Team:Frankfurt/Project
From 2012.igem.org
(→The steviol precursor metabolism in yeast) |
|||
| Line 9: | Line 9: | ||
==The steviol precursor metabolism in yeast== | ==The steviol precursor metabolism in yeast== | ||
| - | [[Image:FPP_Synthase4.png| | + | [[Image:FPP_Synthase4.png|400px|right]] |
It is known from literature that yeast produces some mono-, sesqui- and diterpenes in very small amounts and so possesses a functional isoprenoid pathway. There are two known pathways for the production of isoprenyl pyrophosphate in nature. Isoprenyl pyrophosphate is converted into all the diverse isoprenoid compounts by condensing several units together. In bacteria and plastides isoprenyl pyrophosphate is produced via the methylerytritol pyrophosphate way (MEP) while it is produced via the mevalonate pathway (MVA) in eukaryotes. Many plants including stevia have crosslinks between the plastidic and the cytosolic pathway and produce the majority of isoprenoid compounds via the MEP pathway though they are eukaryotic organisms. | It is known from literature that yeast produces some mono-, sesqui- and diterpenes in very small amounts and so possesses a functional isoprenoid pathway. There are two known pathways for the production of isoprenyl pyrophosphate in nature. Isoprenyl pyrophosphate is converted into all the diverse isoprenoid compounts by condensing several units together. In bacteria and plastides isoprenyl pyrophosphate is produced via the methylerytritol pyrophosphate way (MEP) while it is produced via the mevalonate pathway (MVA) in eukaryotes. Many plants including stevia have crosslinks between the plastidic and the cytosolic pathway and produce the majority of isoprenoid compounds via the MEP pathway though they are eukaryotic organisms. | ||
Revision as of 03:56, 27 September 2012

Contents |
Our project aims for this years competition
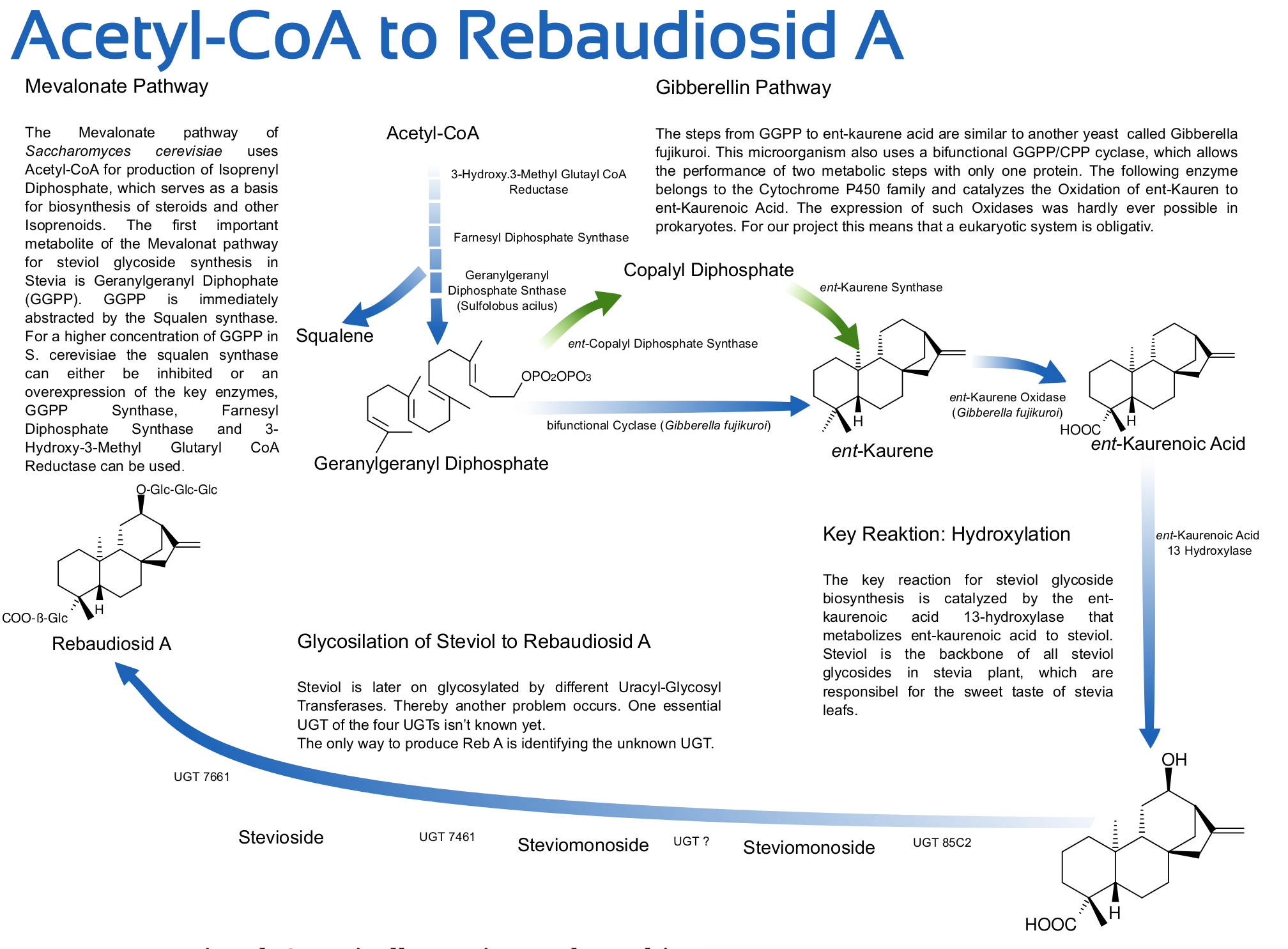
The sweetening agents produced by Stevia rebaudiana are all derived from the common precursor steviol which belongs to the diverse group of isoprenoids (also called terpenes). The term isoprenoid indicates that these compounts can theoretically be decomposed into units of the compound isoprene (2-methylbuta-1,3-dien). A diterpene like steviol is composed of four isopren units. All diterpenes in cell metabolism are derived from geranylgeranyl pyrophosphate (which is a diterpene itself). The synthesis of steviol is the aim for our participation in the iGEM 2012 competition. At least we want to introduce the necessary genes into our host organism Saccharomyces cerevisiae. To reach this goal one part of the project is the increase of the mevalonate pathway activity.
The steviol precursor metabolism in yeast
It is known from literature that yeast produces some mono-, sesqui- and diterpenes in very small amounts and so possesses a functional isoprenoid pathway. There are two known pathways for the production of isoprenyl pyrophosphate in nature. Isoprenyl pyrophosphate is converted into all the diverse isoprenoid compounts by condensing several units together. In bacteria and plastides isoprenyl pyrophosphate is produced via the methylerytritol pyrophosphate way (MEP) while it is produced via the mevalonate pathway (MVA) in eukaryotes. Many plants including stevia have crosslinks between the plastidic and the cytosolic pathway and produce the majority of isoprenoid compounds via the MEP pathway though they are eukaryotic organisms.
In yeast only the mevalonate pathway is present. The compound chiefly produced by yeast (from the isoprenoid family) is squalene. This is a triterpene from which the sterols (like cholesterol and ergosterol) are derived. Those compounds are essential for propagation because they control the membrane fluidity. Yeast with a corrupted sterol production is not able to grow unless ergosterol is present in the medium. Geranylgeranyl pyrophosphate is only produced in small amounts since yeast only uses it for very special purposes like the modification of proteins. We suppose that a proper function of the steviol producing pathway in yeast will only be possible when the flow of the mevalonate pathway is redirected to geranylgeranyl pyrophosphate without disturbing the production of squalene.
The early gibberelline pathway for the production of steviol
The generation of steviol from geranylgeranyl pyrophosphate takes place via four reactions.
The first two steps imply the double cyclization of geranylgeranyl pyrophosphate. In the first reaction ent-copalyl pyrophosphate is formed (induced by the protonation of a double bond, for that reason called proton-induced cyclization). The second reaction step leads to ent-kaurene (induced by dephosphorylation, which leaves a positive charge that acts as electrophile and attacks another double bond).
The following two reactions imply the oxidation of ent-kaurene. The third reaction implies the oxidation of ent-kaurene to form ent-kaurenoic acid. It is divided into three steps (ent-kaurenol, ent-kaurenal, ent-kaurenoic acid). The last reaction leads to a hydroxygroup via oxidation at position 13 to form steviol. It is the key reaction of the steviol glycoside pathway which seperates it from the parallel gibberelline pathway. As far as it is known it only occurs in plants of the genus Stevia and in the blackberry species Rubus suavissimus.
DNA constructs for realisation of our idea
Mevalonate Pathway Overexpression
The vector for improving the MVA pathway flow contains three genes: A trunctated version of the HMG-CoA-Reductase, the native gene of the Farnesylpyrophosphate Synthase (ERG20) and the GGPP Synthase gene from Sulfolobius acileratius. The sources of the physical DNA were as following:
- HMG-CoA-Reductase: The sequence was obtained from the Saccharomyces Genome Database and modified. This version was send to a company for de novo synthesis.
- FPP-Synthase: The gene was obtained from genomic DNA of the strain CEN.PK2. By using primers which contained overlaps with the pre- and suffix sequences it was amplified by PCR.
- GGPP-Synthase : The sequence was obtained from the Pubmed Database and modified. This version was send to a company for de novo synthesis.
construction of the insert:
pHXT7 HMG-CoA tHXT7 pPFK1 ERG20 tPFK2 pPGK1 GGPPS tCYC1
plasmid backbone with URA3
Steviol Production
The vector for steviol production also contains three genes: The bifunctional cyclase from Gibberella fujikuroi (CPS/KS), the ent-kaurene oxidase from Gibberella fujikuroi (KO) and the ent-kaurenoic acid hydroxylase from Stevia rebaudiana (KAH). The sources of the physical DNA were as following:
- bifunctional cyclase: The sequence was obtained from the Pubmed Database and modified. This version was send to a company for de novo synthesis.
- ent-kaurene oxidase: The sequence was obtained from the Pubmed Database and modified. This version was send to a company for de novo synthesis (4 x 500 bp for Gibson Assembly).
- ent-kaurenoic acid hydroxylase: The sequence was obtained from the Pubmed Database and modified. This version was send to a company for de novo synthesis (4 x 500 bp for Gibson Assembly).
construction of the insert:
pHXT7 CPS/KS tTAL1 pTPI1 KO tPDC1 pPGI1 KAH tCYC1
plasmid backbone with HIS3
DNA sources
| Templates | Amplified DNA Fragments |
|---|---|
| synthesized sequence of HMG-CoA | HMG-CoA |
| synthesized sequence of GGPPS | GGPPS |
| chromosomal DNA of CEN.PK2-1C | ERG20 |
| synthesized sequence of Cps/Ks | CPS/KS |
| synthesized sequence of KO | ent-kaurene oxidase |
| synthesized sequence of KAH | ent-kaurenoic acid hydroxylase |
| pUD8e (yeast shuttle plasmid) | pPFK1, pTPI1, tHXT7, tTAL1 |
| pUD22e (yeast shuttle plasmid) | pPGK1, pPGI1, tPFK2, tPDC1 |
Sequence Editing
The most of our genes were synthesized, because they contained several biobrick restriction sites. The relevant codons were replaced by different codons which encode for the same amino acid. Only ERG20 has no biobrick restriction sites.
- HMG-CoA
synthesized sequence
5`: 45 bp overlap to pHXT7 and the prefix
3`: the suffix and 33 bp overlap to tHXT7
- ERG20
gene from chromosomal DNA of CEN.PK2-1C
5`: 16 bp overlap to pPFK1 and the prefix (added via primer)
3`: the suffix and 14 bp overlap to tPFK2 (added via primer)
- GGPPS
synthesized sequence
5`: 18 bp overlap to pPGK1 and the prefix
3`: the suffix and 45 bp overlap to tCYC1
- CPS/KS
synthesized sequence
5': 45 bp overlap to PHXT7 and the prefix
3': the suffix and 17 bp overlap to tTAL1
- KO
synthesized sequence (4 x 500 bp for Gibson Assembly)
5': 15 bp overlap to pTPI1 and the prefix
3': the suffix and 16 bp overlap to tPDC1
- KAH
synthesized sequence (4 x 500 bp for Gibson Assembly)
5': 15 bp overlap to pPGI1 and the prefix
3': the suffix and 45 bp overlap to tCYC1
- promoters and terminators
They were amplified from plasmids via PCR with designed primers.
The primer contain about 20 to 30 bp of the promoter or terminator sequence for PCR and about 30 bp overlap to the neighbouring sequence for homologue recombination.
Gas Chromatographic Analysis for Examination of the Product content of the cells
The metabolites of our modified pathways in particular GGPP and Steviol should be analyzed via Gas Chromatography - Mass Spectrometry. We used overnight cultures of yeast cells at OD600=2. The cells were harvested via centrifugation and washed with physiological NaCl solution. The washed cells were homogenized by addition of glass beads and disruption for 20 minutes. Afterwards hexane was added to the disrupted cells. The Analytes were extracted by shaking for 15 minutes. Afterwards the suspension was centrifuges untill a clear organic phase had established itself on the surface. This phase was pipetted into a reaction tube and concentrated to a volume of 80µl using a Speed-Vac. In a GC vial the samples were treated with a derivation agent (Trimethylchlorsilan, TMS) for better seperation of Alcohols on non-polar columns. We used a simple temperature program for the GC runs: The oven temperature was increased constantly from 150°C to 300°C with 70°C/min.
Unfortunatly we were not able to proof our first intermediate GGPP. GGPP can not be analyzed directly (via GC), only the dephosphorylation product Geranylgeraniol can be analyzed. We abdicate on an active enzymatical digest of GGPP in our first trials. When we conduct further biochemical experiments we will try to proof GGPP both in wildtype and in transformant cells to compare the amounts.
We poofed the general method to be functional because other diterpene compounts such as Farnesol and Squalene which are closely related to our product could be analyzed.
Gap repair Cloning for Vector Assembly
Gap repair means the homologue recombination in yeast in order to assemble desired DNA fragments. It is easier and more effective than cloning via restriction and ligation. We used it to assemble the mevalonate overexpression plasmid and the steviol synthesis plasmid. All genes, promoters and terminators could be amplified except of the fragments for Gibson Assembly of KAH. Thus the mevalonate overexpression plasmid was assembled and transformed into yeast. To verify the correct assembly of the plasmid we did two control restrictions (see "Notebook"). Because we were not able to assemble KAH the gap repair of the steviol synthesis plasmid did not work. We also tried the gap repair with the four 500 bp fragments of KAH but the overlapping sequences are only 30 bp and not 40 bp which is needed for homologue recombination.
 "
"