Team:SJTU-BioX-Shanghai/Project
From 2012.igem.org
AleAlejandro (Talk | contribs) (→Why MEMBRANE?) |
Huanan1991 (Talk | contribs) (→Reference) |
||
| (3 intermediate revisions not shown) | |||
| Line 39: | Line 39: | ||
==Introduction== | ==Introduction== | ||
| - | In this year, we expanded the definition of ''scaffold'' in synthetic biology and developed two universal devices called ''Membrane Accelerator'' and ''Membrane Rudder'' respectively. | + | In this year, we expanded the definition of ''scaffold'' in synthetic biology and developed two universal devices called ''Membrane Accelerator'' and ''Membrane Rudder'' respectively. Together, they made ''Membrane Magic'' happen! |
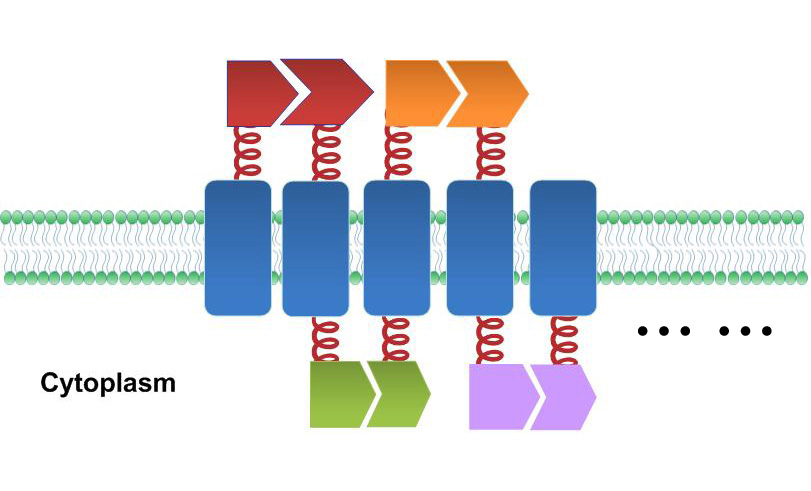
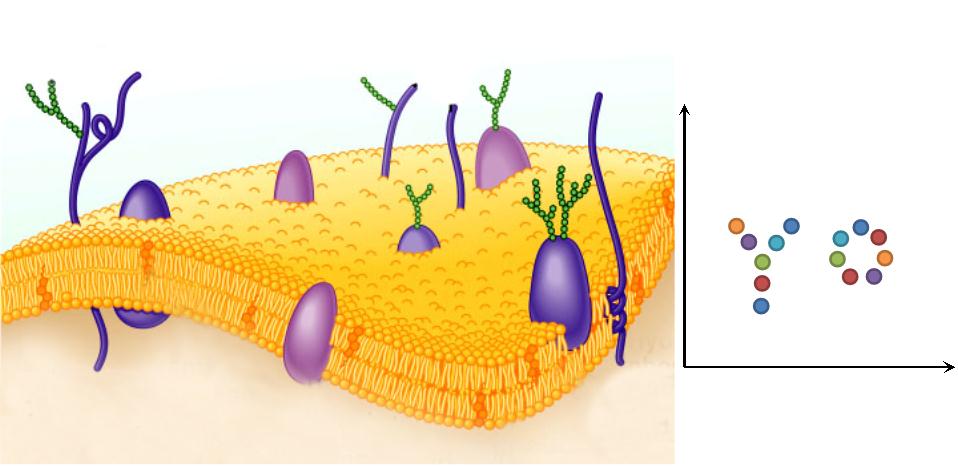
Previous researchers have focused on building protein, RNA or DNA scaffold as constitutive assemblies carrying enzymes. They have succeeded in increasing product yields. However, the amount of those scaffolds could be limited by its expression or copy level, leading to restriction on further acceleration. With ''Membrane Magic'', we made ''E.coli'' membrane into a huge scaffold accommodating enzymes without limitation of scaffold amount. Moreover, protein assembly on membrane could readily receive extracellular or intracellular signal, so the whole system becomes highly tunable. The superiority of Membrane Scaffold is shown in details in '''WHY MEMBRANE'' section below. | Previous researchers have focused on building protein, RNA or DNA scaffold as constitutive assemblies carrying enzymes. They have succeeded in increasing product yields. However, the amount of those scaffolds could be limited by its expression or copy level, leading to restriction on further acceleration. With ''Membrane Magic'', we made ''E.coli'' membrane into a huge scaffold accommodating enzymes without limitation of scaffold amount. Moreover, protein assembly on membrane could readily receive extracellular or intracellular signal, so the whole system becomes highly tunable. The superiority of Membrane Scaffold is shown in details in '''WHY MEMBRANE'' section below. | ||
| Line 70: | Line 70: | ||
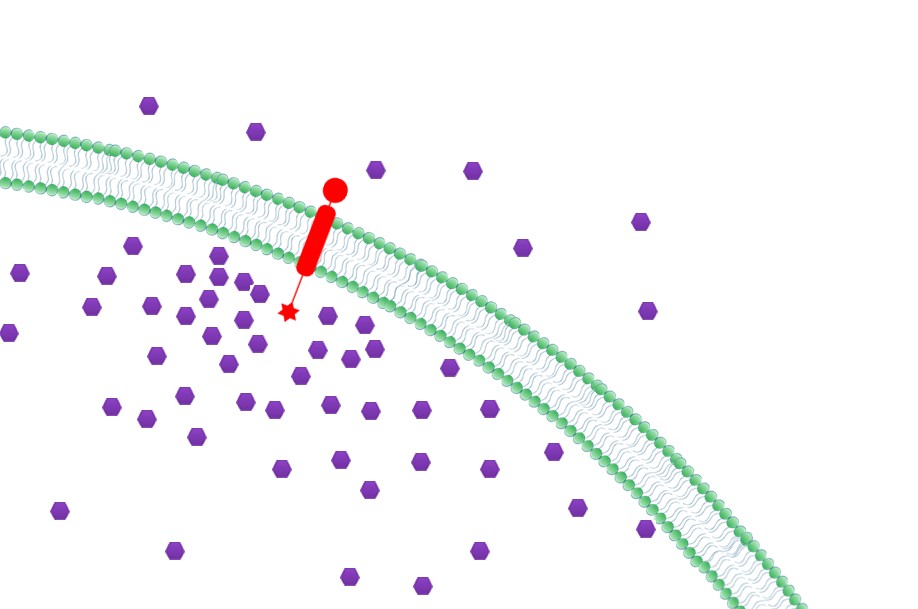
|[[File:12SJTU Why membrane3.jpg|300px|right|thumb|''Fig.5:'' Priority to Exportation]] | |[[File:12SJTU Why membrane3.jpg|300px|right|thumb|''Fig.5:'' Priority to Exportation]] | ||
|- | |- | ||
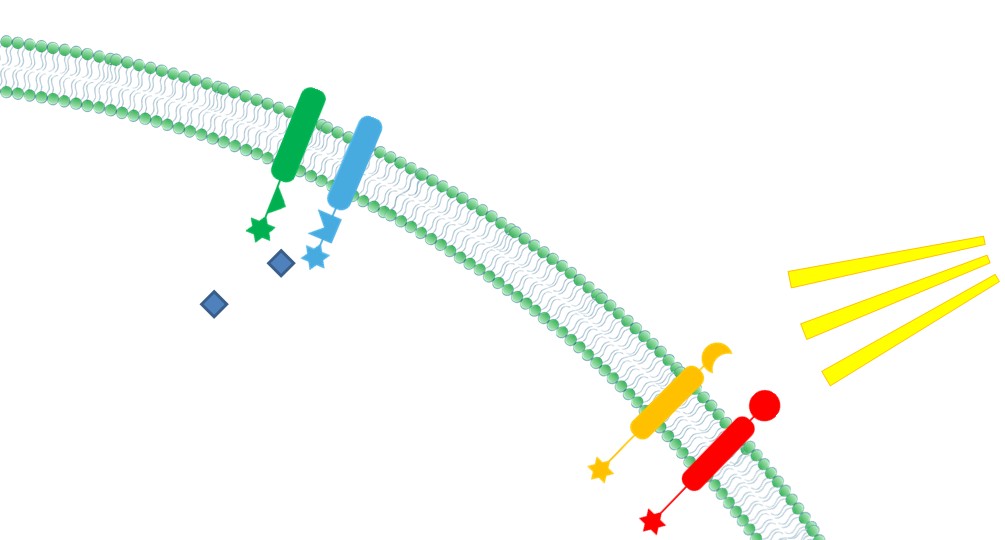
| - | |4. '''Ability to Sense Signals: ''' Membrane Scaffold provides a platform to directly receive environmental and internal signal, So biochemical reactions could be dynamically controlled through those signals(Demonstrated in [https://2012.igem.org/Team:SJTU-BioX-Shanghai/Project/project1.3 Membrane Rudder Design] and [https://2012.igem.org/Team:SJTU-BioX-Shanghai/Project/project2.1 Membrane Rudder Application]). | + | |4. '''Ability to Sense Signals: ''' Membrane Scaffold provides a platform to directly receive environmental and internal signal, So biochemical reactions could be dynamically controlled through those signals(Demonstrated in [https://2012.igem.org/Team:SJTU-BioX-Shanghai/Project/project1.3 Membrane Rudder Design] and [https://2012.igem.org/Team:SJTU-BioX-Shanghai/Project/project2.1 Violacein pathway: Membrane Rudder Application]). |
|[[File:12SJTU Why membrane4.jpg|300px|right|thumb|''Fig.6:'' Ability to sense signals]] | |[[File:12SJTU Why membrane4.jpg|300px|right|thumb|''Fig.6:'' Ability to sense signals]] | ||
|- | |- | ||
| Line 81: | Line 81: | ||
3.Conrado, R. J., G. C. Wu, et al. (2012). "DNA-guided assembly of biosynthetic pathways promotes improved catalytic efficiency." Nucleic acids research 40(4): 1879-1889. | 3.Conrado, R. J., G. C. Wu, et al. (2012). "DNA-guided assembly of biosynthetic pathways promotes improved catalytic efficiency." Nucleic acids research 40(4): 1879-1889. | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
<!----------------------------------------------------到这里结束---------------------------------------> | <!----------------------------------------------------到这里结束---------------------------------------> | ||
Latest revision as of 04:00, 27 October 2012
| ||||||||||
|
 "
"