Project Overview
Advance in molecular cloning technology has made it possible for mankind to entitle engineered organisms to different biochemical reactions. However, the speed of those enzymatic reactions is often limited because intermediates produced from upstream enzyme cannot be passed efficiently to downstream enzyme due to spatial obstacles. Thus, synthetic scaffold built to decrease distance between enzymes for speeding biochemical reactions is a hot topic with promising application prospect.
Moreover, although some progress has been made in fields of reaction acceleration, no one has before succeeded in dynamically controlling direction of the biochemical pathway.
Introduction
In this year, we expanded the definition of scaffold in synthetic biology and developed two universal devices called Membrane Accelerator and Membrane Rudder respectively. Together,they made Membrane Magic happen!
Previous researchers have focused on building protein, RNA or DNA scaffold as constitutive assemblies carrying enzymes. They have succeeded in increasing product yields. However, the amount of those scaffolds could be limited by its expression or copy level, leading to restriction on further acceleration. With Membrane Magic, we made E.coli membrane into a huge scaffold accommodating enzymes without limitation of scaffold amount. Moreover, protein assembly on membrane could readily receive extracellular or intracellular signal, so the whole system becomes highly tunable.
One of our devices, called Membrane Accelerator, functions by localizing and organizing enzymes on membrane surface. E.coli inner membrane serves as a two-dimensional plane that can accommodate various protein assemblies linked with enzymes. Otherwise diffusing enzymes can form clusters on membrane through interacting protein domains and ligands. Enzyme clusters help substrates flow between enzymes, and thus increase yields of sequential biological reactions. We not only applied the Membrane Accelerator into biosynthetic pathway but also biodegradation pathway, which is proposed for the first time in synthetic biology. Previous researches on scaffold system all focused on biosynthesis.
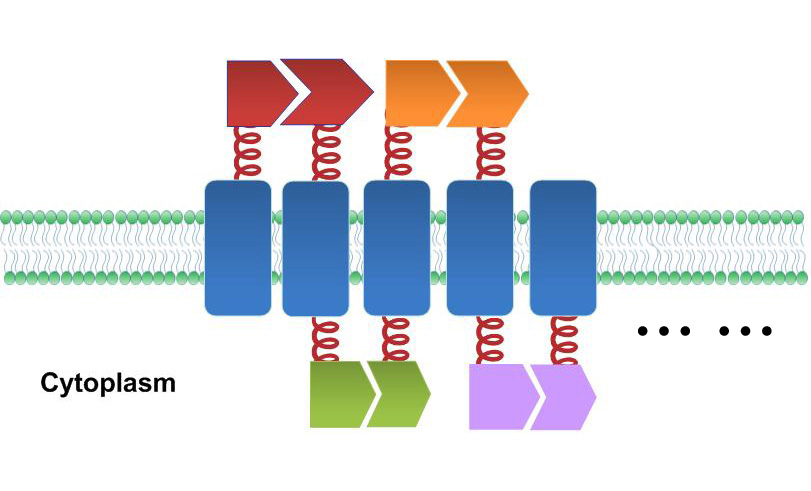 Fig.1: Sketch of Membrane Accelerator Although some work has been done in reaction acceleration, it has always been a challenge to artificially and dynamically control those reactions. Our Membrane Rudder device, however, offers a novel method to control the direction of biochemical reactions through varieties of signals. We further combined the whole post-translational control system with genetic circuits by recruiting RNA aptamer and its corresponding binding protein. Thus RNA signal could also be recruited to dynamically control biochemical reaction.
Fig.2: Sketch of Membrane Rudder
Why MEMBRANE?
Why do we choose membrane as our primary scaffold to assemble enzymes?
| 1. Natural Scaffold: Different from previous synthesized scaffold, membrane scaffold is an innate one. Besides, there is no limitation on scaffold amount.
|
|
| 2. Two-Dimensional Plane: Membrane Scaffold changes restricted the reaction space to a two-dimensional plane, which has been proved to accelerate reaction more sharply than one-dimensional and discrete scaffold.
| 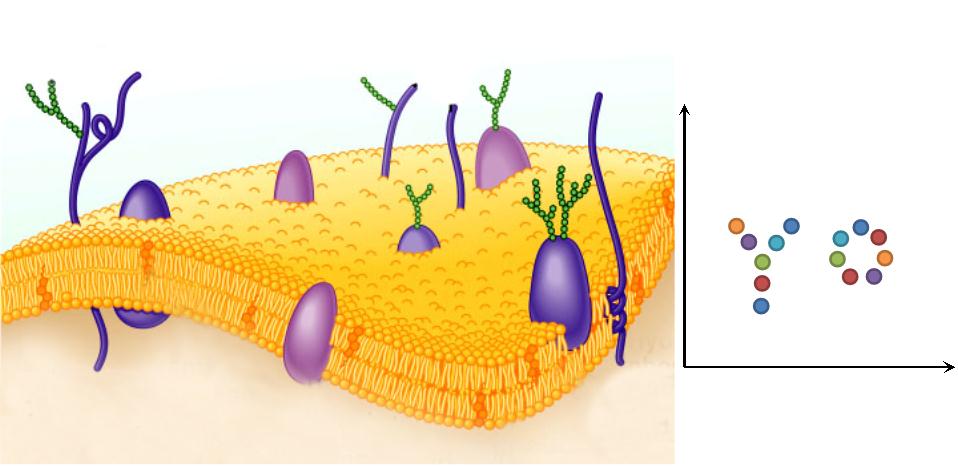 Fig.4: Two-Dimensional Plane |
| 3. Priority to Exportation: Concentration of final products could be effectively increased near the membrane with Membrane Scaffold, which in turn, facilitates the transmembrane transportation. So final products would be more readily to be exported to extracellular media.
| 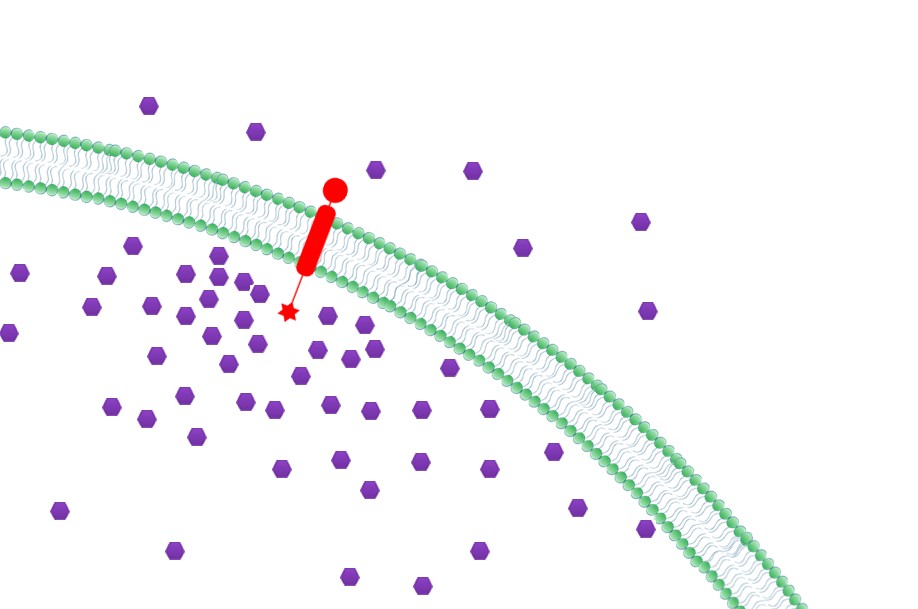 Fig.5: Priority to Exportation |
| 4. Ability to Sense Signals Membrane Scaffold provides a platform to directly receive environmental and internal signal. Thus reactions could be dynamically controlled through those signals.
| 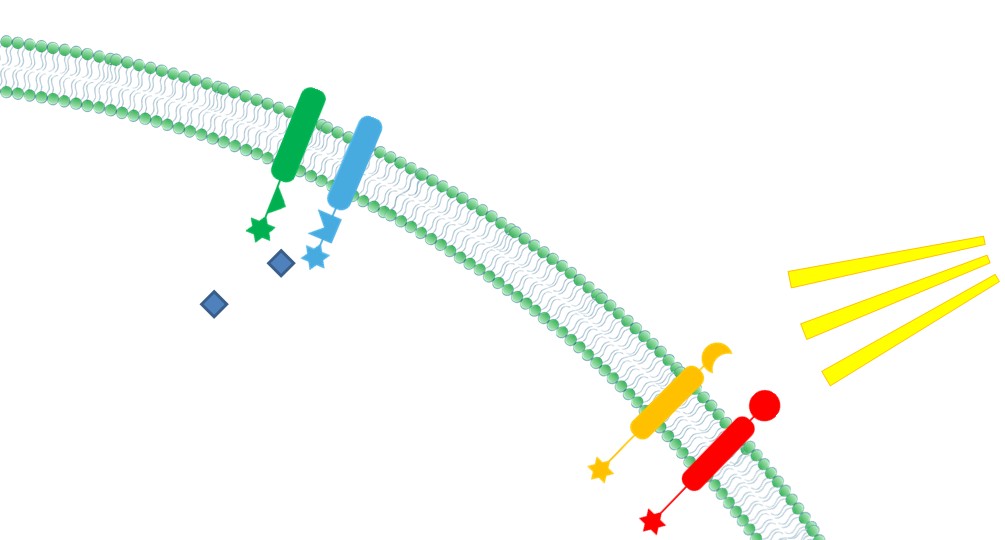 Fig.6: Ability to sense signals |
Reference
1.Delebecque, C. J., A. B. Lindner, et al. (2011). "Organization of intracellular reactions with rationally designed RNA assemblies." Science 333(6041): 470.
2.Dueber, J. E., G. C. Wu, et al. (2009). "Synthetic protein scaffolds provide modular control over metabolic flux." Nature biotechnology 27(8): 753-759.
3.Conrado, R. J., G. C. Wu, et al. (2012). "DNA-guided assembly of biosynthetic pathways promotes improved catalytic efficiency." Nucleic acids research 40(4): 1879-1889.
|