Team:USP-UNESP-Brazil/Associative Memory/Experiments
From 2012.igem.org
(→Tests of System "Cin" of quorum sensing) |
|||
| Line 6: | Line 6: | ||
===Tests of System "Cin" of quorum sensing=== | ===Tests of System "Cin" of quorum sensing=== | ||
This experiment consists in checking the behavior of Biobricks which make part of the Cin system of Quorum Sensing. We will first check the production of QS substance observing the enzymatic activity of cinl (enzyme which produce the QS substance), secondly check the levels of repression and activation of cinR and, lastly, the intensity of positive feedback in the system, comparing the levels of activation and inhibition with the previously checked. | This experiment consists in checking the behavior of Biobricks which make part of the Cin system of Quorum Sensing. We will first check the production of QS substance observing the enzymatic activity of cinl (enzyme which produce the QS substance), secondly check the levels of repression and activation of cinR and, lastly, the intensity of positive feedback in the system, comparing the levels of activation and inhibition with the previously checked. | ||
| - | 3 constructions will be made in order to create 3 different lineages of bacteria. These lineages will be used in the following tests: | + | |
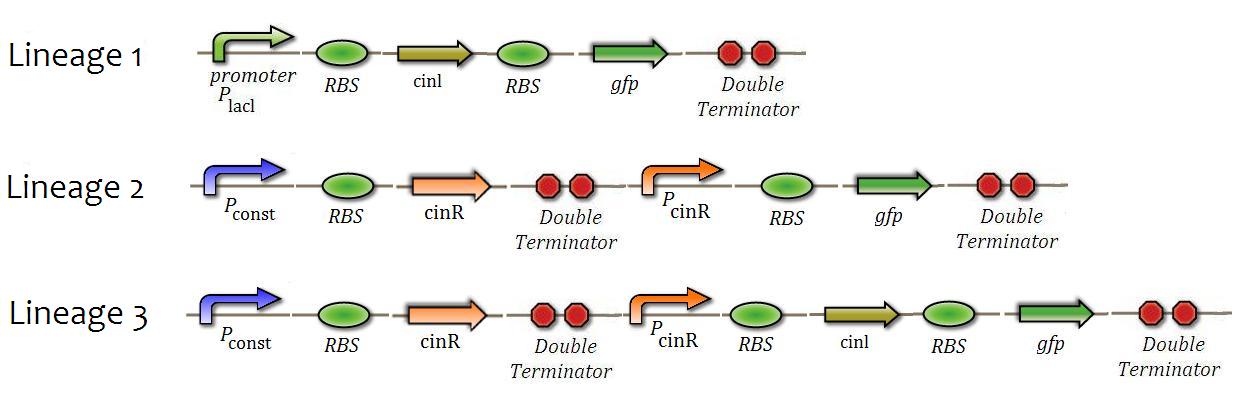
| + | 3 constructions will be made in order to create 3 different lineages of bacteria(figure 7). These lineages will be used in the following tests: | ||
| + | |||
| + | [[File:0010.JPG|center|400px|caption|]] | ||
| + | "Figure 7" | ||
| + | |||
HSL production test. | HSL production test. | ||
| - | Creating the E.coli lineage 1, we intend to observe the fluorescence in bacterial growth. This experiment doesn’t prove the activity of cinl, but shows signals of its operation since the GFP will be produced in the same ORF of the cinl gene. A graph of fluorescence X IPTG concentration used as input will be made. | + | Creating the E.coli lineage 1, we intend to observe the fluorescence in bacterial growth. This experiment doesn’t prove the activity of cinl, but shows signals of its operation since the GFP will be produced in the same ORF of the cinl gene. A graph of fluorescence X IPTG concentration used as input will be made. |
===Test of Response to QS Substance=== | ===Test of Response to QS Substance=== | ||
Revision as of 21:19, 22 September 2012
 Introduction
Introduction Project Overview
Project Overview Plasmid Plug&Play
Plasmid Plug&Play Associative Memory
Associative MemoryNetwork
 Extras
ExtrasContents |
Tests of System "Cin" of quorum sensing
This experiment consists in checking the behavior of Biobricks which make part of the Cin system of Quorum Sensing. We will first check the production of QS substance observing the enzymatic activity of cinl (enzyme which produce the QS substance), secondly check the levels of repression and activation of cinR and, lastly, the intensity of positive feedback in the system, comparing the levels of activation and inhibition with the previously checked.
3 constructions will be made in order to create 3 different lineages of bacteria(figure 7). These lineages will be used in the following tests:
"Figure 7"
HSL production test. Creating the E.coli lineage 1, we intend to observe the fluorescence in bacterial growth. This experiment doesn’t prove the activity of cinl, but shows signals of its operation since the GFP will be produced in the same ORF of the cinl gene. A graph of fluorescence X IPTG concentration used as input will be made.
Test of Response to QS Substance
For this test we will make the lineage 2 of E.coli. We intend to centrifuge the medium from the bacteria of lineage 1, pouring it on lineage 2 and verify if the members of lineage 2 will display fluorescence. Once we perceive fluoresce, we will have a strong signal that the QS system is working properly. In the case of any problem involving methodological variables, the GFP from lineage 2 can be replaced by RFP. In the case, we would mix lineage 1 and 2 members and observe the developing of both simultaneously. If a yellow fluorescence glows (Green + Red) – Like on microarrays – we will make sure that the lineage 2 is producing RFP under influence of the lineage 1.
Test for Internal feedback and basal production rate
In the case of satisfactory results in the previous tests, this one will probably work as well. The main objective is to estimate if the rate of basal expression is not excessive, in other words, if the positive feedback system is low enough. For this, we can compare the rate of basal fluorescence using the graph made on “HSL production test” as parameter. If the fluorescence doesn’t appear, there are 2 main possibilities: The feedback system isn’t working or it’s too low. In order to verify this issue, the system could be stimulated by insertion of QS substance, measuring the period of glowing and comparing it to the glowing time of bacteria from lineage 2 (which doesn’t have feedback system). It is possible to verify how much HSL will be necessary to activate the QS of the system. In order to do this, will be necessary to take samples from the growth medium from lineage 1 with several different “IPTG inductions”. It is likely to exists a threshold concentration of the QS substance in which the QS starts and the fluorescence of the system keeps glowing without external influence. Tests of Rhl system of Quorum Sensing. Just as previously, we intend to check the operation of the QS system, but this time with the rhl system. We will check: production of rhIL enzyme, levels of activation and repression of rhlR substance and ,lastly, intensity positive feedback system, comparing levels of activation and inhibition with tested previously. We will do the constructions presented in Figure 8 in addition to tests similar to the system in each Cin.
BHL production test.
The objective of this test is to confirm if the enzyme responsible by the synthesis of QS substance is being produced. The main evidence of it is the production of GFP under IPTG input. Using this test, a graph relating the quantity of IPTG and fluorescence will be made in order to be a reference for other tests. Reaction to the QS substance test. As seen in the test for reaction of Cin system under HSL, a centrifugation of the medium of lineage 2 will be poured on bacteria of lineage 1. If any issue emerge, the same methodology used on the tests of Cin system will be used. Test of Internal Feedback and Basal production rate Test. The levels of fluorescence of lineage 3 will be compared with the ones of other lineages and the concentration of BHL required to activate the QS system will be checked. The goals and procedures of this test are equivalent to those in QS Cin system.
Experiments of Multi Regulated Promoter
FIGURA The main goal of these experiments is checking if the Biobricks that constitute the system of genic regulation based on multi regulated promoter (Prm) are working properly. This promoter is activated by “cl” factor and inhibited by “cl434” factor. This system will be used to transform all input signals in activation of the system(indicated by cl production) or in inhibition of it(indicated by cl434). The main goal is to make sure if the rates of activating or inhibition of the promoter conform with cl and cl434. For the Prm promoter system functionality, the follow constructions will be made.
Test ofActivationby cl.
The main goal of this test is to create the “lineage 1” above and induce the production of cl and activation of GFP transcription by use of IPTG. It is expected the appearance of fluorescence, starting in a basal level, in the bacterial growth,
Test of Repression by cl434
The lineage 2 of E.coli Will be created and induced, by means of IPTG, to produce cl434 and, consequently, inhibit the production of tetR repressor, which represses PtetR promoter, the controller of GFP production. By this way, the transcription of GFP by PtetR will be stimulated. By this way, like in the previous experiment, it is expected to observe the emerging of fluorescence. There is a possibility that the basal levels of Prm transcription be too high or low to maintain a proper feedback in the QS system. In an ideal situation, the Prm would have transcription rates similar to the QS system promoters (PcinRandPrlhR), emulating the natural feedback of the QS systems- not enough to activate the system. A way to do it would be create mutant Prm promoters with a set of different transcription rates. This would be a good alternative, but probably will not make part in this Project due the deadlines and time required to do it. Assembly diagrams Previously, it was intent to use a light receptor (Red Light Suit) as input system. The strategy was to put the light responsive promoter in sequence to the Prm promoter, making the genic regulation of the following ORF. In order to simplify the Project, this part was removed. To illustrate all constructions used in all experiments, the Venn diagram (HERE(link)) was created, gathering all partial constructions in the main construction. In this LINK, there is the assembly fluxogram. We intend to assembly all biobricks using the 3A assembly method, except the smaller parts, like RBS and terminal sequences in which will be used the Standard Assembly Method. Depending on the method of assembly, a different scheme of digestion will be used. The different types of digestion are on the assembly fluxogram. Alternatively to the initial plan using the light switch, IPTG will be used as input to induce the initial production of GFP.
 "
"