Membrane Rudder
- Violacein & Deoxyviolacein synthetic pathway
Dynamically and artificially regulating the direction of biochemical pathway in vivo has remained a challenge for scientists. We are now to achieve this goal through controlling the aggregation state of different enzymes in branched biological reactions based on Membrane Scaffold. We named this universal device Membrane Rudder.
Violacein is a purple chromobacterial pigment synthesized from the basic amino acid tryptophan. Its biosynthetic pathway contains 3 branches and produces 3 final products, which are violacein, deoxyviolacein and deoxychromoviridans respectively. Thus it is incorporated to test the feasibility of Membrane Rudder
To switch the direction of Violacein and deoxyviolacein synthetic pathway through light signal
To switch the direction of Violacein and deoxyviolacein synthetic pathway through RNA signal
To justify the universality of Membrane Rudder
Successfully switched the direction of Violacein synthetic pathway by light signal
Decreased yields of side product deoxychromaviridans by 8-fold with Membrane Rudder compared to group with free cytoplasmic enzyme
Constructed a device that connects post-translational Membrane Scaffold system to genetic circuits by recruiting RNA as a controlling signal.
Background
Dynamically and artificially regulating the direction of biochemical pathway in vivo has remained a challenge for scientists. Based on Membrane Scaffold, we are now trying to achieve this goal through controlling the aggregation state of different enzymes in branched biological reactions. We named this universal device Membrane Rudder. Theoretically this device could sense a large amount of signals. We chose blue light and rationally designed RNA molecule D0 as controlling signal. Branched violacein & deoxyviolacein synthetic pathway is recruited to verify the feasibility of Membrane Rudder.
There are three branches in violacein & deoxyviolacein synthetic pathway. We could identify amount of final products through high-performance liquid chromatography (HPLC). The change of products proportion will demonstrate the efficiency of direction alteration.
Violacein & Deoxyviolacein Biosynthetic Pathway
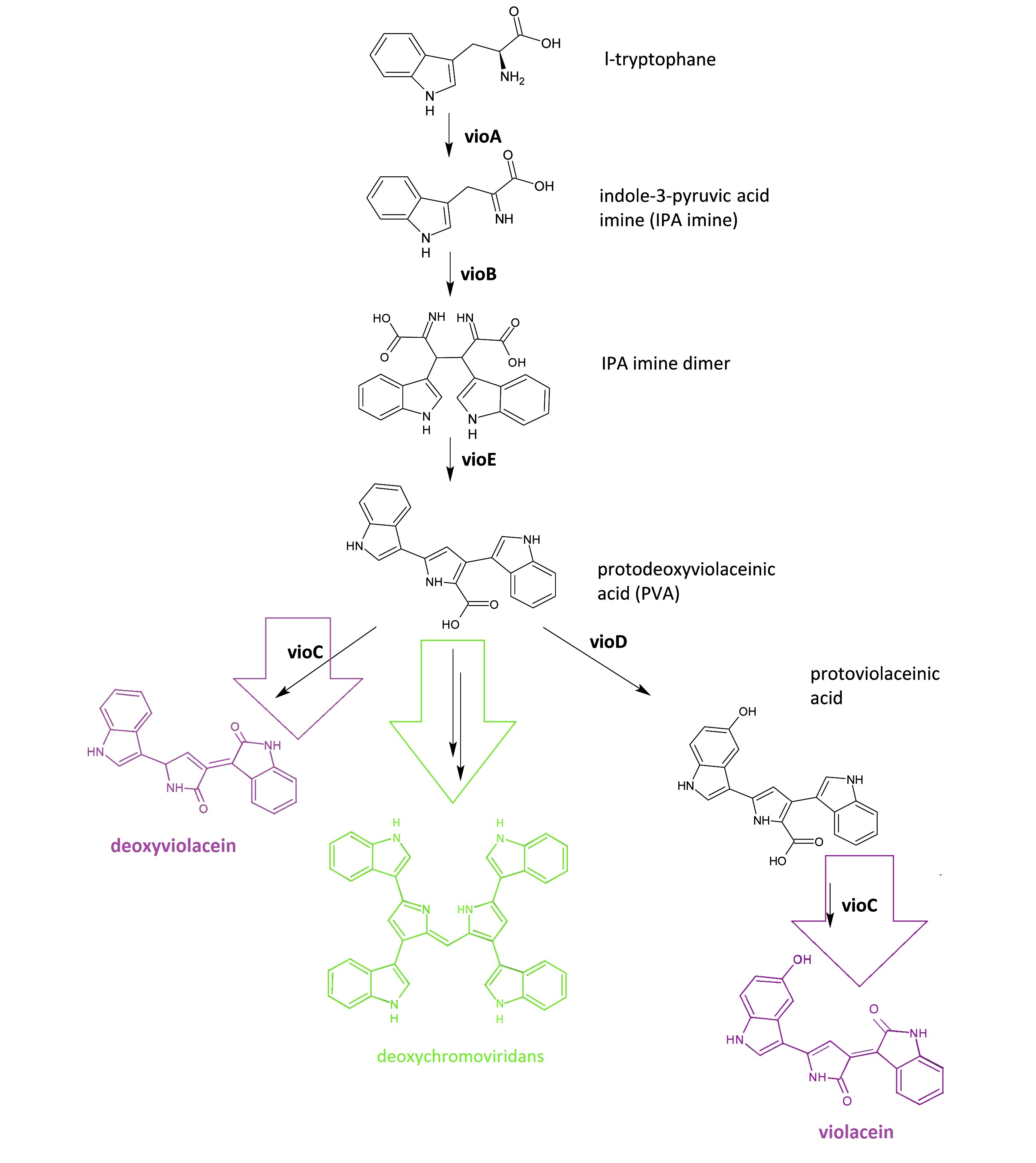 Fig.1: :Details of violacein & deoxyviolacein biosynthetic pathway. The left pathway painted in purple indicates the synthetic pathway of purple pigment deoxyviolacein.The right pathway painted in violet indicates the synthetic pathway of purple pigment violacein. The green pathway indicates the nonenzymatic production of green pigment deoxychromoviridans.
The violacein & deoxyviolacein biosynthetic pathway includes 5 key enzymes which work in sequence: Vio A, B, C, D and E. VioA, a flavoenzyme and VioB, a heme protein, work together to oxidize and dimerize tryptophen into IPA imine dimer. Then VioE induces an indole rearrangement, producing prodeoxyviolacein acid, also known as PVA. In a word, enzyme VioA, VioB and VioE convert tryptophen into PVA. Then this biosynthetic pathway is about to branch.
In E.coli, an additional side reaction could occur, and PVA is converted into a green pigment called deoxychromoviridans.
The last two enzymes, VioC and VioD are flavin-dependent oxygenases. VioC alone transforms PVA into a purple pigment called deoxyviolacein. VioC could also act cooperatively with VioD. VioD hydroxylates 5-position indole ring, and then the other 2-position indole ring is processed by VioC to create the oxindole. In this way, violacein is produced.
Design of Experiment
We tried to create Membrane Rudder controlled by light signal and RNA signal to switch direction of biochemical reaction dynamically and artificially in E.coli.
Membrane Rudder Sensing Light Signal
Light is an intriguing signal to regulate E.coli activity because it is easy to obtain, highly tunable and nontoxic. A metabolite-coupled light-switchable system could be quite fascinating.
We constructed our device as demonstrated in Fig.2. In light-sensing Membrane Rudder, VioA, VioB, VioE and VioC with interacting Membrane Anchors constitutively aggregate. VVD, a photoreceptor of Neurospora crassa, can form dimer in the presence of blue light and disassociate as light is off. When blue light is present, VioD with VVD-coupled Membrane Anchor would aggregate with assembly of VioA, B, C and E, making violacein the dominant final product. But when blue light is off, VioD with VVD coupled Membrane Anchor will disassociate with VioA, B, C and E. Thus the biosynthetic pathway for deoxyviolacein is switched on.
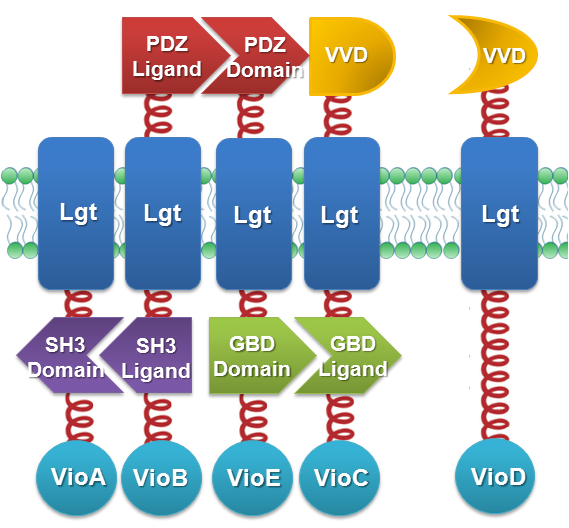 Fig.2 :Construction details of light-sensing Membrane Rudder. VVD acts as a blue light sensor. For VioB and VioE only function normally in dimereic state, free VioB and free VioE were coexpressed with membrane anchored VioA, B, C, D and E to ensure the normal function of the whole system.
Bacteria in experimental group were induced at a L-Arabinose concentration of 0.1%. One group of bacteria expressing full set of light-sensing Membrane Rudder are incubated under blue light. The other group of bacteria expressing full set of light-sensing Membrane Rudder are incubated in the dark.
The BioBrick Part VVD-MA5-vioC and VVD-MA6-vioD is [http://partsregistry.org/Part:BBa_K771205 Part:BBa_K771205] and [http://partsregistry.org/Part:BBa_K771206 Part:BBa_K771206], respectively. For MA2-VioA, MA3-VioB, MA4-VioE, the corresponding part is [http://partsregistry.org/Part:BBa_K771201 Part:BBa_K771201], [http://partsregistry.org/Part:BBa_K771202 Part:BBa_K771202], [http://partsregistry.org/Part:BBa_K771203 Part:BBa_K771203], respectively.
Membrane Rudder Sensing RNA Signal
Membrane Accelerator and Membrane Rudder are both post-translation controlling device to regulate metabolic flux of the host cell. To connect this relatively isolated post-translation control system to genetic circuits, we employed RNA signal, which is present in cytoplasm. Rationally designed RNA D0 with MS2 and PP7 aptamer domain is recruited. When certain RNA molecule with two aptamer domains is present in cells, their cognate aptamer binding proteins can thus aggregate together. Furthermore, if we place RNA D0 (with PP7 and MS2 aptamer domains) under various promoters regulated by different signals, approaches to induce dimerization would be expanded sharply. Thus, Membrane Rudder could sense much more signals.
We constructed our device as demonstrated in Fig.3. In RNA-sensing Membrane Rudder, VioA, VioB, VioE and VioC with interacting Membrane Anchors constitutively aggregate. RNA D0, can aggregate with RNA aptamer binding protein MS2 and PP7. So when RNA D0 is present, VioD with MS2-coupled Membrane Anchor would aggregate with assembly of VioA, B, C and E, making violacein the dominant final product. But when RNA D0 is absent, VioD with MS2-coupled Membrane Anchor will disassociate with VioA, B, C and E. Thus the biosynthetic pathway for deoxyviolacein is switched on.
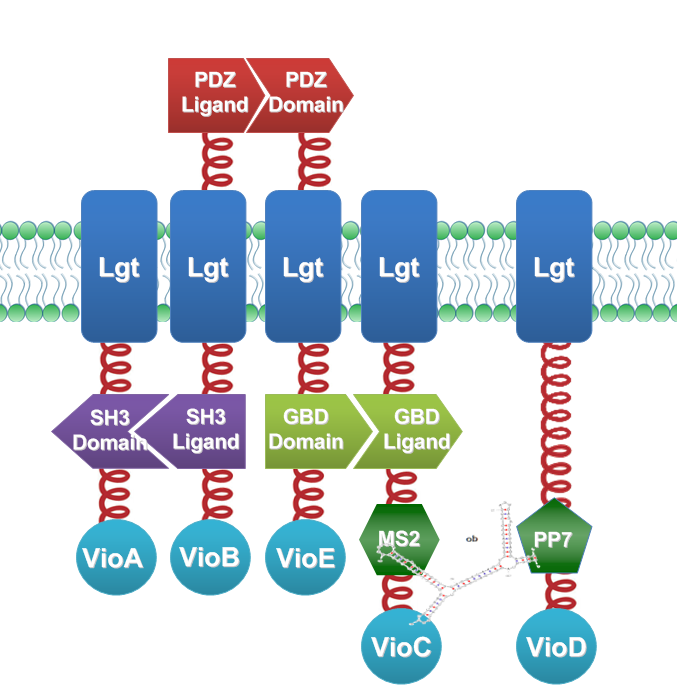 Fig.3 :Construction details of RNA-sensing Membrane Rudder. RNA aptamer binding protein MS2 and PP7 acts as an RNA sensor. Free VioB and free VioE were coexpressed with membrane anchored VioA, B, C, D and E to ensure the normal function of the whole system.
Bacteria in experimental group were induced at a L-Arabinose concentration of 0.1%. One group of bacteria expressing full set of light-sensing Membrane Rudder are incubated under blue light. The other group of bacteria expressing full set of light-sensing Membrane Rudder are incubated in the dark.
Results and Discussion
Membrane Rudder Sensing Light Signal
Overview
By attaching Enzymes VioA, B, C, D and E to Membrane Anchors that could sense blue signal, we managed to change the proportion of final products by switching on or off blue light signal. When blue light is on, VioA, B, C, D and E aggregate together, making violacein the dominant final product. When blue light is off, VioD with Membrane Anchor won't associate with VioA, B, C and E. Thus the biosynthetic pathway for deoxyviolacein is switched on.
Moreover, a significant decrease of side products such as deoxychomoviridans was observed when Membrane Rudder system was present.
Direction Alteration
The switch of the reaction can be realized by the extracellular signals. Through the light induction, the reaction producing deoxyviolacein by VioC alone is inhibited due to the lack of interaction with PVA. On the other hand, as long as light is restrained from the bacteria, however, the above reaction is initiated leading to the production of deoxyviolacein.
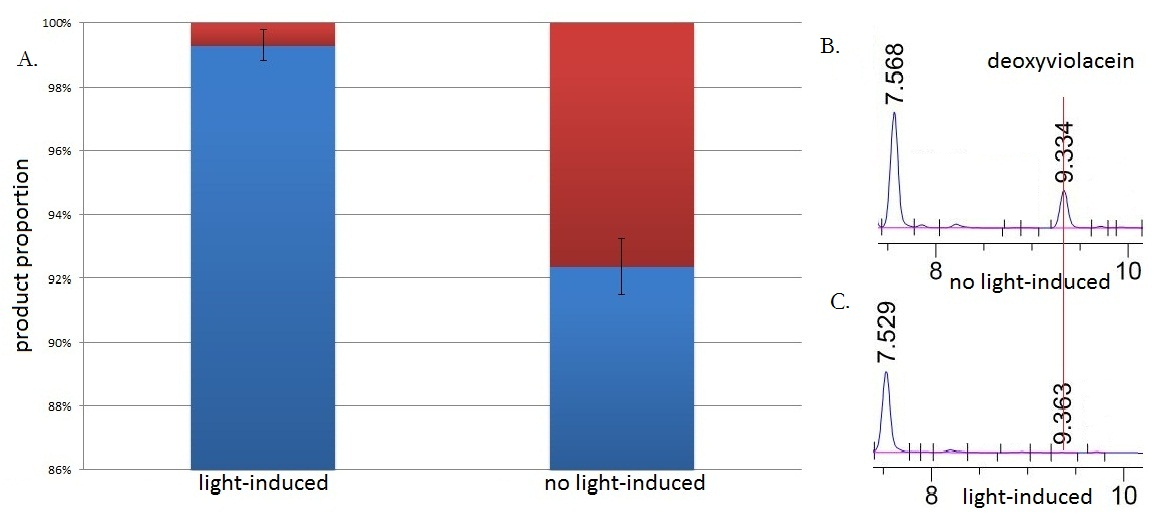 Fig.3 :HPLC result (right) and the ratio(left) of deoxyviolacein in sample with light induction and without light induction. Fig. B represents the deoxyviolacein HPLC peak results from sample with no light induction. Fig. C represents the deoxyviolacein HPLC peak results from sample with light induction. Results indicate that the amount of the deoxyviolacein from the sample without light induction is tremendously larger than that from the sample with light induction. The red part in the Fig.A represents the portion of deoxyviolacein and the blue part represents violacein We ran an HPLC (SHIMADZU LC-20AP,C18 reversed column) test with the purple samples extracted from the bacterium culture.Then we ran an mass spectrometry (Thermo Ultra GC-ISQ) test and verified the corresponding molecule role of every main peak.
The HPLC results show that the peak of deoxyviolacein appears at about 9 minutes after the injection. We find out that under the induction of light, production of deoxyviolacein was inhibited (to almost 0%) while most products were violacein. Without the light induction, however, the pathway leading to the deoxyviolacein was initiated and thus deoxyviolacein was produced(about 8%). Such results confirmed our membrane rudder system and indicated that by controlling the extracellular signal, such as light, we are able to manipulate the branched chain reactions, thus producing the target products we desire.
Decrease in Side Products
Membrane complex system would help to reduce the amount of side-products to some extent. Under the case that the enzymes involved in the side reaction are situated in the cytoplasm, they are less competent than the core enzymes attached on the membrane to mediate the subsequent reactions due to the spatial obstacle.
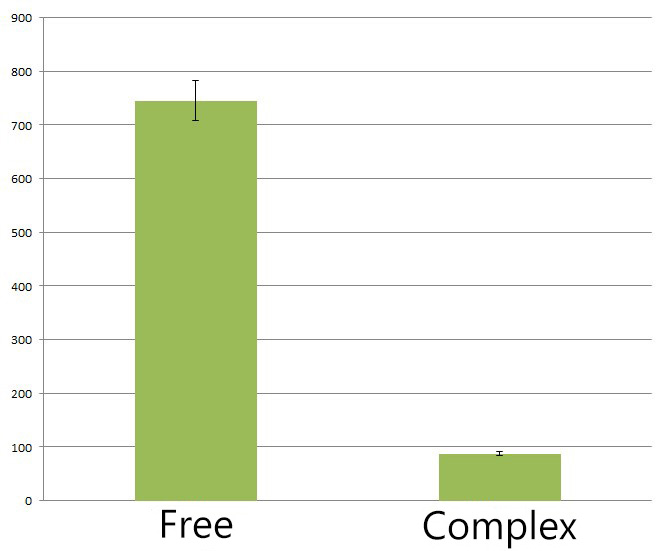 Fig.4 :The HPLC peak area of deoxychromoviridans from sample with light induction and free vioABCDE control group. From the HPLC results, we detected a distinct decrease of side-product, deoxychomoviridans. Judging from the peak area, we find out that the amount of deoxychromoviridans from rbs-vio control group is nearly eight times that from light-induced group. Such experiment results coincided with our anticipation and demonstrated that the membrane complex system would enhance the usage of substrates and thus reduce the amount of intermediate products and other subsequent side-products.
Reference
1. Balibar, C. J. and C. T. Walsh (2006). "In vitro biosynthesis of violacein from L-tryptophan by the enzymes VioA-E from Chromobacterium violaceum." Biochemistry 45(51): 15444-57.
2. Hoshino, T. "Violacein and related tryptophan metabolites produced by Chromobacterium violaceum: biosynthetic mechanism and pathway for construction of violacein core." Appl Microbiol Biotechnol 91(6): 1463-75.
3. Shrode, L. B., Z. A. Lewis, et al. (2001). "vvd is required for light adaptation of conidiation-specific genes of Neurospora crassa, but not circadian conidiation." Fungal Genet Biol 32(3): 169-81.
|