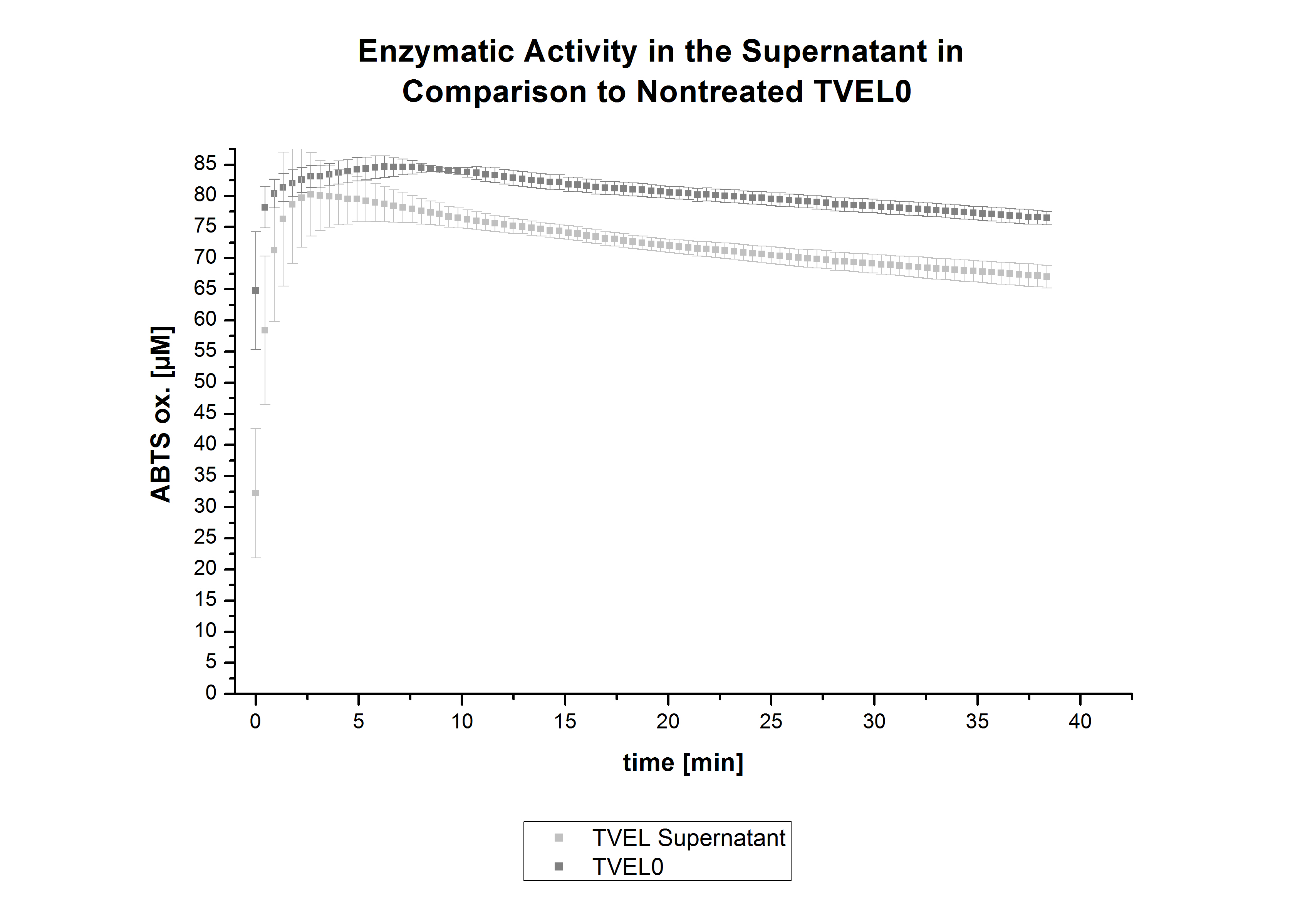
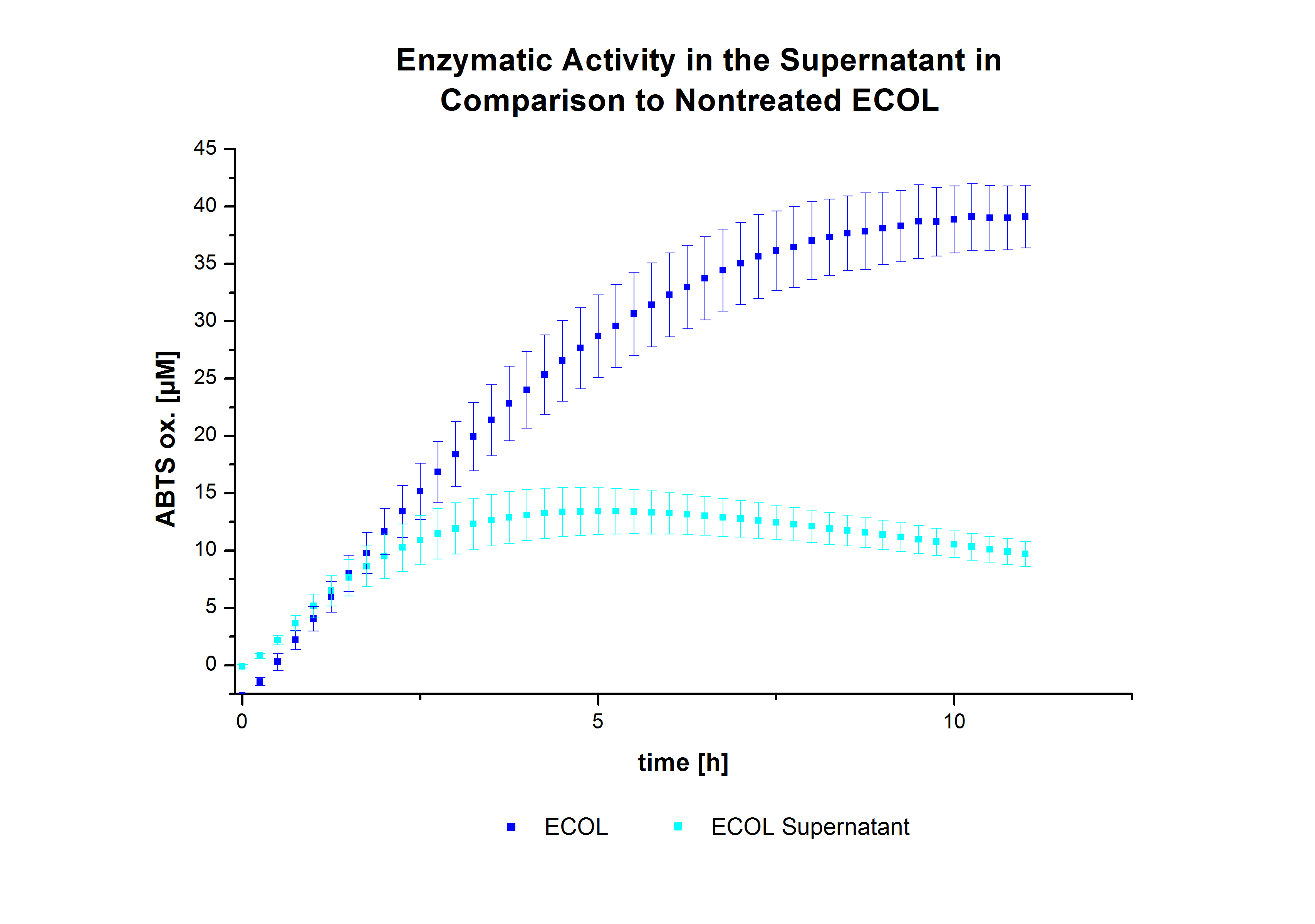
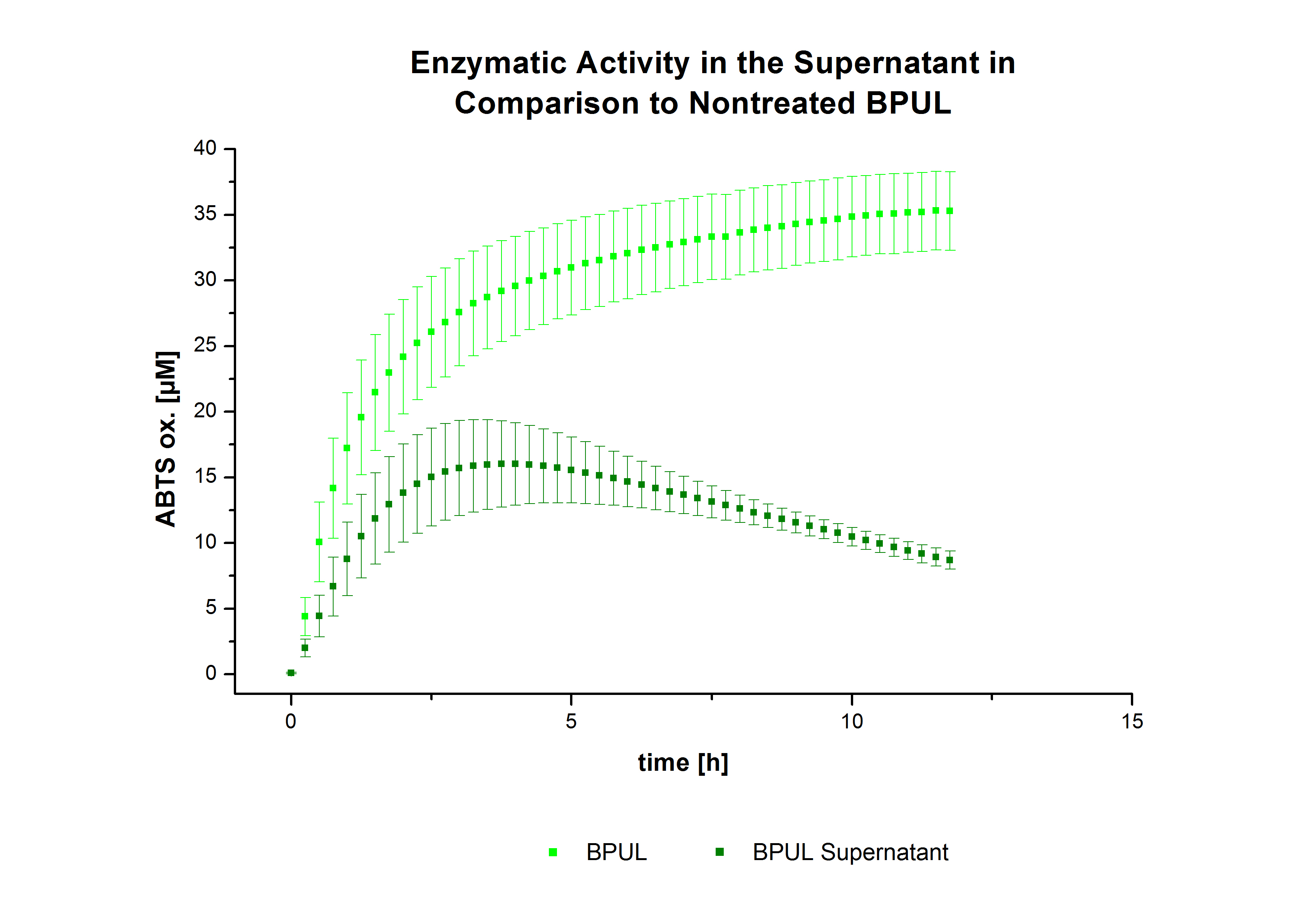
The activity of laccases in the supernatant was also measured and compared to the activity of nontreated laccases (Fig. 8-10). The results show that TVEL0 almost preserve their activity in the supernatant, whereas the activity of ECOL dramatically decreases compared to a slight decrease in the activity of BPUL. However, the graphs 9 and 10 show a similar behavior in the activity of ECOL and PBUL due to the higher concentration of ECOL used compared to PBUL. Still, all three laccases show indeed an activity.
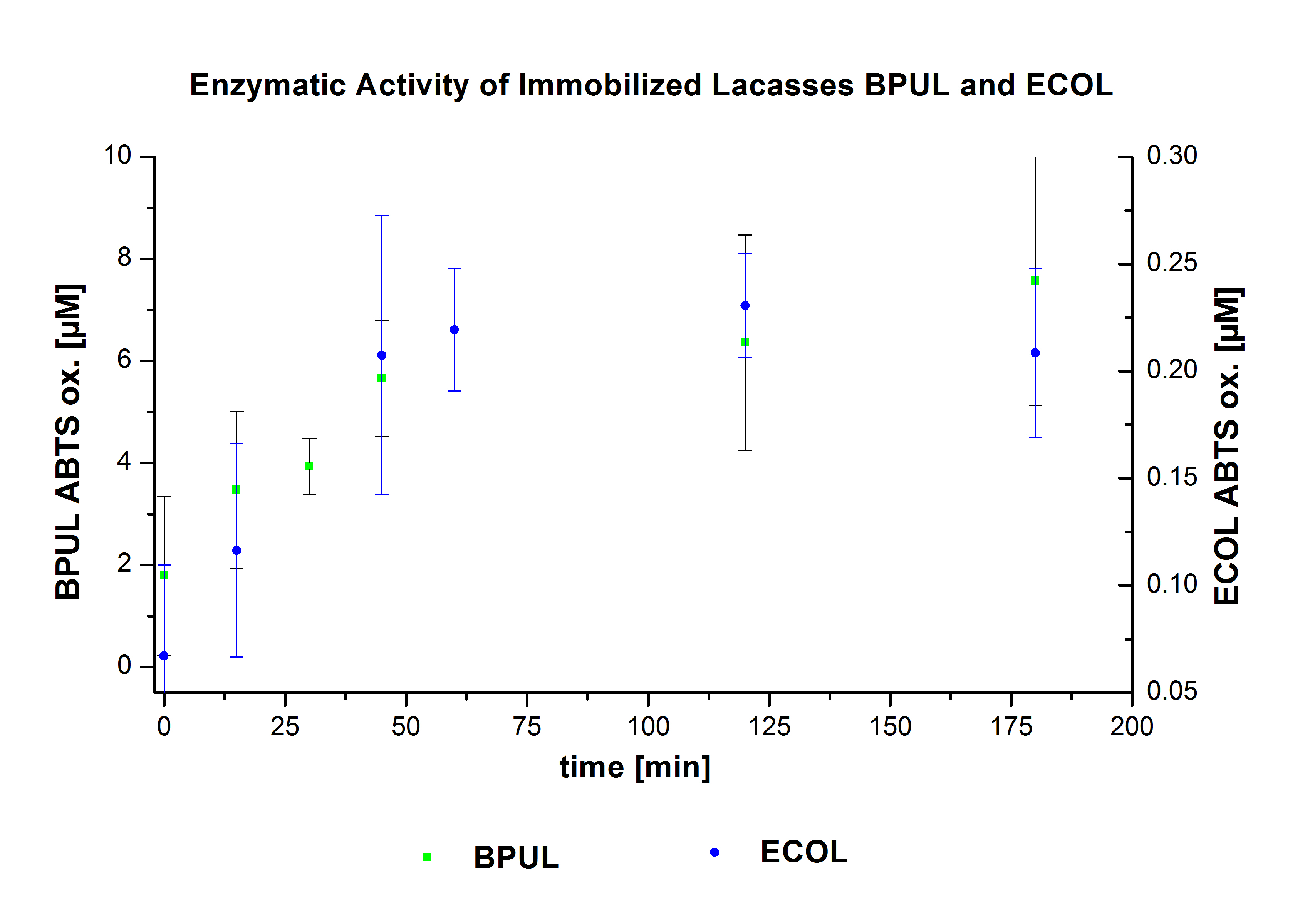
On the other hand, the enzymatic activity of the bound laccases was also measured (Fig. 11-12). TVEL0 shows a very rapid and high activity within few minutes. The activity of bound ECOL on the contrary is very low, even lower than that in the supernatant. BPUL shows a delayed increase in its activity, yet with higher values compared to ECOL despite the low binding capacity.
Literature
[1]Fernández-Fernández M et al. (2012) Recent developments and applications of immobilized laccase. Biotechnol Adv. 2012 Feb 28. [Epub ahead of print]
[2]P.-P. Champagne and J.A. Ramsay (2007) Reactive blue 19 decolouration by laccase immobilized on silica beads. Appl Microbiol Biotechnol. Oct;77:819–823
[3]Chantale Cardinal-Watkins and Jim A. Nicell (2011)Enzyme-Catalyzed Oxidation of 17ß-Estradiol Using Immobilized Laccase from Trametes versicolor. Enzyme Research, vol. 2011, Article ID 725172, 11 pages
| 55px | | | | | | | | | | |
 "
"