Team:NTU-Taida/PEPDEX
From 2012.igem.org
(→Stabilization Modules) |
(→2. Partition System:from Pseudomonas putidaKT2440) |
||
| Line 155: | Line 155: | ||
====2. Partition System:from '''Pseudomonas putida'''KT2440==== | ====2. Partition System:from '''Pseudomonas putida'''KT2440==== | ||
| - | : | + | :Bacterium distributes chromosomes/plasmids evenly to its progenies by a dynamic system that includes SMC like proteins, type Ia partition system and so on. Type Ia partition system segregates chromosomes/plasmids in a process akin to Eukaryotic mitosis. It can be found on most eubacteria. However, E. coli is not the case. |
[[FIle:NTU-Taida-Par2.png|700px|thumb|center]] | [[FIle:NTU-Taida-Par2.png|700px|thumb|center]] | ||
| - | :Previous study have shown that when provide parAB of P.putida in trans, | + | :Previous study have shown that when provide parAB of P.putida in trans and carrying parS site in cis, segregation of low copy plasmids (mini F) can be stabilized in E. coli.(Anne-Marie etl. 2002). The BioBrick vector pSB2K3 is a mini F plasmid, thus we expect its segregation can be stabilized in this way. Our team have made a modified version of pSB2K3 with parS site, and plan to use it to harbor our PepdEx system (a low copy number plasmid allows us to fine tune our systems controls e.g. with smaller number of operator sites). |
| + | |||
[[FIle:NTU-Taida-Par3.png|500px|thumb|center]] | [[FIle:NTU-Taida-Par3.png|500px|thumb|center]] | ||
Revision as of 16:14, 26 September 2012
Overall Project
Introduction
There has been a wide array of peptides, either innate or synthetic, used as drugs or vaccine in combat of different diseases. For instance, insulin was synthesized and mass produced in bacteria; long, synthetic peptide vaccine has also brought to bedside to help treat patient with invasive cancers. (Nat Rev Cancer. 2008 May;8(5):351-60.) However, the delivery of the peptide into human body is a big issue, since it cannot be administered orally, and has a poor distribution and absorption as compared to small molecules drugs in human body. With the advent of biotechnology and the growing forum of synthetic biology, we try to engineer and finely design different circuits in bacteria, which can deliver peptides through epidermal, buccal, rectal, or enteral. As for a simple exemplification of our ultimate goal of microbial peptide delivery system, we choose GLP-1, the endogenous hormone as our delivery model. Our design features efficiency, quick response to the environment changes, and sustainable release of GLP-1. This is the first model of our peptide delivery system, and opens a new room for the synthetic biology in medicine applications.
GLP-1, a human innate neuro-peptide for energy balance, is chosen to combat for obesity and metabolic syndrome. We engineer the non-pathogenic E. coli which senses fatty acids in intestines and secretes synthetic GLP-1. Appropriate signal peptides and penetratin are used to facilitate peptide secretion and intestinal uptake. Furthermore, we design a circuit with quorum sensing and double repressors, which aims to generate quick but sustainable responses and serves as an anti-noise filter. Plasmid stabilization modules including partition system and multimer resolution system are also incorporated to circumvent the undesirable loss or segregational instability of our artificial device. With this general concept of delivery of short peptide into human body, we can also target other human diseases with alternative circuit designs.
Circuit
The main circuit is designed to detect the presence of fatty acid in the intestinal environment and produce the peptide drug (GLP-1 in this case) and cell penetrating peptide (CPP) as response. There are two core systems: double repressors and quorum sensing.
Double Repressors
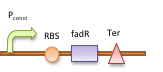
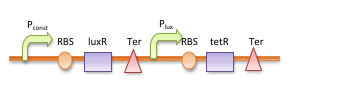
The double repressor system consists of the genes encoded Tet Repressor Protein (tetR) and lac reprresor (lacI). We put tetR downstream of fad promoter (Pfad) and lacI downstream of tet promoter (Ptet). The DNA sequence encoded GLP-1 and CPP are placed downstream of lac promoter (Plac).
In the absence of fatty acid, a constitutive expressed fatty acid metabolism regulator protein FadR binds to Pfad, which represses the transcription of tetR and makes Ptet free of TetR. Therefore LacI is expressed, binds to Plac and blocks the production of GLP-1 and CPP. After intake of a fat-rich meal, the fatty acid biosensor FadR is inhibited by the fatty acyl-CoA, which leads to the expression of TetR. The repression of the transcription of lacI by TetR frees the Plac from LacI and leads the final production of GLP-1 and CPP. When there is lactose in the intestinal environment (after drinking milk), the lactose can halt the repression of Plac directly by binding to LacI. GLP-1 and CPP can also be produced in this case.
Quorum Sensing
In order to amplify the response and to recruit other bacteria to work together when there are only a few of bacteria sense the fatty acid, a quorum sensing system is also included in our circuit. We place luxI downstream of Pfad and another tetR downstream of lux promoter (Plux). At the same time, the expression of LuxR is constantly driven by a constitutive promoter. When a single bacterium detects the fatty acid, LuxI is expressed and it catalyzes the synthesis of N-Acyl homoserine lactone (AHL). AHL activates the LuxR and binds to Plux, results in extra expression of TetR and amplifies the production of GLP-1 and CPP. AHL can be released into the environment and other bacteria that uptake the AHL can be recruited and start producing GLP-1 and CPP as well.
Appetite Regulating Hormone
Obesity has been regarded as chronic disease recent decades. It is reported that obese people are in a higher risk of type II diabetes, cardiovascular disease and hypertension. Nowadays, there are 1 billion people encountering over-weight problem, and 30 million people are diagnosed of obesity. Modern dietary pattern has been considered the major cause contributing to the phenomena.[12]
Incretins are a group of gastrointestinal hormones secreted by endocrine cells in small intestine that mainly work to increase insulin levels. Among all the incretins, glucagon-like peptide-1 (GLP-1) is one of the most widely used appetite inhibiting hormone for treating obese patients [1]. In normal physiological condition GLP-1 is secreted by intestinal L cells after a meal, promoting insulin release and inhibit energy intake [2]. The effect on feeding is signaling through G-protein coupled receptor in solitary tract and brainstem [3]. Beside appetite regulating effect, GLP-1 is also well known for its insulinotropic effect, which is useful for treating type II diabetes[14].
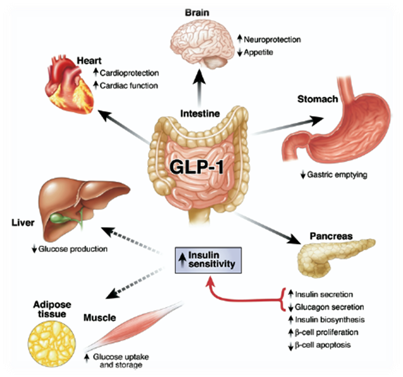
We want to generate a device which is not only helps people annoyed by obese problem, but also potentially effective for treating patients with type II diabetes. The half-life of GLP-1 in plasma is pretty short (5 minutes) because of the degradation by DPP-4 peptidase in serum[13], which is one of the reasons why we are thinking about using supplementary GLP-1 to treat obese patients. In addition, previous study has shown that patients with type II diabetes can be treated with delivery of GLP-1 plasmid construct into hepatic cells[4]. Therefore, by applying the similar philosophy to our bacterial device, we are looking forward to building a device that secret GLP-1 into intestinal lumen when the host is replete.
Fatty Acid Biosensor
in order to regulate the release of GLP-1, we decide to use a fatty acid biosensor which will switch on when the intestinal environment is filled with fatty nutrient. Our team chose fatty acid as a sensor of metabolic states for two main reasons. First, fatty acid is considered as the major cause to obesity because of their highly calorigenic effect. Second, GLP-1 level increases once the blood glucose concentration rises, but there is no significant GLP-1 secretion when the free fatty acid concentration increases in blood stream [15]. Therefore, we hope to compensate this weakness in the appetite regulating mechanisms. By designing the device that secrets GLP-1 on sensing fatty acid around, we aim to make a dietary bacteria.
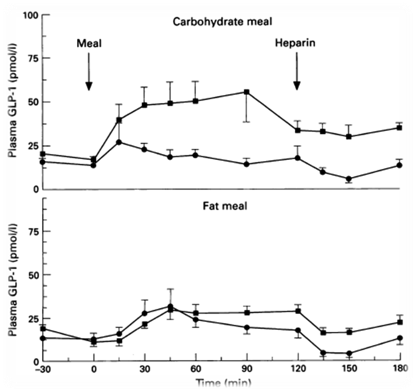
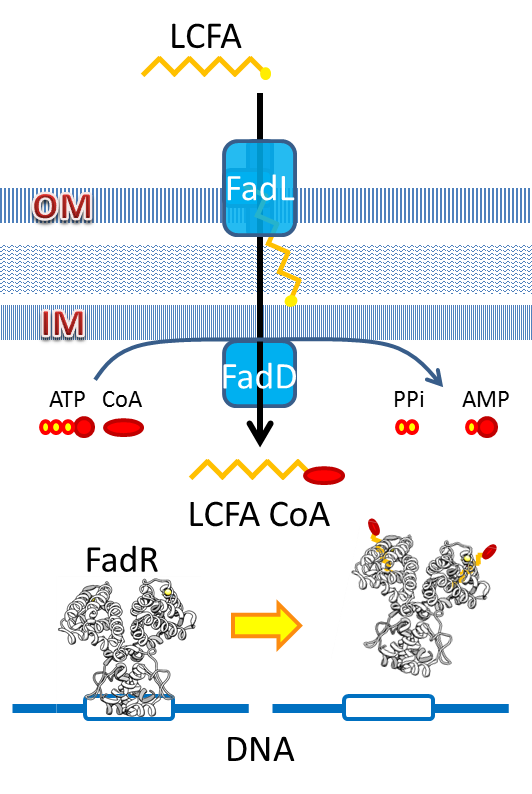
We use the natural fatty acyl-CoA biosensor, the fadBA promoter (PfadBA) from E. coli as the sensor in our design. [5] fadBA is one of the β-oxidation gene which E. coli turns on when the fatty acyl-CoA concentration in the surrounding rises. Therefore, we plan to clone the PfadBA as the promoter in our device, which enables the bacteria to detect fatty acid in the environment.
However, the baseline expression of the reportor gene (mRFP) downstream of PfadBA is so high that we cannot see significant signal rise after induction of fatty acid (oleic acid). Therefore, we decide to lower the baseline expression by overexpression of FadR, an endogenous repressor of PfadBA, whose repressive function is antagonized by fatty acyl-CoA. [6] By co-transforming constructs with PfadBA and FadR into the bacterial platform, it is made capable of changing gene expression in response to environmental fatty acid concentration and produce GLP-1 when the host is in taking a meal.
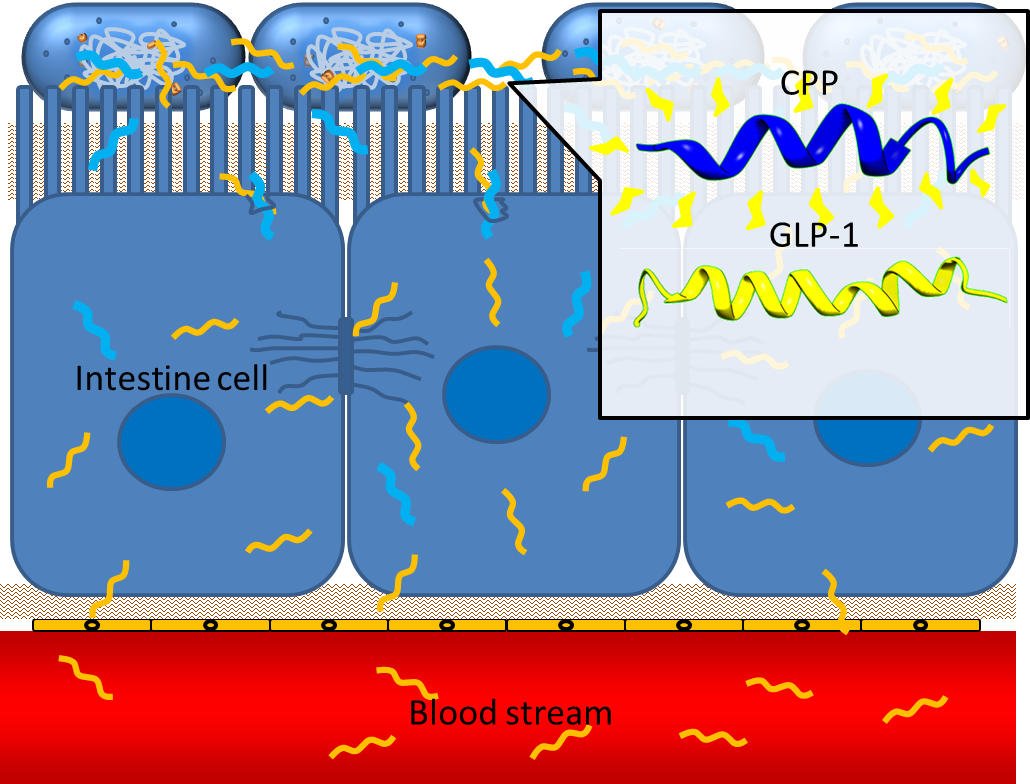
Cell Penetrating Peptide
One obstacle needs to be overcome is the poor penetrating ability of GLP-1 through the intestinal epithelia, since GLP-1 is neither small molecule nor hydrophobic. However, several studies have shown that some short peptides known as cell penetrating peptides (CPPs) can help to deliver biodrugs through intestinal mucosa and increase their bioavailability. [7] CPPs are usually arginine-rich, tend to interact with extracellular matrix and can enhance macropinocytosis of epithelial cells. [9] One recent study even demonstrated that co-administration of GLP-1 and Penetratin, one of the CPPs, greatly increased the intestinal absorption rate to 5%. [8] By applying cell penetrating peptide to our device, we aim to promote the delivery of GLP-1 from intestinal lumen to blood stream, therefore enhance their appetite-regulating effect.
Secretion
In order to achieve the extracellular secretion of our recombinant peptide GLP-1 and CPP, we utilize the endogenous type 2 secretion system (T2SS) in bacteria with specific signal peptides added to the N-terminus of GLP-1 and CPP.
Type 2 Secretion System (T2SS)
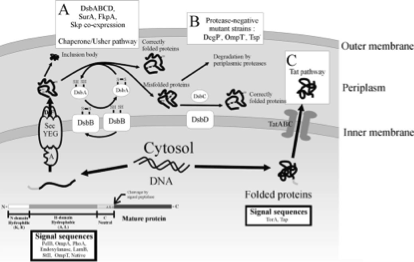
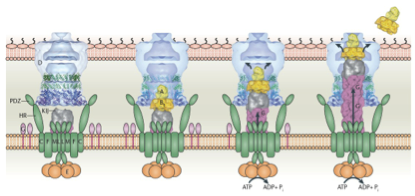
Secretion systems of gram-negative bacteria are categorized into 6 types, from type 1 secretion system (T1SS) to type 6 secretion system (T6SS). Among these secretion systems, type 1 and type 2 secretion systems are in charge of transport bacterial protein products into the extracellular space. [1] Here we choose type 2 secretion system (T2SS) as our secretion apparatus which is widely found in gram-negative bacteria, including E. coli. Unlike the single-step secretion of T1SS such as hemolysin transport system (Hly), T2SS has two-step secretion processes. Unfolded or folded proteins with specific signal sequences on their N-terminus are first transported through the inner membrane into the periplasmic space via Sec translocon or twin-arginine translocation (Tat) pathway, respectively (Fig. 1). [1, 4, 5] The cleavage of signal peptides is performed by signal peptidase inside the periplasm. Proteins of proper structural features are then transported through the outer membrane into the extracellular space via general secretory pathway (GSP) which consists of 4 distinguished subassemblies: inner membrane platform, outer membrane complex, pseudopilus, and secretion ATPase (Fig. 2) [2, 3], while small proteins (in our case, short peptides) can also leak from the periplasmic into the environment, regarding the integrity of the outer membrane. Co-expressions of some genes, such as kil, out, tolAIII, and bacteriocin release protein (BRP), were reported to facilitate the extracellular secretory processes. [1]
Signal Peptides
The selection of signal sequence for delivery of specific recombinant proteins is mostly based on empirical data. Some representative signal sequences, such as those of PhoA (alkaline phosphatase), PelB (pectate lysase B), OmpA (outer-membrane protein A), OmpF (outer-membrane protein F), StII (heat-stable enterotoxin 2), or LTB (heat-labile enterotoxin subunit B), are commonly used. We survey papers of successfully secretion of recombinant proteins with specific signal sequences, and choose 3 sequences which were reported to work in the bacterial strains (E. coli DH5α and BL21) we use. They are signal sequences of PhoA [6], LTB [7], and StII [8], named signal peptide 1 (SP1), signal peptide 2 (SP2), and signal peptide 3 (SP3), respectively. Targeting to the Sec pathway, these signal sequences are cleaved in the periplasmic space, which helps to preserve the structural and functional integrity of the original the peptide. This is also an advantage to choose the T2SS as our delivery system.
| Signal Peptide | Source | Sequence |
|---|---|---|
| SP1 | PhoA | MKQSTIALALLPLLFTPVTKA |
| SP2 | LTB | MNKVKCYVLFTALLSSLYAHG |
| SP3 | StII | MKKNIAFLLASMFVFSIATNAYA |
Thermal Promoter
Phs thermal promoter (toxin, anti-toxin)
E. coli is pretty sensitive and responsive to temperature changes. We brought promoter Phs, located within dnaG, and transient induced to temperature upshift, to our circuit. As described by Wayne E. Talyor et al., Phs can forward upregulation of a series of proteins. The increase activity of Phs promoter is partially compatible with the increase synthesis of sigma factor. The quick respsonse of Phs is suitable since its the sigma factor synthesis peaks 10 minutes after the temperature upshift, and so does Phs RNA levels. The increase ratio before and after the temperature can be more than 20 folds. On particular note, Phs is lack of consensus region over -10 region, which explains its poor activity in 30 Celsuius degree. Thus, Phs is marked as a candidate in our circuit, which can only be functional after ingestion into human body.
We incorporate toxin and anti-toxin into our circuit, as a sensitive device to the environment. Holin, a membrane bound protein, can form complex with each other and trigger the disruption of the membrane. Anti-holin, which is bound to inner membrane, can bind and inactivate Holin. We put Holin under lac repressor control, and when temperature raises, the synthesis of Holin is repressed by LacI, and anti-Holin synthesis also add on to circumvent the lysis of the bacteria. However, when the temperature goes down, the synthesis Holin would not be suppressed, and thus poise for the cellular lysis. This design can make sure that our E. coli would not spread and grow outside of human body.
Modified promoter cI with CIts
This circuit involves a thermosensitive cI promoter under the expression of a strong promoter, J23119, and is followed by a CI promoter (PcI) region and reporter of desire. The whole composite circuit is designed by Harvard iGEM team, 2008. Under conditions demonstrated before, its increase of reporter expression after temperature upshift is minimal (1.8 fold). And thus, we design novel cI promoter region with alterer binding affinity to CIts repressor in search of better efficiency. The circuit aims to provide stable and sustainable GLP-1 synthesis inside human body, that is, the bacteria showed no ability to secret GLP-1 outside of human body under room temperature, under which the PcI is suppressed by CIts.
Stability and Safety
Every GM system that will function outside lab will face two major problems:system stability and safety! Without selective pressure ,we have to deal with plasmid instability ; to make our coli colonized bowel we have to use recA+ strains which may cause plasmid multimer. Our GM coli will also contact with many kinds of bacteria at the risk of horizontal gene transfer.
The following segment is our struggle against these obstacles.
Stability of Delivery System
Briefing
As our PepdEx system consists of several plasmids and will function outside of the laboratory (human gut) which lacks of antibiotic selection pressure, plasmid segregation stability is critical that determines whether every single E. Coli cell contains the original system with the designated function. Inspired by natural plasmid & mobile gene element, we cope with segregation instability by incorporating three modules, i.e. partition system, Multimer resolution system and toxin antitoxin system, on top of our system. We think that this layer of regulation will potentially benefit the entire synthetic biology community, as synthetic systems are becoming increasingly complex with fine controls (e.g. on low copy plasmids).
Obstacles
Sources of plasmid instability:
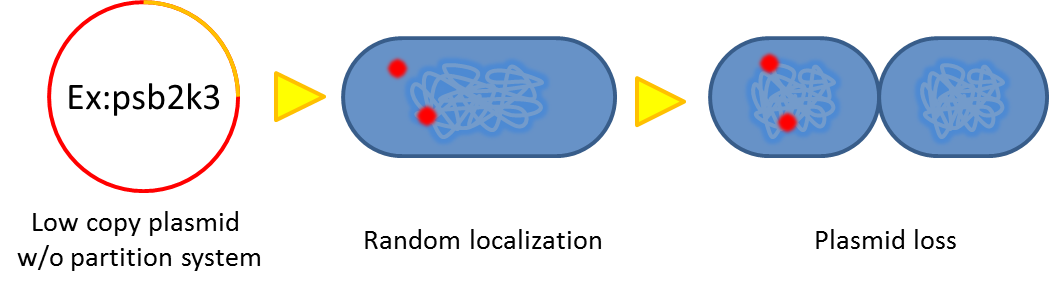
- Segregational instability
Plasmids are randomly (possibly uneven) distributed inside a bacterium. After cell division, one of the progenies may lose plasmids.
- Burden Effect
Cells bearing our PepdEx system have to spend extra energy/resource, so will not grow as good as plasmid-free cells. This metabolic burden will cause our coli to gradually lose pieces of circuits, and the growth rate difference will eventually eliminate plasmid-bearing bacteria in the population.
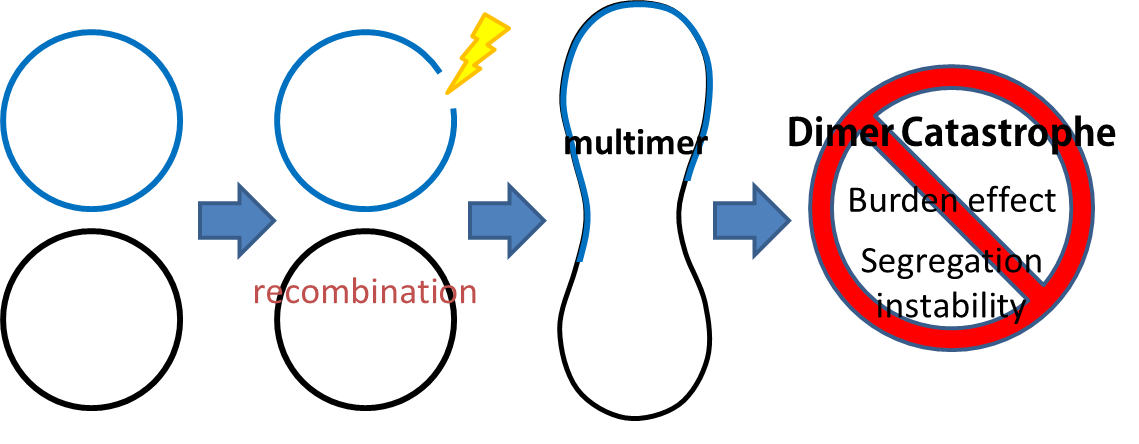
- Dimer Catastrophe
Homologues recombination may cause plasmid multimers which will increases segregational instability and burden. This is the reason why most lab coli are recA1 mutants. But since our coli must be able to colonize in the gut, it should be recA+ wild type strain. That's the problem!
Stabilization Modules
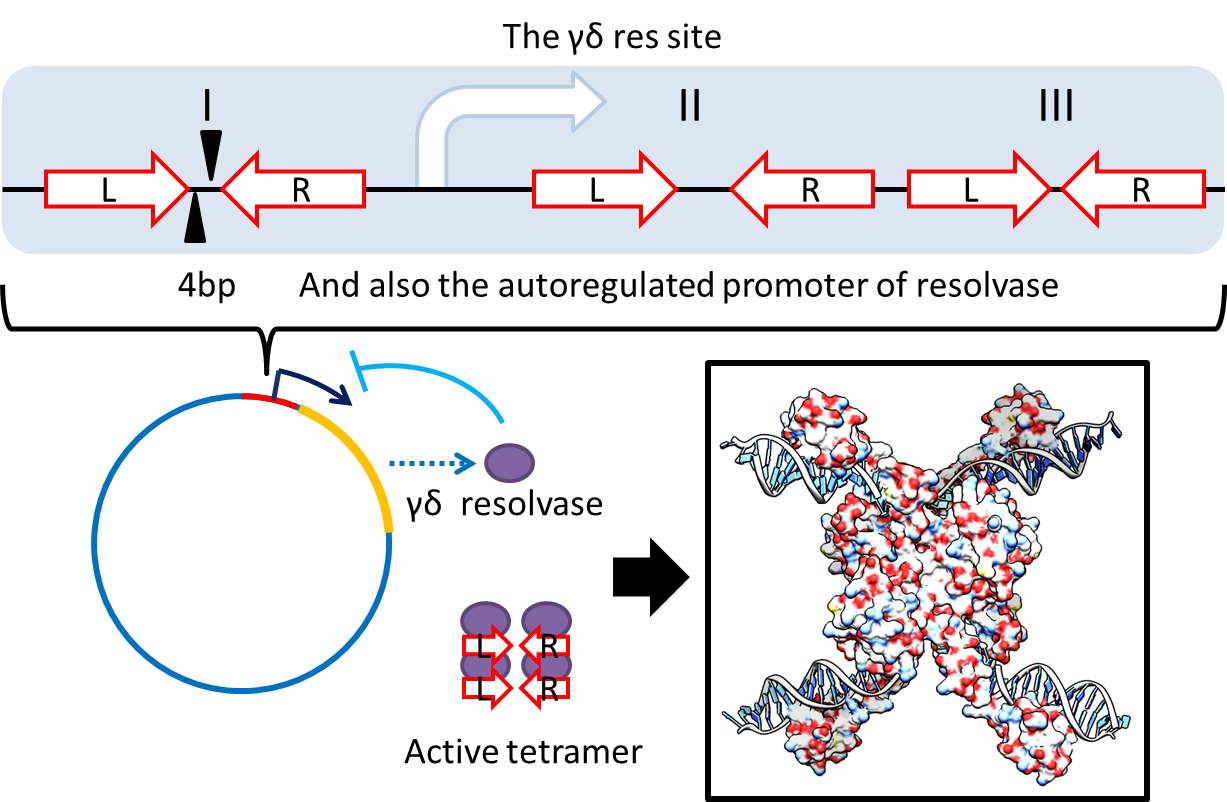
1. Multimer Resolution System:Tn1000(gamma delta) resolution system
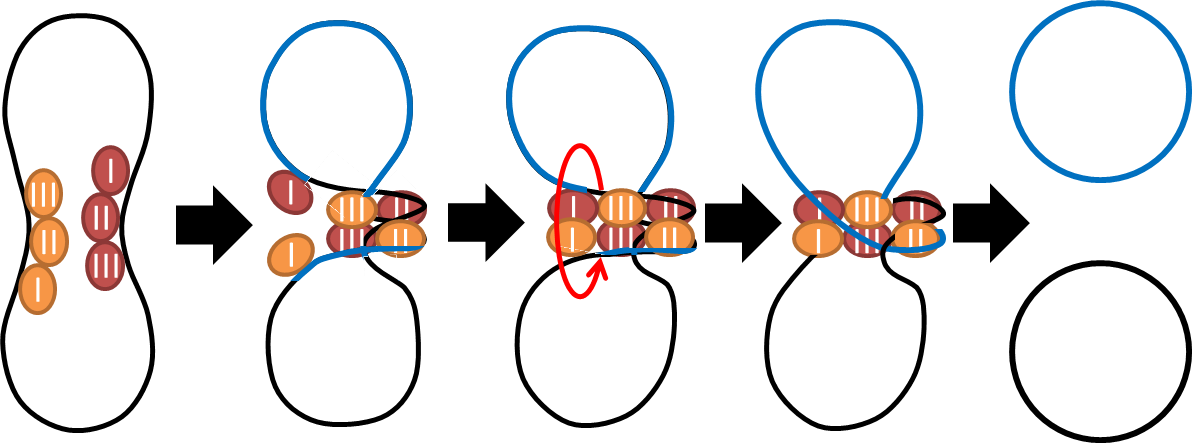
- To deal with multimerization, we cloned an cassette which encodes an autoregulated resolves (serine type recombinase) from E.coli F plasmid transposon tn1000 (tn3 family). Its promoter region consists of 3 sub-sites (res site) and can process recombination. This cassette can resolve multimer that formed during replicative transposition. It can also resolve plasmid multimer to increase plasmid stability by avoiding dimer catastrophe. Multimer resolution system (MRS) provides analogous functions to E.coli chromosome XerCD/dif but acts independent of cell cycle, DNA localization and may have higher efficiency on plasmids compared with slow XerCD system.
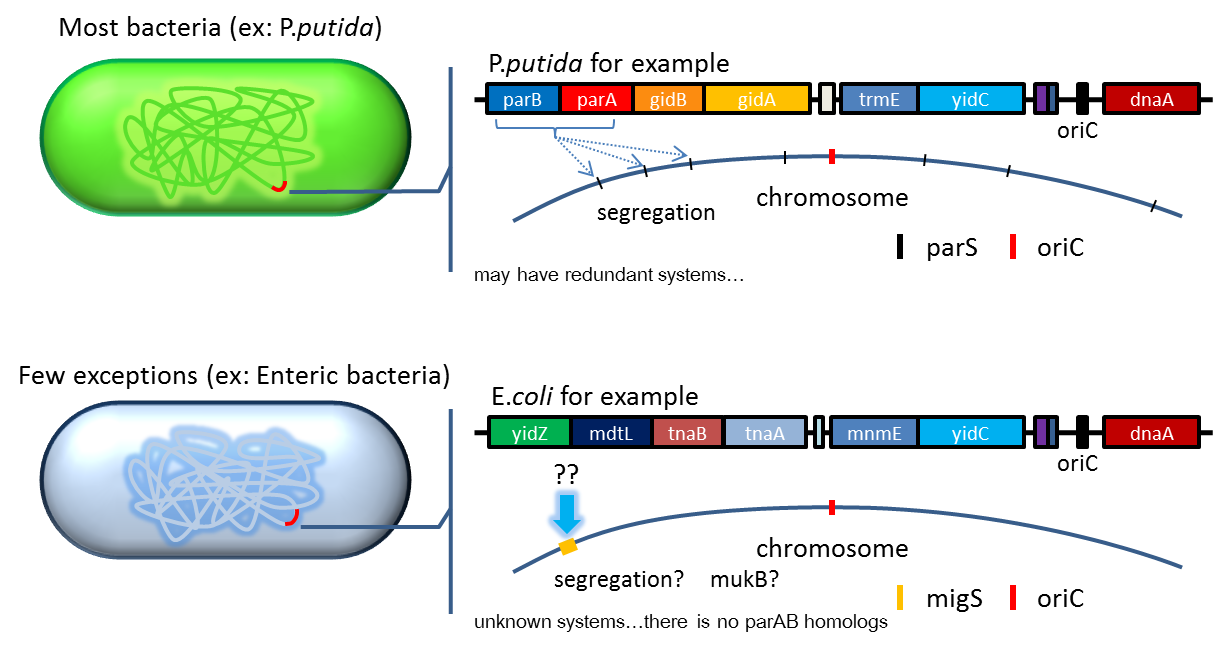
2. Partition System:from Pseudomonas putidaKT2440
- Bacterium distributes chromosomes/plasmids evenly to its progenies by a dynamic system that includes SMC like proteins, type Ia partition system and so on. Type Ia partition system segregates chromosomes/plasmids in a process akin to Eukaryotic mitosis. It can be found on most eubacteria. However, E. coli is not the case.
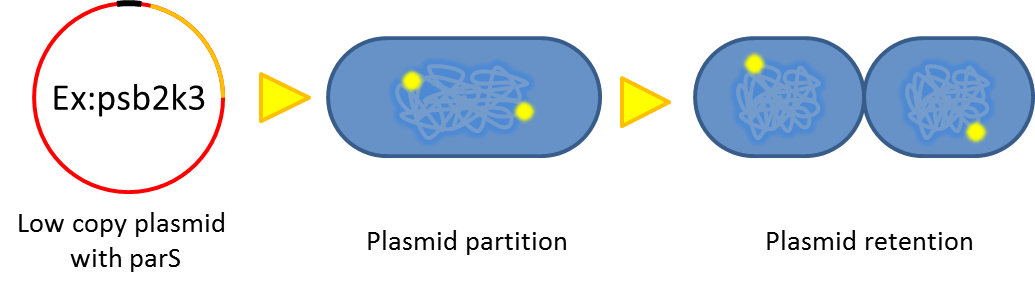
- Previous study have shown that when provide parAB of P.putida in trans and carrying parS site in cis, segregation of low copy plasmids (mini F) can be stabilized in E. coli.(Anne-Marie etl. 2002). The BioBrick vector pSB2K3 is a mini F plasmid, thus we expect its segregation can be stabilized in this way. Our team have made a modified version of pSB2K3 with parS site, and plan to use it to harbor our PepdEx system (a low copy number plasmid allows us to fine tune our systems controls e.g. with smaller number of operator sites).
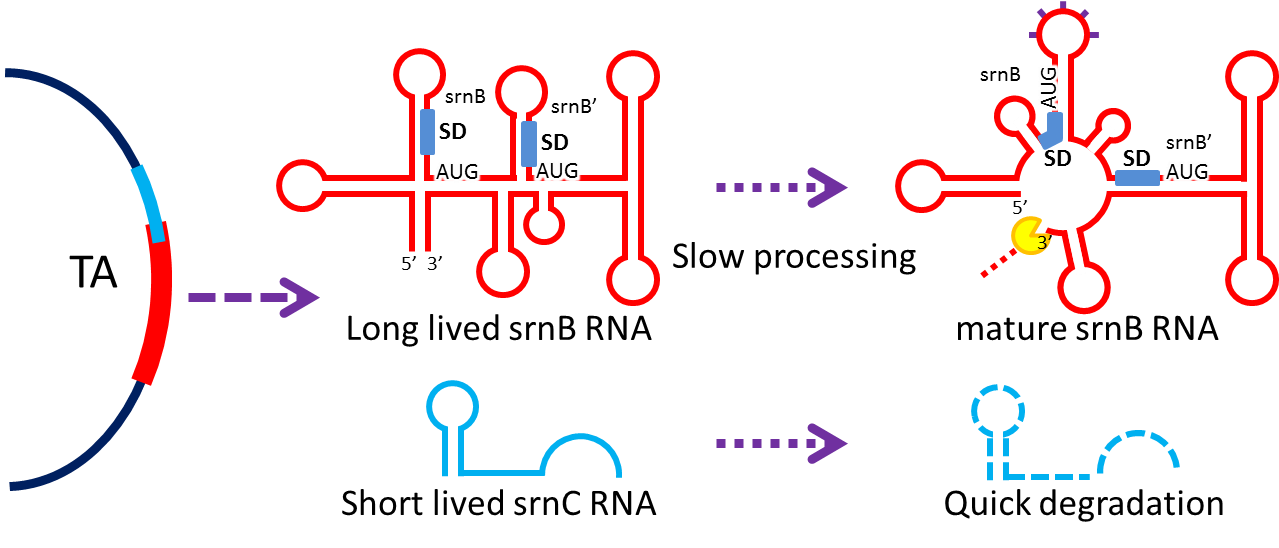
3. Post-Segregation Killing:srnBC toxin-antitoxin system
- No matter how well partition system and multimer resolution system work, inevitably there still will have some bacteria losing plasmid. We sentence them to death to solve the problem.

| 
|
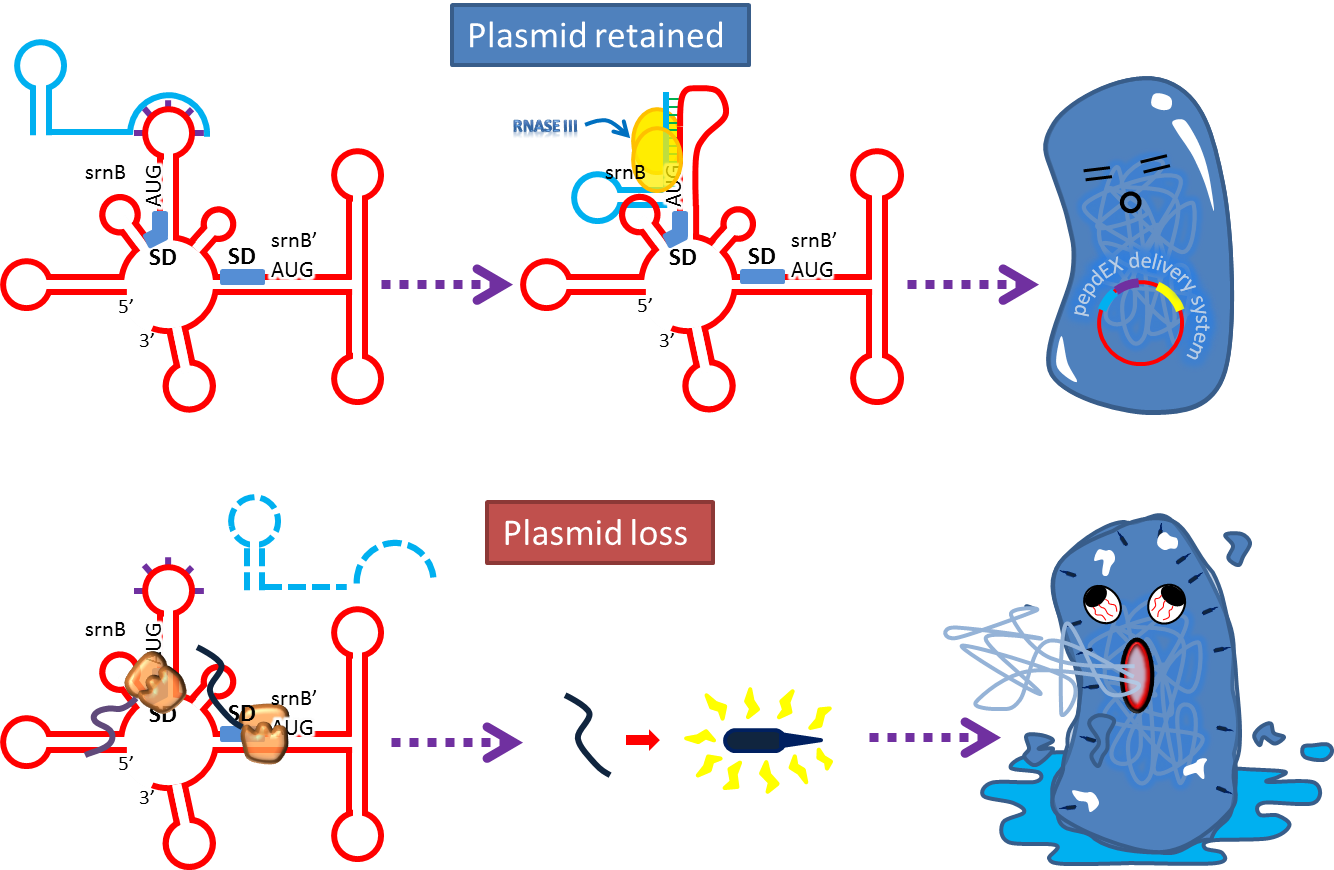
- Type I toxin-antitoxin srnBC is an ideal executor which belongs to hok/sok homologues, it expresses stable toxin encoded RNA and short lived antitoxin RNA that can neutralize toxin RNA by RNA interaction and RNase III cleavage. It acts as post segregation killing system, which kills bacteria when it loss the DNA(genomic islands, plasmids, mobile gene elements) that contains it. Therefore we use it to reduce plasmid loss rate and make applications without antibiotic selection more feasible.
4. Reduce Burden Effect
- Besides these modules, reducing the burden effect is also important to the system stability. Well designed system and lower gene dosage may help.
- High level gene expression and gene dosage cause burden effect. If having the same outcome, low copy plasmid is preferred to high copy ones. If having the same outcome and not for regulatory purpose, stabilizing mRNA is preferred to overexpression it. Place yourselves in E.coli's position, reduce its metabolic burden as much as possible then it can work for you.
Modeling and Application
How to model plasmid instability:
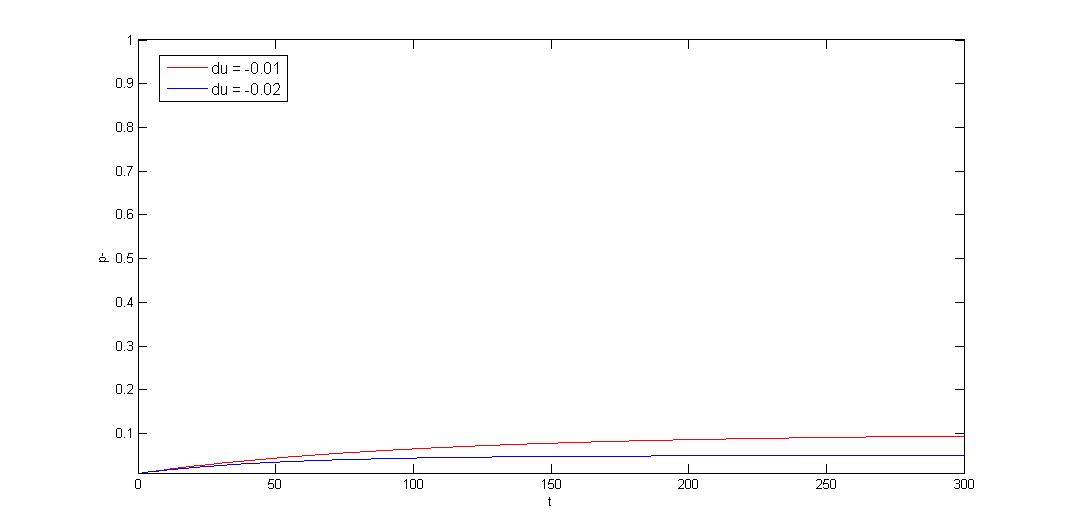
We use Cooper's model (Cooper, N.S., M.E. Brown, and C.A. Caulcott, A ) to model plasmid instability, and set a protocol to suggest users which modules can be used to prove their system stability. Click upper button "modeling" for detail or press following link.
Safety of Delivery System
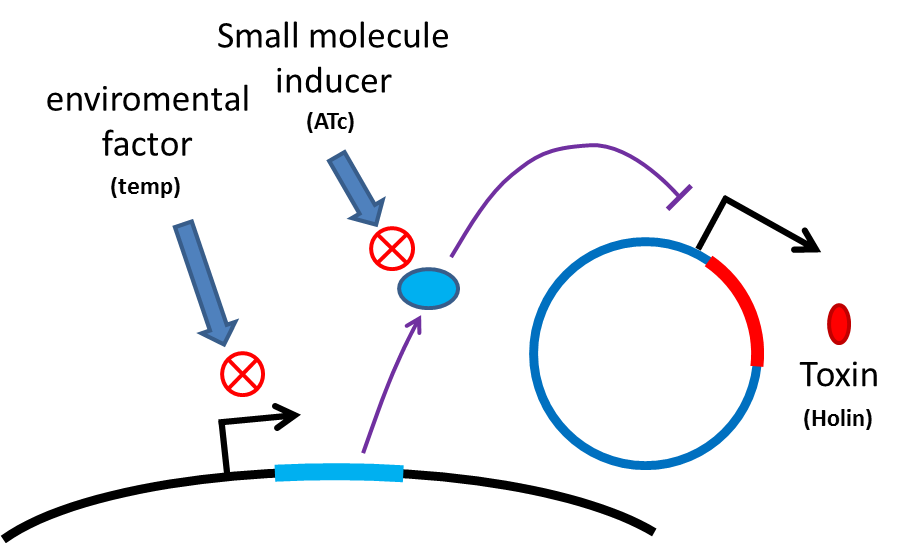
Many turn off strategies have been developed, most of these are the inducible suicide system that can be activated at certain condition. For instance, in our project, we plan to use temperature and small molecule as activating signals( following picture). When the course of treatment ends, administration of small amount of tetracycline agonist will induce bacterial to commit suicide, leaving human body also cause suicide gene activation thereby avoid recombinant strain/gene polluting. And splitting suicide system to provide repression in trans can prevent plasmid transfering to wild type strains. There have been many off-the-rack parts can be used.
However, these design cannot totally eliminate the risk of horizontal gene transfer(HGT), which recombinant genes can move to other organisms independent of suicide system. So besides suicide system, we have a new idea to deal with these kinds of HGT risks by RNA interaction.
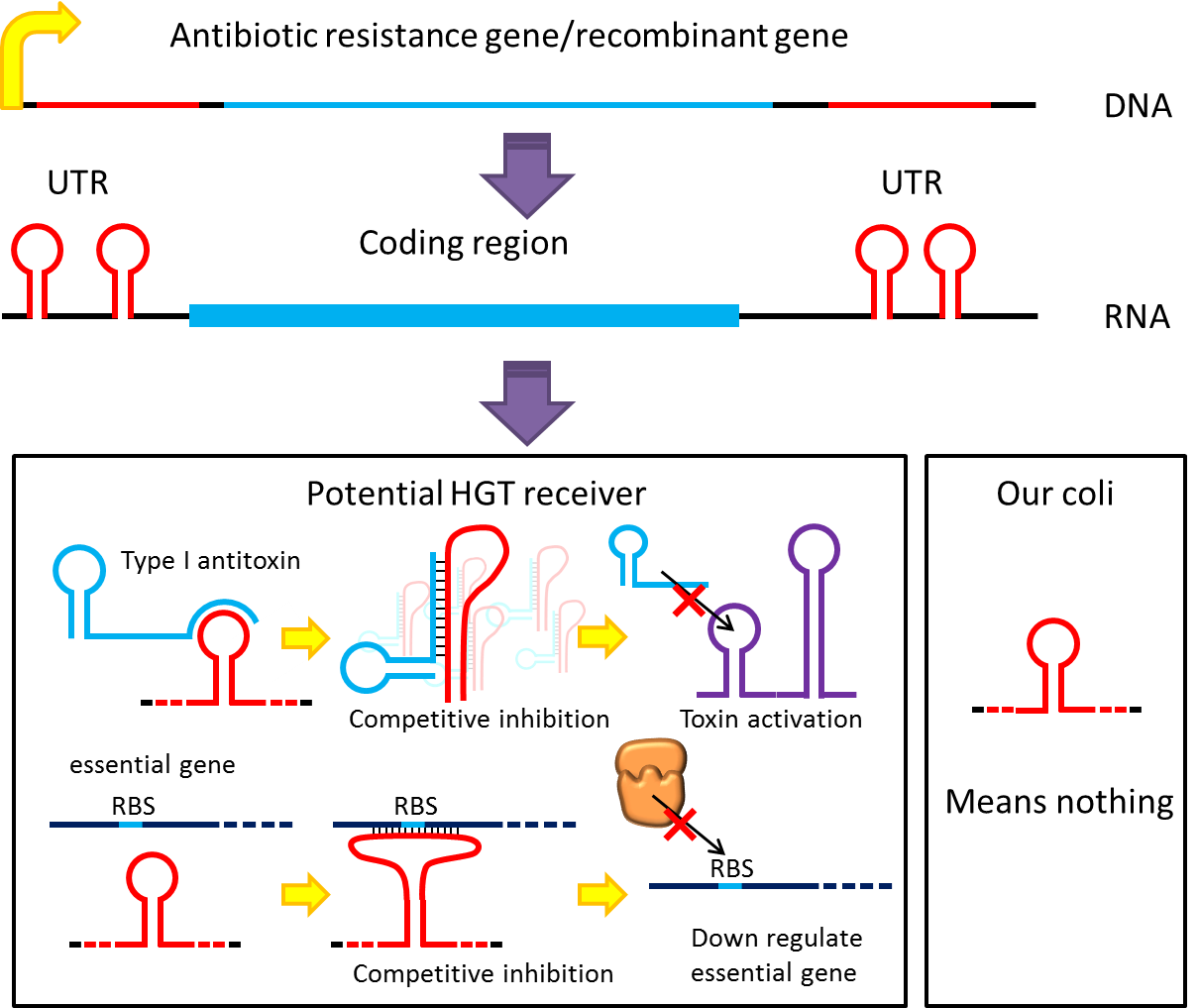
Although bacteria lack for RNAi pathway, expressing well designed antisense RNAs have been shown to have inhibitory effect on target RNAs through competitive inhibition, and recent study showed that peptide nucleic acid (PNA) that antisense to antitoxin RNA 5' sequence can cause bacteria death. Putting appropriate antisense RNAs on untranslated region of transcripts may interfere target RNA function or translation. This property might be used to prevent HGT. For instance, HGT is more likely to occur between related species like lab E.coli & O157, laboratory E.coli have inactivated all its hok/sok toxin-antitoxin system by mutation, but wild bacteria especially pathogenic bacteria usually have more active TA locus on its chromosome like E.coli O157. we plan to put a stem loop from hok mRNA which can pair with sok RNA 5’sequence on UTR of antibiotic resistance genes we used in pepdEX system. IF wild bacteria steal our antibiotic resistance genes and express it, its antitoxin will be competitive inhibited and its toxin will express and kill the thief thus preventing HGT between lab & wild coli. This idea can have wide extension. Besides targeting antitoxin (functional RNA) of type I TA, designing antisense sequences that target RBS to down regulate targeted protein is also possible. Targeting antitoxin of type II TA, essential genes for metabolism, housekeeping genes and any sequences exist in potential HGT receivers but not our coli can be used. Even if the design cannot kill thieves, it can weaken receivers and reduce advantages antibiotic genes bring about thus reduce possibility and danger of HGT.
In the past the repression efficiencies of antisense RNA in bacteria are low, but after invention of the paired termini antisense RNA(PTasRNA) method and incorporate U turn/YUNR motif etc., this idea will become more and more feasible.
Reference of Safety of Delivery System
- Field, C.M. and D.K. Summers, Multicopy plasmid stability: revisiting the dimer catastrophe. Journal of Theoretical Biology, 2011. 291: p. 119-27.
- Grindley, N.D.F., et al., Transposon-Mediated Site-Specific Recombination - Identification of 3 Binding-Sites for Resolvase at the Res-Sites of Gamma-Delta and Tn3. Cell, 1982. 30(1): p. 19-27.
- Wells, R.G. and N.D.F. Grindley, Analysis of the Gamma-Delta Res Site - Sites Required for Site-Specific Recombination and Gene-Expression. Journal of Molecular Biology, 1984. 179(4): p. 667-687.
- Mullins, R.D., Bacterial Chromosome Segregation. Annu Rev Cell Dev Biol, 2009.
- Godfrin-Estevenon, A.M., F. Pasta, and D. Lane, The parAB gene products of Pseudomonas putida exhibit partition activity in both P. putida and Escherichia coli. Molecular Microbiology, 2002. 43(1): p. 39-49.
- Gerdes, K., et al., Mechanism of killer gene activation. Antisense RNA-dependent RNase III cleavage ensures rapid turn-over of the stable hok, srnB and pndA effector messenger RNAs. Journal of Molecular Biology, 1992. 226(3): p. 637-49.
- Thisted, T. and K. Gerdes, Mechanism of post-segregational killing by the hok/sok system of plasmid R1. Sok antisense RNA regulates hok gene expression indirectly through the overlapping mok gene. Journal of Molecular Biology, 1992. 223(1): p. 41-54.
- Szekeres, S., et al., Chromosomal toxin-antitoxin loci can diminish large-scale genome reductions in the absence of selection. Molecular Microbiology, 2007. 63(6): p. 1588-1605.
- Faridani, O.R., et al., Competitive inhibition of natural antisense Sok-RNA interactions activates Hok-mediated cell killing in Escherichia coli. Nucleic Acids Research, 2006. 34(20): p. 5915-5922.
- Gerdes, K. and E.G.H. Wagner, RNA antitoxins. Current Opinion in Microbiology, 2007. 10(2): p. 117-124.
- Good, L. and J.E. Stach, Synthetic RNA silencing in bacteria - antimicrobial discovery and resistance breaking. Front Microbiol, 2011. 2: p. 185.
- Faridani, O.R., et al., Competitive inhibition of natural antisense Sok-RNA interactions activates Hok-mediated cell killing in Escherichia coli. Nucleic Acids Research, 2006. 34(20): p. 5915-22.
- Lucks, J.B., et al., Versatile RNA-sensing transcriptional regulators for engineering genetic networks. Proc Natl Acad Sci U S A, 2011. 108(21): p. 8617-22.
- Franch, T., et al., Antisense RNA regulation in prokaryotes: rapid RNA/RNA interaction facilitated by a general U-turn loop structure. Journal of Molecular Biology, 1999. 294(5): p. 1115-25.
Reference
- The Gut Hormones PYY3-36 and GLP-17-36 amide Reduce Food Intake and Modulate Brain Activity in Appetite Centers in Humans.
- The gastrointestinal tract and the regulation of appetite.
- The Multiple Actions of GLP-1 on the Process of Glucose-Stimulated Insulin Secretion
- Glucagon-like Peptide-1 Plasmid Construction and Delivery for the Treatment of Type 2 Diabetes.
- Design of a dynamic sensor-regulator system for production of chemicals and fuels derived from fatty acids.
- Unexpected Functional Diversity among FadR Fatty Acid Transcriptional Regulatory Proteins.
- Oral biodrug delivery using cell-penetrating peptide
- Efficiency of cell-penetrating peptides on the nasal and intestinal absorption of therapeutic peptides and proteins.
- Cellular Uptake of Arginine-Rich Peptides: Roles for Macropinocytosis and Actin Rearrangement
- Biology of Incretins: GLP-1 and GIP
- Crystal structure of FadR, a fatty acid-responsive transcription factor with a novel acyl coenzyme A-binding fold.
- Obesity and Overweight
- Active glucagon-like peptide-1 (GLP-1): Storage of human plasma and stability over time.
- One week's treatment with the long-acting glucagon-like peptide 1 derivative liraglutide (NN2211) markedly improves 24-h glycemia and alpha- and beta-cell function and reduces endogenous glucose release in patients with type 2 diabetes
- Attenuated GLP-1 secretion in obesity: cause or consequence?
 "
"