Team:Washington/Flu
From 2012.igem.org
(→“For the first time in history we can track the evolution of a pandemic in real time. Influenza viruses are notorious for their rapid mutation and unpredictable behavior.”--Margaret Chan, WHO Director General) |
(→Test: Yeast Surface Display) |
||
| (30 intermediate revisions not shown) | |||
| Line 2: | Line 2: | ||
__NOTOC__ | __NOTOC__ | ||
==<i>“For the first time in history we can track the evolution of a pandemic in real time. Influenza viruses are notorious for their rapid mutation and unpredictable behavior.”--Margaret Chan, WHO Director General</i>== | ==<i>“For the first time in history we can track the evolution of a pandemic in real time. Influenza viruses are notorious for their rapid mutation and unpredictable behavior.”--Margaret Chan, WHO Director General</i>== | ||
| + | |||
<h1 id='Background'>Background</h1> | <h1 id='Background'>Background</h1> | ||
[[Image:FluTree.jpg|border|330px|right|thumb|There are 16 subtypes of Hemagglutinin(HA). They are divided into two groups based on their phylogenicity. We focused our efforts on the Group I HAs<html><a href="#Sources"><font size="1"><sup>[1]</sup></font></a></html>. ]] | [[Image:FluTree.jpg|border|330px|right|thumb|There are 16 subtypes of Hemagglutinin(HA). They are divided into two groups based on their phylogenicity. We focused our efforts on the Group I HAs<html><a href="#Sources"><font size="1"><sup>[1]</sup></font></a></html>. ]] | ||
| - | + | Influenza, also known as the flu, is a deadly virus that has decimated large populations of various species including humans. Even with the advent of vaccinations, high rates of mortality still occur because of the constant evolution of the virus. Variations within each strain of influenza are due to the different proteins that coat the virus' surface. | |
| - | + | ||
| - | One such protein is Hemagglutinin (HA), which is represented by the H when describing flu subtypes (H1N1, H2N3, etc). Hemagglutinin is primarily involved in viral transfer as it mediates the binding and entry into a host cell through a sialic acid linkage. Each strain of Influenza is characterized by it's variant of Hemagglutinin. There are 16 known subtypes of Hemagglutinin | + | [[File:Flu_virus.jpg|border|380px|left|thumb|Hemagglutinin is an viral protein that coats the surface of Influenza.]] |
| + | One such protein is Hemagglutinin (HA), which is represented by the H when describing flu subtypes (H1N1, H2N3, etc). Hemagglutinin is primarily involved in viral transfer as it mediates the binding and entry into a host cell through a sialic acid linkage. Each strain of Influenza is characterized by it's variant of Hemagglutinin. There are 16 known subtypes of Hemagglutinin which give rise to the multitude of strains that exist to today and usually only differ by a relatively few number of amino acid mutations. The World Health Organization uses the history of influenza breakouts to predict upcoming strains, which threaten to cause future pandemics, and vaccinations are created using these predictions. | ||
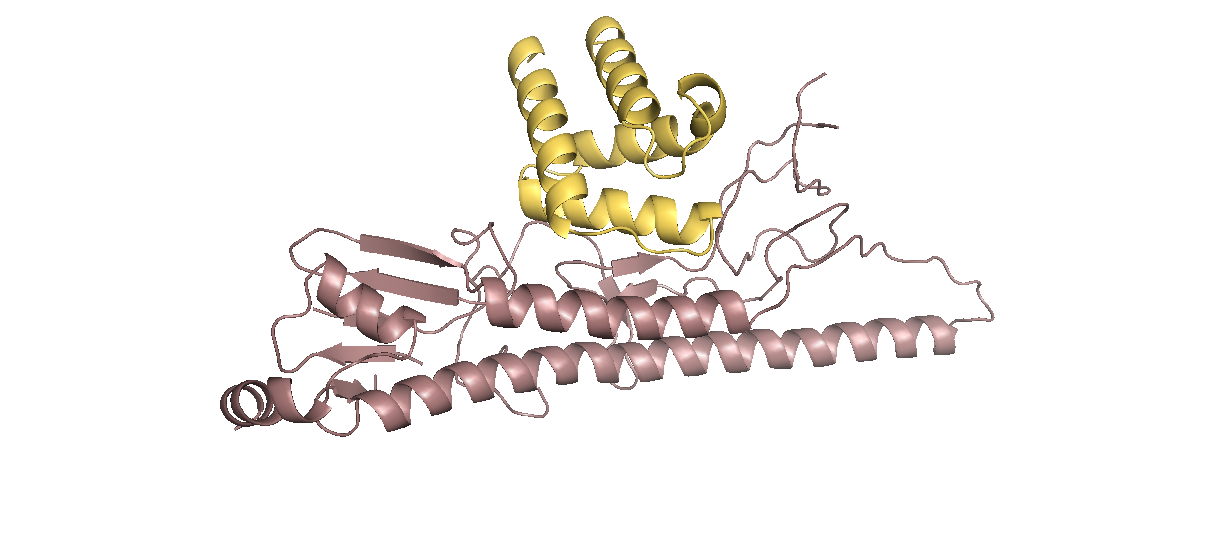
[[Image:Washington_HB36.png|border|350px|left|thumb|HB36.5 original flu binder, shown as a four-bundle helix in yellow, bound to a monomer subunit of the trimer Hemagglutinin in purple. We focused our design efforts on finding differences near the HB36.5 binding epitope(same as HB80.4 binding epitope) on the different HAs tested.]] | [[Image:Washington_HB36.png|border|350px|left|thumb|HB36.5 original flu binder, shown as a four-bundle helix in yellow, bound to a monomer subunit of the trimer Hemagglutinin in purple. We focused our design efforts on finding differences near the HB36.5 binding epitope(same as HB80.4 binding epitope) on the different HAs tested.]] | ||
In 2011, Fleishman et al., detailed two small proteins which had binding capabilities to various subtypes of hemagglutinin. In 2012, Whitehead et al., optimized the flu binders and showed that HB80.4 was broadly binding, meaning it bound to multiple HAs with high affinity. They also showed that HB36.5 bound preferentially to HA 1. | In 2011, Fleishman et al., detailed two small proteins which had binding capabilities to various subtypes of hemagglutinin. In 2012, Whitehead et al., optimized the flu binders and showed that HB80.4 was broadly binding, meaning it bound to multiple HAs with high affinity. They also showed that HB36.5 bound preferentially to HA 1. | ||
| - | We aimed to further optimize the binding of HB80.4 to the group 1 HAs, | + | |
| - | + | We aimed to further optimize the binding of HB80.4 to the group 1 HAs, but knowing that HB80.4 was already broadly binding to many HAs, we wanted to focus our efforts on the flu binder HB36.5 which until had not yet been tested against any other HAs is group 1. Therefore, we tested HB36.5 against the other group 1 HAs and assessed its native binding capabilities. After testing, we discovered that in addition to HA1, HB36.5 bound to H5, H6, H9 and H13. Binding was not observed to HA2 or HA12 subtypes however. Through application based design using FoldIt and real world testing we were able to find multiple single amino acid mutations to the binders that '''improved binding to the flu subtypes HA2, HA6, and HA9.''' Having two broadly-binding flu binders, with subtype specific capabilities would allow for enhanced rapid and reliable global tracking of current and future Influenza strains. | |
| + | |||
| Line 25: | Line 28: | ||
| - | + | ---- | |
<h1 id='Methods'>Methods <html><a href="#Background"><font size="3">[Top]</font></a></html></h1> | <h1 id='Methods'>Methods <html><a href="#Background"><font size="3">[Top]</font></a></html></h1> | ||
[[Image:flu buildtestdesign.png|border|1500px|center|thumb|An overview of our our project.]] | [[Image:flu buildtestdesign.png|border|1500px|center|thumb|An overview of our our project.]] | ||
| Line 31: | Line 34: | ||
== Design: Method of computational design == | == Design: Method of computational design == | ||
===Gathering information about the structures of Hemagglutinin=== | ===Gathering information about the structures of Hemagglutinin=== | ||
| - | Since we wanted to improve the binding of HB80.4 and HB36.5 to specific HAs, we had to understand the structural differences of the many subtypes of hemagglutinin. Initially, we searched for specific hemagglutinin | + | Since we wanted to improve the binding of HB80.4 and HB36.5 to specific HAs, we had to understand the structural differences of the many subtypes of hemagglutinin. Initially, we searched for specific hemagglutinin structure on the [http://www.rcsb.org/pdb/home/home.do '''Protein Data Bank'''] (PDB). If we found a desired hemagglutinin structures, we downloaded the associated PDB file and made a model of the binder-HA complex in Pymol. These models would then later be used in [http://fold.it/portal/ '''Foldit''']. |
| + | |||
If we could not find the exact structure of a particular hemagglutinin, we then searched for the protein sequence of that hemagglutinin on the National Center for Biotechnology Information (www.ncbi.nih.gov). We then aligned the protein sequence of the structurally unknown hemagglutinin we obtained to that of the H1 HA (which has a known structure) by using [http://www.ebi.ac.uk/Tools/msa/clustalw2/ '''Clustalw2''']. Next, we created homology models in FoldIt of the desired hemaggluttinin by changing the side chains of the H1 HA in Foldit according to the protein sequence differences we found. | If we could not find the exact structure of a particular hemagglutinin, we then searched for the protein sequence of that hemagglutinin on the National Center for Biotechnology Information (www.ncbi.nih.gov). We then aligned the protein sequence of the structurally unknown hemagglutinin we obtained to that of the H1 HA (which has a known structure) by using [http://www.ebi.ac.uk/Tools/msa/clustalw2/ '''Clustalw2''']. Next, we created homology models in FoldIt of the desired hemaggluttinin by changing the side chains of the H1 HA in Foldit according to the protein sequence differences we found. | ||
===Using the computational application Foldit to design our models=== | ===Using the computational application Foldit to design our models=== | ||
| - | Crystal stuctures of HB36.3 and HB80.4 in complex with H1HA are available on the PDB as PDB ID 3R2X and 4EEF, respectively. To create models | + | Crystal stuctures of HB36.3 and HB80.4 in complex with H1HA are available on the PDB as PDB ID 3R2X and 4EEF, respectively. To create models of the binder bound to different HAs, we used PYMOL to align our alternative HA structures to the H1HA from the published crystal structures. Then we would record the difference in side chains between that specific hemagglutinin and H1HA and use FoldIt to implement those changes on the known crystal structures. The models we created are linked below: |
{|align="center" | {|align="center" | ||
![[File:HB80_H12_model.zip]] | [[File:HB80_H9_model.zip ]] | [[File:HB80_H5_model.zip]] | [[File:HB36_H13_model.zip ]] | [[File:HB36_H12_model.zip]] | ![[File:HB80_H12_model.zip]] | [[File:HB80_H9_model.zip ]] | [[File:HB80_H5_model.zip]] | [[File:HB36_H13_model.zip ]] | [[File:HB36_H12_model.zip]] | ||
| Line 44: | Line 48: | ||
|} | |} | ||
| - | |||
| - | |||
| - | [[Image:Washington H312A.jpg|border| | + | [[Image:Washington H312A.jpg|border|600px|left|thumb| On the left: A histidine residue on the HB36.5 binder (purple) clashed with a phenylalanine reside on H2 (green) and showed a positive-predicted score, which indicated poor binding. |
On the right: the histidine residue was mutated to an alanine, thus reducing clashing and predicting better binding.]] | On the right: the histidine residue was mutated to an alanine, thus reducing clashing and predicting better binding.]] | ||
| - | == | + | ===Proposing Mutations based on FoldIt Models=== |
| - | + | After preparing the FoldIt model, we proposed mutations on the original flu binder in order to improve its binding to the desired hemagglutinin. There is a score total in FoldIt which indicates the energy of the whole protein structure. The protein structure is more stable when its energy is lower. Thus, our primary goal was to decrease the total score in FoldIt. We did that in three different ways, filling the holes, making space, and balancing the electrostatic interactions. Holes that did not exist in H1HA would be created with the replacement of different side chains. We would mutate the corresponding side chain on the flu binder so that it extends out to fill a hole in the hemagglutinin. Making space is the exact opposite of filling the holes. Some side chains of certain hemaggluttinins are more projected out in space than that of hemagluttinin one. Therefore, we would mutate the corresponding side chains of the flu binder in order to provide room for the more extended side chains. Some variations of side chains on hemaggluttinin cause electrostatic changes. We would mutate the corresponding side chains of the flu biunder so as to counter balance the electrostatic changes. For example, if a certain area on the helix is changed to be more electronegative, we would introduce an electropositive side chain on the flu binder for improving the binding and vice versa. | |
| + | |||
[[Image:Washington Kunkels.png|500px|thumb|left|Overview of how Kunkel Mutagenesis works]] | [[Image:Washington Kunkels.png|500px|thumb|left|Overview of how Kunkel Mutagenesis works]] | ||
| - | + | == Build: Kunkel Mutagenesis - Mutagenizing HB80.4 and HB36.5 flu binders == | |
| - | + | [https://2012.igem.org/Team:Washington/Protocols/Kunkel '''Kunkel mutagenesis'''] is a classic procedure for incorporating targeted mutations into a plasmid and we used it to create many variants of the original flu binders. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | The proteins tested were encoded on the shuttle vector pETCON which contained the yeast display components and was compatible with expression in both ''E. Coli'' (bacteria) and ''S. Cerevisiae'' (Yeast). This allowed us to do Kunkel Mutagenesis in ''E. Coli'' and not have to switch vectors when we transformed into yeast for Yeast Surface Display testing. | ||
| + | The first step to producing our specially designed flu binders was to change the original flu binding protein with our desired amino acid substitutions. We designed mutagenic oligonucleotide primers that would anneal to the original flu binding protein and incorporate point mutations that, when expressed, would result in a variant of the binder with the desired amino acid alteration. To incorporate these mutations, we first isolated single stranded DNA (ssDNA) of our vector harboring the original binding sequence. To do this we infected cells with bacteriophage M13, which packages its own ssDNA genome identified by length, and so in tandem packaged our vector in single stranded form. We then harvested the phage from the lysed culture of ''E. Coli'', and extracted our single stranded vector DNA. Next, we annealed and extended our mutagenic oligos to incorporate the specified mutations into the newly synthesized antisense strand. This hybrid vector was transformed into ''E. Coli'' which degraded the original DNA and replaced it with sections complementary to the mutagenized strand. | ||
==Test: Yeast Surface Display== | ==Test: Yeast Surface Display== | ||
[[Image:Washington ysdexplanation Screen shot 2012-10-03 at 2.45.23 PM.png|border|450px|right|thumb|Overview of Yeast Surface Display protocol.]] | [[Image:Washington ysdexplanation Screen shot 2012-10-03 at 2.45.23 PM.png|border|450px|right|thumb|Overview of Yeast Surface Display protocol.]] | ||
| - | To test our designs, we [https://2012.igem.org/Team:Washington/Protocols/Yeast '''transformed'''] our mutants into the organism ''Saccharomyces cerevisiae'' in order to perform surface display and binding efficiency analysis. We then used [https://2012.igem.org/Team:Washington/Protocols/Display '''yeast surface display'''], a method adapted from Boder and Wittrup (1997) [3]. This method allowed us to analyze a subset of our constructs at 4 different antigen (HA) concentrations: 0,1, 10, and 100 nM. In this process, our mutant plasmids were first expressed in yeast and grown in SDCAA growth media. Next, cells were grown in SCGAA media which contained glucose and galactose, required for the activation of the galactose inducible promoter on the yeast plasmid. This allows the mutant protein to be expressed on the yeast surface. All yeast cell samples were standardized to an OD600 of 2, and then labeled using different amounts of a single type of biotinylated antigen (HA). After an incubation step, the samples were | + | To test our designs, we [https://2012.igem.org/Team:Washington/Protocols/Yeast '''transformed'''] our mutants into the organism ''Saccharomyces cerevisiae'' in order to perform surface display and binding efficiency analysis. We then used [https://2012.igem.org/Team:Washington/Protocols/Display '''yeast surface display'''], a method adapted from Boder and Wittrup (1997)<html><a href="#Sources"><font size="1"><sup>[3]</sup></font></a></html>. This method allowed us to analyze a subset of our constructs at 4 different antigen (HA) concentrations: 0,1, 10, and 100 nM. In this process, our mutant plasmids were first expressed in yeast and grown in SDCAA growth media. Next, cells were grown in SCGAA media which contained glucose and galactose, required for the activation of the galactose inducible promoter on the yeast plasmid. This allows the mutant protein to be expressed on the yeast surface. All yeast cell samples were standardized to an OD600 of 2, and then labeled using different amounts of a single type of biotinylated antigen (HA). After an incubation step, the samples were washed to remove unbound antigen. The samples were then labeled with Streptavidin-PE (to monitor binding of antigen, see in red in the diagram below) and anti-cymc FITC (to monitor surface expression, seen in green in the diagram below). |
<br> | <br> | ||
| - | We then analyzed the fluorescent signature of the yeast cells using flow cytometery. Flow cytometry beams a laser into a stream of the samples containing the mutant and uses fluorescence detectors to measure the volume of fluorescently labeled cells. This generates graphs of the mean PE fluorescence of the surface displaying the population of cells as a function of antigen | + | We then analyzed the fluorescent signature of the yeast cells using flow cytometery. Flow cytometry beams a laser into a stream of the samples containing the mutant and uses fluorescence detectors to measure the volume of fluorescently labeled cells. This generates graphs of the mean PE fluorescence of the surface, displaying the population of cells as a function of antigen. The KD is calculated that fits to that data assuming 1:1 binding stoichiometry. |
| Line 84: | Line 77: | ||
<h1 id='Results'>Results Summary <html><a href="#Background"><font size="3">[Top]</font></a></html></h1> | <h1 id='Results'>Results Summary <html><a href="#Background"><font size="3">[Top]</font></a></html></h1> | ||
| - | We were able to design and test 6 HB36.5 variants and 14 HB80.4 variants. Including the starting designs we tested over 150 binder/HA combinations (see below spreadsheet). The results showed that most of the suggested mutations we found did not change the binding affinity to the different hemagglutinins. However, we did find 7 variants (listed below the table), that had an effect on binding. Our most dramatic results were with HB36.5 H312A, which was able to bind H2HA strongly where the original design variant could not. H2HA is particularly important as it has not been seen in humans since 1958. As it is known to still circulate in birds and has the potential to cross back over into humans it can be considered a future pandemic strain. We hope that our mutations to HB36.5 will be useful in a diagnostic that can rapidly test for the presence of H2 hemagglutinin which would be useful in global health monitoring. We have not been able to find any other protein that can differentiate between H1(the most common) and H2HA and believe we have created the first such example. | + | We were able to design and test 6 HB36.5 variants and 14 HB80.4 variants. Including the starting designs, we tested over 150 binder/HA combinations (see below spreadsheet). The results showed that most of the suggested mutations we found did not change the binding affinity to the different hemagglutinins. However, we did find 7 variants (listed below the table), that had an effect on binding. Our most dramatic results were with HB36.5 H312A, which was able to bind H2HA strongly where the original design variant could not. H2HA is particularly important as it has not been seen in humans since 1958. As it is known to still circulate in birds and has the potential to cross back over into humans, it can be considered a future pandemic strain. We hope that our mutations to HB36.5 will be useful in a diagnostic that can rapidly test for the presence of H2 hemagglutinin which would be useful in global health monitoring. We have not been able to find any other protein that can differentiate between H1(the most common) and H2HA and believe we have created the first such example. |
[[Image:UW FLU HB36 results graph.png|border|800px|center|thumb|HB36.5 and its variants tested against hemagglutinin subtype 2 at four different concentrations. The binding signal was determined by taking the geometric mean of a labeled population of 25,000 yeast cells at three different HA concentrations. The H312A mutation shows particularly strong binding signal at 100nM. ]] | [[Image:UW FLU HB36 results graph.png|border|800px|center|thumb|HB36.5 and its variants tested against hemagglutinin subtype 2 at four different concentrations. The binding signal was determined by taking the geometric mean of a labeled population of 25,000 yeast cells at three different HA concentrations. The H312A mutation shows particularly strong binding signal at 100nM. ]] | ||
| Line 93: | Line 86: | ||
[[Image:UWashington HB36cytomutant.jpg|border|500px|center|thumb|HB36.5 flu binder tested using Yeast surface display at 100 nM H2 on the left, H312A mutant tested on the right at 100 nM H2 on the right. HB36.5 Shows little to no binding at this HA concentration. H312A dramatically increases binding to H2.]] | [[Image:UWashington HB36cytomutant.jpg|border|500px|center|thumb|HB36.5 flu binder tested using Yeast surface display at 100 nM H2 on the left, H312A mutant tested on the right at 100 nM H2 on the right. HB36.5 Shows little to no binding at this HA concentration. H312A dramatically increases binding to H2.]] | ||
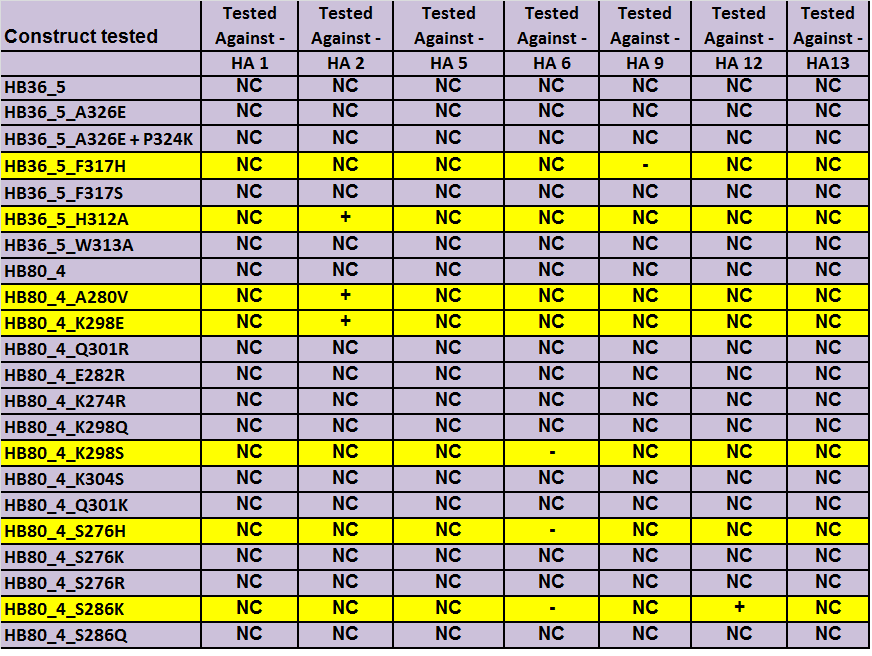
| - | [[Image:Results All .png|border|500px|center|thumb|Summary of the results, NC (No changes for the binding), + (increased binding), - (decreased binding)]] | + | [[Image:Results All .png|border|500px|center|thumb|Summary of the results, NC (No changes for the binding), + (increased binding), - (decreased binding). Each box represents an analysis of testing at 4 HA concentrations in comparison to the original starting binder designs.]] |
HB36.5_F317H was predicted to increase HA5 binding but it decreased the binding to HA9. Nevertheless, it can be used for specific hemagglutinin subtypes binder. | HB36.5_F317H was predicted to increase HA5 binding but it decreased the binding to HA9. Nevertheless, it can be used for specific hemagglutinin subtypes binder. | ||
Latest revision as of 03:54, 4 October 2012
“For the first time in history we can track the evolution of a pandemic in real time. Influenza viruses are notorious for their rapid mutation and unpredictable behavior.”--Margaret Chan, WHO Director General
Background

Influenza, also known as the flu, is a deadly virus that has decimated large populations of various species including humans. Even with the advent of vaccinations, high rates of mortality still occur because of the constant evolution of the virus. Variations within each strain of influenza are due to the different proteins that coat the virus' surface.
One such protein is Hemagglutinin (HA), which is represented by the H when describing flu subtypes (H1N1, H2N3, etc). Hemagglutinin is primarily involved in viral transfer as it mediates the binding and entry into a host cell through a sialic acid linkage. Each strain of Influenza is characterized by it's variant of Hemagglutinin. There are 16 known subtypes of Hemagglutinin which give rise to the multitude of strains that exist to today and usually only differ by a relatively few number of amino acid mutations. The World Health Organization uses the history of influenza breakouts to predict upcoming strains, which threaten to cause future pandemics, and vaccinations are created using these predictions.
In 2011, Fleishman et al., detailed two small proteins which had binding capabilities to various subtypes of hemagglutinin. In 2012, Whitehead et al., optimized the flu binders and showed that HB80.4 was broadly binding, meaning it bound to multiple HAs with high affinity. They also showed that HB36.5 bound preferentially to HA 1.
We aimed to further optimize the binding of HB80.4 to the group 1 HAs, but knowing that HB80.4 was already broadly binding to many HAs, we wanted to focus our efforts on the flu binder HB36.5 which until had not yet been tested against any other HAs is group 1. Therefore, we tested HB36.5 against the other group 1 HAs and assessed its native binding capabilities. After testing, we discovered that in addition to HA1, HB36.5 bound to H5, H6, H9 and H13. Binding was not observed to HA2 or HA12 subtypes however. Through application based design using FoldIt and real world testing we were able to find multiple single amino acid mutations to the binders that improved binding to the flu subtypes HA2, HA6, and HA9. Having two broadly-binding flu binders, with subtype specific capabilities would allow for enhanced rapid and reliable global tracking of current and future Influenza strains.
Foldit - A protein folding application[Top]
To design our binder mutants we employed the purpose-built application (app), [http://fold.it/portal/ FoldIt]. This program enables you to view a desired protein complex and score conformational or mutational changes to the structure. FoldIt also indicates potential issues within a protein by indicating clashes between resides and offers many options of resolving it. Clashing residues, for example, can be manually resolved by mutating a particular amino acid to a different one, the effect of your choice of mutation will then be evaluated immediately and given a score. This program was employed in designing mutants of the native binders that would bind better to the different flu subtypes. By utilizing the computational application Foldit, we were able to design many mutants and then quickly assess whether or not they would produce the desired binding affect.
Methods [Top]
Design: Method of computational design
Gathering information about the structures of Hemagglutinin
Since we wanted to improve the binding of HB80.4 and HB36.5 to specific HAs, we had to understand the structural differences of the many subtypes of hemagglutinin. Initially, we searched for specific hemagglutinin structure on the [http://www.rcsb.org/pdb/home/home.do Protein Data Bank] (PDB). If we found a desired hemagglutinin structures, we downloaded the associated PDB file and made a model of the binder-HA complex in Pymol. These models would then later be used in [http://fold.it/portal/ Foldit].
If we could not find the exact structure of a particular hemagglutinin, we then searched for the protein sequence of that hemagglutinin on the National Center for Biotechnology Information (www.ncbi.nih.gov). We then aligned the protein sequence of the structurally unknown hemagglutinin we obtained to that of the H1 HA (which has a known structure) by using [http://www.ebi.ac.uk/Tools/msa/clustalw2/ Clustalw2]. Next, we created homology models in FoldIt of the desired hemaggluttinin by changing the side chains of the H1 HA in Foldit according to the protein sequence differences we found.
Using the computational application Foldit to design our models
Crystal stuctures of HB36.3 and HB80.4 in complex with H1HA are available on the PDB as PDB ID 3R2X and 4EEF, respectively. To create models of the binder bound to different HAs, we used PYMOL to align our alternative HA structures to the H1HA from the published crystal structures. Then we would record the difference in side chains between that specific hemagglutinin and H1HA and use FoldIt to implement those changes on the known crystal structures. The models we created are linked below:
| File:HB80 H12 model.zip | File:HB80 H9 model.zip | File:HB80 H5 model.zip | File:HB36 H13 model.zip | File:HB36 H12 model.zip |
|---|
| File:HB36 H9 model.zip | File:HB36 H6 model.zip | File:HB36 H5 model.zip | File:HB36 H2 model.zip |
|---|
Proposing Mutations based on FoldIt Models
After preparing the FoldIt model, we proposed mutations on the original flu binder in order to improve its binding to the desired hemagglutinin. There is a score total in FoldIt which indicates the energy of the whole protein structure. The protein structure is more stable when its energy is lower. Thus, our primary goal was to decrease the total score in FoldIt. We did that in three different ways, filling the holes, making space, and balancing the electrostatic interactions. Holes that did not exist in H1HA would be created with the replacement of different side chains. We would mutate the corresponding side chain on the flu binder so that it extends out to fill a hole in the hemagglutinin. Making space is the exact opposite of filling the holes. Some side chains of certain hemaggluttinins are more projected out in space than that of hemagluttinin one. Therefore, we would mutate the corresponding side chains of the flu binder in order to provide room for the more extended side chains. Some variations of side chains on hemaggluttinin cause electrostatic changes. We would mutate the corresponding side chains of the flu biunder so as to counter balance the electrostatic changes. For example, if a certain area on the helix is changed to be more electronegative, we would introduce an electropositive side chain on the flu binder for improving the binding and vice versa.
Build: Kunkel Mutagenesis - Mutagenizing HB80.4 and HB36.5 flu binders
Kunkel mutagenesis is a classic procedure for incorporating targeted mutations into a plasmid and we used it to create many variants of the original flu binders.
The proteins tested were encoded on the shuttle vector pETCON which contained the yeast display components and was compatible with expression in both E. Coli (bacteria) and S. Cerevisiae (Yeast). This allowed us to do Kunkel Mutagenesis in E. Coli and not have to switch vectors when we transformed into yeast for Yeast Surface Display testing.
The first step to producing our specially designed flu binders was to change the original flu binding protein with our desired amino acid substitutions. We designed mutagenic oligonucleotide primers that would anneal to the original flu binding protein and incorporate point mutations that, when expressed, would result in a variant of the binder with the desired amino acid alteration. To incorporate these mutations, we first isolated single stranded DNA (ssDNA) of our vector harboring the original binding sequence. To do this we infected cells with bacteriophage M13, which packages its own ssDNA genome identified by length, and so in tandem packaged our vector in single stranded form. We then harvested the phage from the lysed culture of E. Coli, and extracted our single stranded vector DNA. Next, we annealed and extended our mutagenic oligos to incorporate the specified mutations into the newly synthesized antisense strand. This hybrid vector was transformed into E. Coli which degraded the original DNA and replaced it with sections complementary to the mutagenized strand.
Test: Yeast Surface Display
To test our designs, we transformed our mutants into the organism Saccharomyces cerevisiae in order to perform surface display and binding efficiency analysis. We then used yeast surface display, a method adapted from Boder and Wittrup (1997)[3]. This method allowed us to analyze a subset of our constructs at 4 different antigen (HA) concentrations: 0,1, 10, and 100 nM. In this process, our mutant plasmids were first expressed in yeast and grown in SDCAA growth media. Next, cells were grown in SCGAA media which contained glucose and galactose, required for the activation of the galactose inducible promoter on the yeast plasmid. This allows the mutant protein to be expressed on the yeast surface. All yeast cell samples were standardized to an OD600 of 2, and then labeled using different amounts of a single type of biotinylated antigen (HA). After an incubation step, the samples were washed to remove unbound antigen. The samples were then labeled with Streptavidin-PE (to monitor binding of antigen, see in red in the diagram below) and anti-cymc FITC (to monitor surface expression, seen in green in the diagram below).
We then analyzed the fluorescent signature of the yeast cells using flow cytometery. Flow cytometry beams a laser into a stream of the samples containing the mutant and uses fluorescence detectors to measure the volume of fluorescently labeled cells. This generates graphs of the mean PE fluorescence of the surface, displaying the population of cells as a function of antigen. The KD is calculated that fits to that data assuming 1:1 binding stoichiometry.
Results Summary [Top]
We were able to design and test 6 HB36.5 variants and 14 HB80.4 variants. Including the starting designs, we tested over 150 binder/HA combinations (see below spreadsheet). The results showed that most of the suggested mutations we found did not change the binding affinity to the different hemagglutinins. However, we did find 7 variants (listed below the table), that had an effect on binding. Our most dramatic results were with HB36.5 H312A, which was able to bind H2HA strongly where the original design variant could not. H2HA is particularly important as it has not been seen in humans since 1958. As it is known to still circulate in birds and has the potential to cross back over into humans, it can be considered a future pandemic strain. We hope that our mutations to HB36.5 will be useful in a diagnostic that can rapidly test for the presence of H2 hemagglutinin which would be useful in global health monitoring. We have not been able to find any other protein that can differentiate between H1(the most common) and H2HA and believe we have created the first such example.

HB36.5_F317H was predicted to increase HA5 binding but it decreased the binding to HA9. Nevertheless, it can be used for specific hemagglutinin subtypes binder.
HB36.5_H312A was predicted to increase HA2 binding and it did improve binding to HA2.
HB80.4_A280Vwas predicted to decrease HA2 binding but it improved the binding to HA2.
HB80.4_K298E was predicted to increase HA2, HA5, HA6 and HA12 binding and it did improve binding to HA2.
HB80.4_S276H was predicted to increase HA6 binding but it decreased the binding to HA6.
HB80.4_K298S was predicted to increase HA12 binding but it decreased the binding to HA6.
HB80.4_S286K was predicted to increase HA12 binding and it increased the binding to HA12. However, it decreased the binding to HA6.
Future Directions [Top]
We intend to combine the HB36.5 single-point mutants H312A and W313A, both successfully having increased binding to HA2, to create a superior combinatorial mutant with multiple-fold improvement. Simultaneously, we intend to create a mutant of HB36.5 that will bind H12 preferentially, as HB36.5 does not currently do so. After obtaining a version of HB36.5 that binds to HA12 more efficiently, a diagnostic system can be made to track the types of hemagglutinin on the surface of influenza. Specifically, this system can identify HA2 and HA12 from the 16 subtypes of hemagglutinin.
Parts Submitted [Top]
We have submitted six parts to the registry. HB80.4, HB36.5 and four of our most efficacious mutants. A short description of each part is provided below.
[http://partsregistry.org/wiki/index.php/Part:BBa_K892401 BBa_K892401: Starting HB36.5]
The starting HB36.5 flu binder originally shown to bind to H1 by Whitehead et al.
[http://partsregistry.org/wiki/index.php/Part:BBa_K892402 BBa_K892402: HB36.5_F317S]
A mutated HB36.5 flu binder aimed to increase binding to H2, a subtype of hemagglutinin on the surface of the Influenza virus. This mutant has a point mutation at residue 317 from Phenylalanine to Serine
[http://partsregistry.org/wiki/index.php/Part:BBa_K892403 BBa_K892403: HB36.5_H312A]
A mutated HB80.4 flu binder aimed to increase binding to H2, a subtype of hemagglutinin on the surface of the Influenza virus. This mutant has a point mutation at residue 312 from Histidine to Alanine, which dramatically increases the binding to H2 over the native HB36.5 binder.
[http://partsregistry.org/wiki/index.php/Part:BBa_K892404 BBa_K892404: HB36.5_A326E]
A mutated HB36.5 flu binder aimed to increase binding to H2, a subtype of hemagglutinin on the surface of the Influenza virus. This mutant has a point mutation at residue 326 from Alanine to Glutamate
[http://partsregistry.org/wiki/index.php/Part:BBa_K892405 BBa_K892405: Starting HB80.4]
The starting HB80.4 flu binder originally shown to bind to various subtypes of Hemagglutinin in group 1. Originally identifited by Fleishman et al., further optimized by Whitehead et al.
[http://partsregistry.org/wiki/index.php/Part:BBa_K892410 BBa_K892410: HB36.5_F317H]
A mutated HB36.5 flu binder aimed to increase binding to H2, a subtype of hemagglutinin on the surface of the Influenza virus. This mutant has a point mutation at residue 317 from Phenylalanine to Histidine
References[Top]
[1] Whitehead, Timothy, et al. "Optimization of affinity, specificity and function of designed influenza inhibitors using deep sequencing." Nature Biotechnology 30.6 (2012):543-548.
[2] Fleishman, Sarel, et al."Computational design of proteins targeting the conserved stem region of influenza hemagglutinin." Science 332.6031 (2011): 816-821.
[3] Boder, Eric and Wittrup, Dane. "Yeast surface display for screening combinatorial polypeptide libraries." Nature Biotechnology 15.6 (1997):553-557.
 "
"