Team:SDU-Denmark/labwork/Notebook
From 2012.igem.org
| (7 intermediate revisions not shown) | |||
| Line 292: | Line 292: | ||
<p> <b>02-07-2012 to 08-07-2012</b> </p> | <p> <b>02-07-2012 to 08-07-2012</b> </p> | ||
| - | + | <p>The first week in lab started shortly after our exams. We had a goal, but were still uncertain about how to get there. We needed to get our hands on the coding sequence for a naturaly occouring inulin producing unit and there were several possible ways to get it.</br> | |
| - | <p> | + | We could synthesise it, isolate it from a plant with a suitible kit or something less handy. Since the iGEM project is a learning experience to us, we chose something less handy |
| - | + | and designed our own experiment to isolate the mRNA from a suitable plant, using only the equipment and materials our lab had acces to from the beginning. <br/> | |
| - | + | After having sought out information on the net, we decided on the plant Helianthus Tuberosus, also known as Jerusalem artichoke, to be the source of the coding sequence(s). Helianthus Tuberosus has a two-part inulin synthesis consisting of 1-SST and 1-FFT. Both genes are needed for the complete synthesis and thus both needed to be aquired. <br/> | |
| - | + | During the summer, June-July-August, the Helianthus Tuberosus is growing and producing alot of inulin in the tubers of the plant. We took advantage of that and used our newly developed plant mRNA isolation protocol to isolate mRNA from the tubers.<br/> | |
| - | + | The process involved cutting up the tubers, flash-freezing with liquid nitrogen to halt RNase activity, which is VERY important when isolating RNA. Following, the plant material was grinded into fine dust while kept frozen to keep the plant cell walls rigid in an attempt to destroy them and release the mRNA and dissolved in RTL buffer. <br/> | |
| - | + | The solution was treated with ultrasound to homogenize it to further disrupt the remaining cell wall. Cell wall and larger organelles were pelleted by centrifuge. From here on a mRNA isolation kit for mamalian cells was used to extract the RNA from the remaining solution.<br/> | |
| - | + | See *mRNA isolation protocol* for further details.<br/> | |
| - | The now isolated mRNA solutions were measured on a | + | Using reverse transcriptase we were able to convert the isolated mRNA to cDNA.<br/><br/> |
| - | < | + | |
| - | Control: 627 ng/uL <br/> | + | The now isolated mRNA solutions were measured on a Nanodrop to ascertain their concentrations:<br/> |
| - | Sonic: 747 ng/uL <br/> | + | |
| - | <p> | + | Control: 627 ng/uL<br/> |
| + | Sonic: 747 ng/uL <br/><br/> | ||
| + | |||
| + | Furthermmore we designed primers that would anneal to the ends of the 1-SST and 1-FFT coding sequences, with the help of Steffen Smidth, and ordered them home.<br/> | ||
| + | |||
| + | <center><img src="https://static.igem.org/mediawiki/igem.org/a/ae/IGEM098.jpg" width="100%" /></center> | ||
| + | |||
| + | |||
| + | |||
| + | </p> | ||
Latest revision as of 02:03, 27 September 2012














Laboratory Notebook
Here you will find the log book for the procedures carried out in the laboratory, starting from week 27.02-07-2012 to 08-07-2012
The first week in lab started shortly after our exams. We had a goal, but were still uncertain about how to get there. We needed to get our hands on the coding sequence for a naturaly occouring inulin producing unit and there were several possible ways to get it.
We could synthesise it, isolate it from a plant with a suitible kit or something less handy. Since the iGEM project is a learning experience to us, we chose something less handy
and designed our own experiment to isolate the mRNA from a suitable plant, using only the equipment and materials our lab had acces to from the beginning.
After having sought out information on the net, we decided on the plant Helianthus Tuberosus, also known as Jerusalem artichoke, to be the source of the coding sequence(s). Helianthus Tuberosus has a two-part inulin synthesis consisting of 1-SST and 1-FFT. Both genes are needed for the complete synthesis and thus both needed to be aquired.
During the summer, June-July-August, the Helianthus Tuberosus is growing and producing alot of inulin in the tubers of the plant. We took advantage of that and used our newly developed plant mRNA isolation protocol to isolate mRNA from the tubers.
The process involved cutting up the tubers, flash-freezing with liquid nitrogen to halt RNase activity, which is VERY important when isolating RNA. Following, the plant material was grinded into fine dust while kept frozen to keep the plant cell walls rigid in an attempt to destroy them and release the mRNA and dissolved in RTL buffer.
The solution was treated with ultrasound to homogenize it to further disrupt the remaining cell wall. Cell wall and larger organelles were pelleted by centrifuge. From here on a mRNA isolation kit for mamalian cells was used to extract the RNA from the remaining solution.
See *mRNA isolation protocol* for further details.
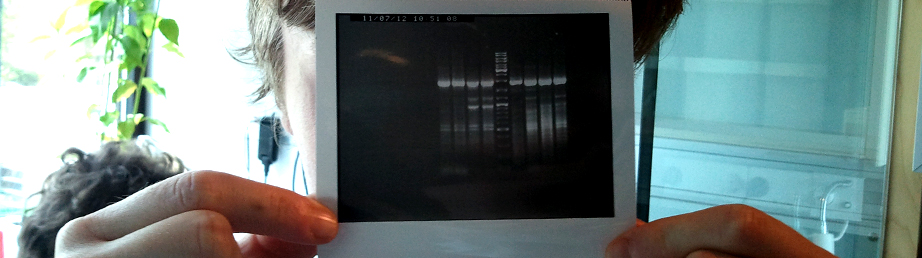
Using reverse transcriptase we were able to convert the isolated mRNA to cDNA.
The now isolated mRNA solutions were measured on a Nanodrop to ascertain their concentrations:
Control: 627 ng/uL
Sonic: 747 ng/uL
Furthermmore we designed primers that would anneal to the ends of the 1-SST and 1-FFT coding sequences, with the help of Steffen Smidth, and ordered them home.

 "
"