Team:HokkaidoU Japan/Notebook/aggregation Week 2
From 2012.igem.org
| Line 11: | Line 11: | ||
Colony PCR for assembly products.Each product reacted recipes written below. | Colony PCR for assembly products.Each product reacted recipes written below. | ||
#picked up each 12(16 is best but there were only 12 colonies) colonies from LB plates by Autoclaved toothpicks. | #picked up each 12(16 is best but there were only 12 colonies) colonies from LB plates by Autoclaved toothpicks. | ||
| - | #Dipped into | + | #Dipped into 10 ul DW in 1.5 ml eppendorf tubes. |
| - | #from | + | #from 10 ul DW, 4 ul was added in PCR reaction solution (below), 6 ul was mixed with 200 ul LB. |
#Ran PCR machine in recipe below. | #Ran PCR machine in recipe below. | ||
#Electrophoresis for confirmation of PCR results. | #Electrophoresis for confirmation of PCR results. | ||
| Line 19: | Line 19: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | | | + | |4 ul |
|- | |- | ||
|KapaTaq ready mix | |KapaTaq ready mix | ||
| - | | | + | |5 ul |
|- | |- | ||
|BioBrick prefix forward primer | |BioBrick prefix forward primer | ||
| - | |0. | + | |0.5 ul |
|- | |- | ||
|BioBrick suffix reverse primer | |BioBrick suffix reverse primer | ||
| - | |0. | + | |0.5 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |10 ul |
|} | |} | ||
| Line 61: | Line 61: | ||
Cycle:2~3 x 40 | Cycle:2~3 x 40 | ||
(Ag43 + dT) | (Ag43 + dT) | ||
| - | Ag43 + dT (+ Biobrick prefis & suffix) is about 3290bp. Extension step needed > | + | Ag43 + dT (+ Biobrick prefis & suffix) is about 3290bp. Extension step needed >180 seconds. |
{|class="hokkaidou-table-pcr-time" | {|class="hokkaidou-table-pcr-time" | ||
|- | |- | ||
| Line 112: | Line 112: | ||
<p> | <p> | ||
Liquid culture for mini-prep((pT7 + RBS) on pSB1K3 and (Ag43 + dT) on pSB1AK3). | Liquid culture for mini-prep((pT7 + RBS) on pSB1K3 and (Ag43 + dT) on pSB1AK3). | ||
| - | #Prepared | + | #Prepared 1800 ul LB solutions. |
| - | #To these LB solutions, added | + | #To these LB solutions, added 200 ul of LB solutions (colony PCR solutions were pre-cultivated in about 2 hrs) and each antibiotics(Km (for (pT7 + RBS) on pSB1K3) and Amp(for (Ag43 + dT) on pSB1AK3)). |
| - | #Cultivated | + | #Cultivated 15 hrs and 30 min. |
</p> | </p> | ||
</div></div> | </div></div> | ||
| Line 124: | Line 124: | ||
===Mini-prep=== | ===Mini-prep=== | ||
<p> | <p> | ||
| - | Mini-prep for some colonies (we selected colonies which showed more correct bands than other colonies, pT7+RBS were No.4, 5, 9, 10 colonies and Ag43 + dT were No.1, 2, 3, 4 colonies)cultivated in LBA(Ag43 + dT) and LBK(pT7 + RBS) in | + | Mini-prep for some colonies (we selected colonies which showed more correct bands than other colonies, pT7+RBS were No. 4, 5, 9, 10 colonies and Ag43 + dT were No. 1, 2, 3, 4 colonies)cultivated in LBA(Ag43 + dT) and LBK(pT7 + RBS) in 15 hrs and 30 min. |
#Mini-prep for LB culturing products along the protocol of FastGene Plasmid Mini kit(Nippon Genetics). | #Mini-prep for LB culturing products along the protocol of FastGene Plasmid Mini kit(Nippon Genetics). | ||
| - | #Electrophoresis (Used pre-migrated 1% agarose gels with | + | #Electrophoresis (Used pre-migrated 1% agarose gels with 5 ul EtBr)in 30 min. |
</p> | </p> | ||
| Line 147: | Line 147: | ||
|- | |- | ||
|pT7-RBS | |pT7-RBS | ||
| - | |1 | + | |1.5 ul |
|- | |- | ||
|EcoRI | |EcoRI | ||
| - | | | + | |1 ul |
|- | |- | ||
|PstI | |PstI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |2 ul |
|- | |- | ||
|DW | |DW | ||
| - | |14. | + | |14.5 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |20 ul |
|} | |} | ||
| Line 171: | Line 171: | ||
|- | |- | ||
|Ag43-dT | |Ag43-dT | ||
| - | | | + | |4 ul |
|- | |- | ||
|EcoRI | |EcoRI | ||
| - | | | + | |1 ul |
|- | |- | ||
|PstI | |PstI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |2 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |12 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |20 ul |
|} | |} | ||
| - | Digestioned at 37c in | + | Digestioned at 37c in 2 hrs. |
| Line 217: | Line 217: | ||
|- | |- | ||
|DNA | |DNA | ||
| - | | | + | |17 ul |
|- | |- | ||
|EcoRI | |EcoRI | ||
| - | | | + | |1 ul |
|- | |- | ||
|SpeI | |SpeI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |3 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |8 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |30 ul |
|} | |} | ||
| Line 240: | Line 240: | ||
|- | |- | ||
|DNA | |DNA | ||
| - | |12. | + | |12.5 ul |
|- | |- | ||
|XbaI | |XbaI | ||
| - | | | + | |1 ul |
|- | |- | ||
|PstI | |PstI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |2 ul |
|- | |- | ||
|DW | |DW | ||
| - | |3. | + | |3.5 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |20 ul |
|} | |} | ||
| Line 263: | Line 263: | ||
|- | |- | ||
|DNA | |DNA | ||
| - | | | + | |20 ul |
|- | |- | ||
|EcoRI | |EcoRI | ||
| - | | | + | |1 ul |
|- | |- | ||
|PstI | |PstI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |3 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |5 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |30 ul |
|} | |} | ||
| - | Reacted in | + | Reacted in 2 hrs at 37c. |
</p> | </p> | ||
</div></div> | </div></div> | ||
| Line 298: | Line 298: | ||
|- | |- | ||
|pT7-RBS | |pT7-RBS | ||
| - | | | + | |2 ul |
|- | |- | ||
|Ag43-dT | |Ag43-dT | ||
| - | | | + | |2 ul |
|- | |- | ||
|pSB1C3 | |pSB1C3 | ||
| - | | | + | |3 ul |
|- | |- | ||
|Ligation Mighty Mix(TAKARA) | |Ligation Mighty Mix(TAKARA) | ||
| - | | | + | |8 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |16 ul |
|} | |} | ||
| Line 339: | Line 339: | ||
===Transformation=== | ===Transformation=== | ||
Transformation for pT7-RBS + Ag43-dT + pSB1C3 into BL21(DE3)(E.coli which have T7 polymerase coadin site in their genomic DNA). | Transformation for pT7-RBS + Ag43-dT + pSB1C3 into BL21(DE3)(E.coli which have T7 polymerase coadin site in their genomic DNA). | ||
| - | #Added DNA soltions (Ligation products) | + | #Added DNA soltions (Ligation products) 1 ul to BL21(DE3) compitent cell. |
| - | #Stood on ice in | + | #Stood on ice in 30 min. |
#Heatshock for 1min at 42c. | #Heatshock for 1min at 42c. | ||
| - | #Added | + | #Added 200 ul of LB to transformed BL21(DE3) solution. |
| - | #Pre-cultivate in | + | #Pre-cultivate in 2 hrs |
| - | #Splead | + | #Splead 200 ul of LB&BL21(DE3) solution supernant to LBC. |
| - | #50ul of LB&BL21(DE3) solution were added to 200ul LB solution then spread | + | #50ul of LB&BL21(DE3) solution were added to 200ul LB solution then spread 200 ul to LBC plate. |
#Cultivated. | #Cultivated. | ||
</p> | </p> | ||
| Line 358: | Line 358: | ||
Colony PCR for ligation product.Each products were reacted in recipes written below. | Colony PCR for ligation product.Each products were reacted in recipes written below. | ||
#picked up each 16 colonies from LB plates by Autoclaved toothpicks. | #picked up each 16 colonies from LB plates by Autoclaved toothpicks. | ||
| - | #Dipped into | + | #Dipped into 10 ul DW in 1.5 ml eppendorf tubes. |
| - | #from | + | #from 10 ul DW, 4 ul was added in PCR reaction solution (below), 6 ul was mixed with 200 ul LB. |
#Ran PCR machine in recipe below. | #Ran PCR machine in recipe below. | ||
#Electrophoresis for confirmation of PCR results. | #Electrophoresis for confirmation of PCR results. | ||
| Line 368: | Line 368: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | | | + | |4 ul |
|- | |- | ||
|KapaTaq ready mix | |KapaTaq ready mix | ||
| - | | | + | |5 ul |
|- | |- | ||
|BioBrick prefix forward primer | |BioBrick prefix forward primer | ||
| - | |0. | + | |0.5 ul |
|- | |- | ||
|BioBrick suffix reverse primer | |BioBrick suffix reverse primer | ||
| - | |0. | + | |0.5 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |10 ul |
|} | |} | ||
| Line 415: | Line 415: | ||
<p> | <p> | ||
Liquid culture for some colonies used in colony PCR. | Liquid culture for some colonies used in colony PCR. | ||
| - | #Prepared | + | #Prepared 200 ul LB solutions. |
| - | #To these LB solutions, added | + | #To these LB solutions, added 6 ul of LB solutions (colony PCR solutions were pre-cultivated in about 3 hrs) and added 2 ml LB and antibiotic(Cp). |
| - | #Cultivated | + | #Cultivated 18 hrs and 30 min. |
</p> | </p> | ||
</div></div> | </div></div> | ||
| Line 427: | Line 427: | ||
===Mini-prep=== | ===Mini-prep=== | ||
<p> | <p> | ||
| - | Mini-prep for colony No.1~8 | + | Mini-prep for colony No. 1~8 |
We used mini-prep kit FastGene Plasmid Mini Kit (Nippon Genetics). | We used mini-prep kit FastGene Plasmid Mini Kit (Nippon Genetics). | ||
| Line 445: | Line 445: | ||
|- | |- | ||
|DNA | |DNA | ||
| - | | | + | |4 ul |
|- | |- | ||
|EcoRI | |EcoRI | ||
| - | |0. | + | |0.5 ul |
|- | |- | ||
|PstI | |PstI | ||
| - | |0. | + | |0.5 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |2 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |13 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |20 ul |
|} | |} | ||
| Line 485: | Line 485: | ||
|- | |- | ||
|Insert | |Insert | ||
| - | | | + | |5 ul |
|- | |- | ||
|Vector | |Vector | ||
| - | | | + | |1 ul |
|- | |- | ||
|Ligation Mighty Mix(TAKARA) | |Ligation Mighty Mix(TAKARA) | ||
| - | | | + | |6 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |12 ul |
|} | |} | ||
| Line 514: | Line 514: | ||
Ligation result | Ligation result | ||
| - | with colony no.1 (see colony pcr result in 9th) | + | with colony no. 1 (see colony pcr result in 9th) |
[[image:HokkaidoU2012 120714 pT7-RBS on 1K3 pSB1C3 Ag43-dT on 1AK3 pSB1T3 Ligation1,2 coloP-No.jpg]] | [[image:HokkaidoU2012 120714 pT7-RBS on 1K3 pSB1C3 Ag43-dT on 1AK3 pSB1T3 Ligation1,2 coloP-No.jpg]] | ||
| Line 524: | Line 524: | ||
| - | #Added DNA soltions (Ligation products) | + | #Added DNA soltions (Ligation products) 1 ul to DH5α compitent cell. |
| - | #Stood on ice in | + | #Stood on ice in 30 min. |
| - | #Added | + | #Added 600 ul of LB to transformed DH5α solution. |
| - | #Pre-cultivate in | + | #Pre-cultivate in 2 hrs |
| - | #Splead | + | #Splead 300 ul of LB&DH5α solution to LBC and LBT , 100 ul added into 900 ul of LB. |
| - | #Splead | + | #Splead 300 ul of LB&DH5α solution from 1000 ul LB (100 ul added into 900 ul) to LBC and LBT. |
| - | #Cultivated in | + | #Cultivated in 21 hrs. |
| Line 567: | Line 567: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | | | + | |6 ul |
|- | |- | ||
|EcoRI | |EcoRI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |1 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |2 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |10 ul |
|} | |} | ||
| Line 587: | Line 587: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | | | + | |6 ul |
|- | |- | ||
|XbaI | |XbaI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xM buffer | |10xM buffer | ||
| - | | | + | |1 ul |
|- | |- | ||
|BSA | |BSA | ||
| - | | | + | |1 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |1 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |10 ul |
|} | |} | ||
| Line 610: | Line 610: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | | | + | |6 ul |
|- | |- | ||
|PstI | |PstI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |1 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |2 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |10 ul |
|} | |} | ||
| Line 631: | Line 631: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | | | + | |6 ul |
|- | |- | ||
|SpeI | |SpeI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xM buffer | |10xM buffer | ||
| - | | | + | |1 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |2 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |10 ul |
|} | |} | ||
| Line 651: | Line 651: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | | | + | |6 ul |
|- | |- | ||
|EcoRI | |EcoRI | ||
| - | | | + | |1 ul |
|- | |- | ||
|PstI | |PstI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |1 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |11 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |20 ul |
|} | |} | ||
| Line 674: | Line 674: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | | | + | |6 ul |
|- | |- | ||
|XbaI | |XbaI | ||
| - | | | + | |1 ul |
|- | |- | ||
|SpeI | |SpeI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xM buffer | |10xM buffer | ||
| - | | | + | |1 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |11 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |20 ul |
|} | |} | ||
</p> | </p> | ||
| Line 696: | Line 696: | ||
<p> | <p> | ||
Ethanol precipitation for digestion products. | Ethanol precipitation for digestion products. | ||
| - | #Added | + | #Added 2 ul of NaoAc, 1.5 ul of glycogen and 50 ul of 100% ethanol. |
| - | #Centrifuged in | + | #Centrifuged in 15000 rpm, 10 min at 4C. |
| - | #Remove supernatant and added | + | #Remove supernatant and added 220 ul of 70% ethanol. |
| - | #Centrifuged in | + | #Centrifuged in 15000 rpm, 5 min at 4C. |
| - | #Remove supernatant and air drying in room temperature then added | + | #Remove supernatant and air drying in room temperature then added 5 ul of DW. |
</p> | </p> | ||
| Line 706: | Line 706: | ||
<p> | <p> | ||
Electrophoresis for digest-ethanol precipitation products. | Electrophoresis for digest-ethanol precipitation products. | ||
| - | #added | + | #added 5 ul of EtBr. |
| - | #Migrated in | + | #Migrated in 30 min. |
| Line 735: | Line 735: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | | | + | |9 ul |
|- | |- | ||
|SpeI | |SpeI | ||
| - | | | + | |1 ul |
|- | |- | ||
|PstI | |PstI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |2 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |7 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |20 ul |
|} | |} | ||
| Line 758: | Line 758: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | |1. | + | |1.1 ul |
|- | |- | ||
|XbaI | |XbaI | ||
| - | | | + | |1 ul |
|- | |- | ||
|PstI | |PstI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xM buffer | |10xM buffer | ||
| - | | | + | |2 ul |
|- | |- | ||
|DW | |DW | ||
| - | |14. | + | |14.9 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |20 ul |
|} | |} | ||
| Line 781: | Line 781: | ||
|- | |- | ||
|DNA solution | |DNA solution | ||
| - | | | + | |6 ul |
|- | |- | ||
|EcoRI | |EcoRI | ||
| - | | | + | |1 ul |
|- | |- | ||
|10xH buffer | |10xH buffer | ||
| - | | | + | |2 ul |
|- | |- | ||
|DW | |DW | ||
| - | | | + | |11 ul |
|- | |- | ||
|Total | |Total | ||
| - | | | + | |20 ul |
|} | |} | ||
</p> | </p> | ||
Revision as of 08:32, 6 August 2012
Contents |
July 9th
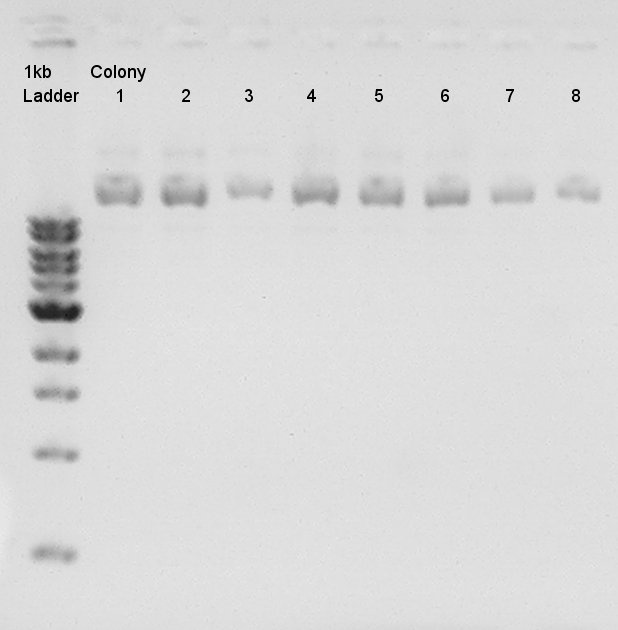
pT7 + RBS (3A Assembly) and Ag43 + dT (standard assembly) ligation products were transformed and cultivated then some colonies were formed (12~16 colonies) so we confirmed really insert DNA (3A:pT7 and RBS, standard:Ag43 and dT) were inserted to vector or not by colony PCR.
Colony PCR
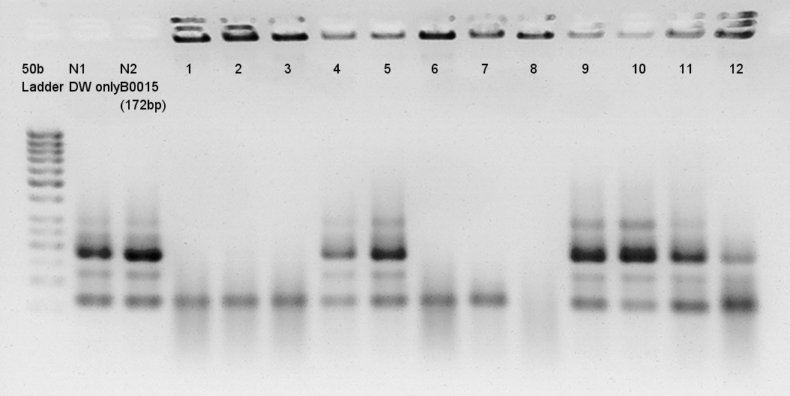
Colony PCR for assembly products.Each product reacted recipes written below.
- picked up each 12(16 is best but there were only 12 colonies) colonies from LB plates by Autoclaved toothpicks.
- Dipped into 10 ul DW in 1.5 ml eppendorf tubes.
- from 10 ul DW, 4 ul was added in PCR reaction solution (below), 6 ul was mixed with 200 ul LB.
- Ran PCR machine in recipe below.
- Electrophoresis for confirmation of PCR results.
| DNA solution | 4 ul |
| KapaTaq ready mix | 5 ul |
| BioBrick prefix forward primer | 0.5 ul |
| BioBrick suffix reverse primer | 0.5 ul |
| Total | 10 ul |
PCR recipe
(pT7 + RBS)
| Number | Degree | Second |
| 1 | 94 | 120 |
| 2 | 94 | 30 |
| 3 | 68 | 60 |
| 4 | 4 | HOLD |
Cycle:2~3 x 40 (Ag43 + dT) Ag43 + dT (+ Biobrick prefis & suffix) is about 3290bp. Extension step needed >180 seconds.
| Number | Degree | Second |
| 1 | 94 | 120 |
| 2 | 94 | 30 |
| 3 | 68 | 180 |
| 4 | 4 | HOLD |
Cycle:2~3 x 35
Electrophoresis results
Electrophoresis for (pT7 + RBS) in 2% agarose gel and (Ag43 + dT) in 1% agarose gel. pT7 + RBS on pSB1K3 bbp-Insert-bbs:86bp
Ag43 + dT on pSB1AK3
bbp-Insert-bbs:3290bp
We couldn't confirm insert DNA were really ligated with Vector or not.
Next we tried confirmation of insert DNA by Electrophoresis of mini-prep products.
For mini-prep, we needed do liquid culture.
Liquid culturing
Liquid culture for mini-prep((pT7 + RBS) on pSB1K3 and (Ag43 + dT) on pSB1AK3).
- Prepared 1800 ul LB solutions.
- To these LB solutions, added 200 ul of LB solutions (colony PCR solutions were pre-cultivated in about 2 hrs) and each antibiotics(Km (for (pT7 + RBS) on pSB1K3) and Amp(for (Ag43 + dT) on pSB1AK3)).
- Cultivated 15 hrs and 30 min.
July 10th
Mini-prep
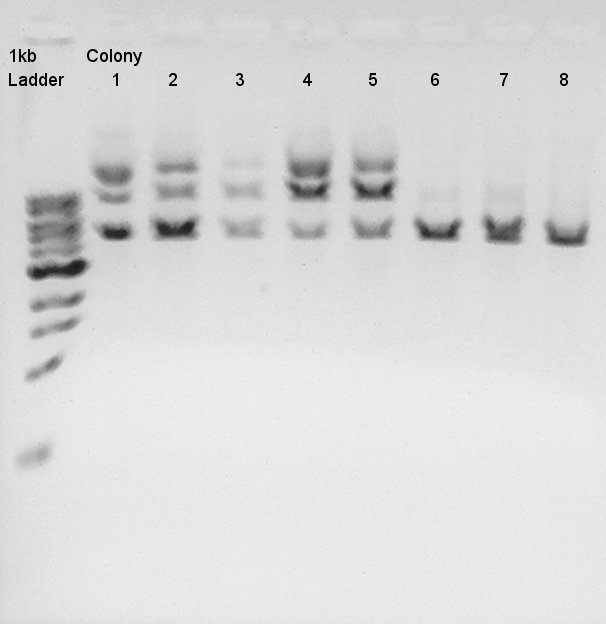
Mini-prep for some colonies (we selected colonies which showed more correct bands than other colonies, pT7+RBS were No. 4, 5, 9, 10 colonies and Ag43 + dT were No. 1, 2, 3, 4 colonies)cultivated in LBA(Ag43 + dT) and LBK(pT7 + RBS) in 15 hrs and 30 min.
- Mini-prep for LB culturing products along the protocol of FastGene Plasmid Mini kit(Nippon Genetics).
- Electrophoresis (Used pre-migrated 1% agarose gels with 5 ul EtBr)in 30 min.
pT7 + RBS on pSB1K3(Total 2247bp)
Ag43 + dT on pSB1AK3(Total 6444bp)
To confirm more about insert DNA, we tried to digest these DNA with EcoRI and PstI.
Digestion
Digestion for confirmation of insert DNA. Each DNA mini-prep products were digested with E & P. Digestion recipe pT7-RBS
| pT7-RBS | 1.5 ul |
| EcoRI | 1 ul |
| PstI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 14.5 ul |
| Total | 20 ul |
Digestion recipe
Ag43-dT
| Ag43-dT | 4 ul |
| EcoRI | 1 ul |
| PstI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 12 ul |
| Total | 20 ul |
Digestioned at 37c in 2 hrs.
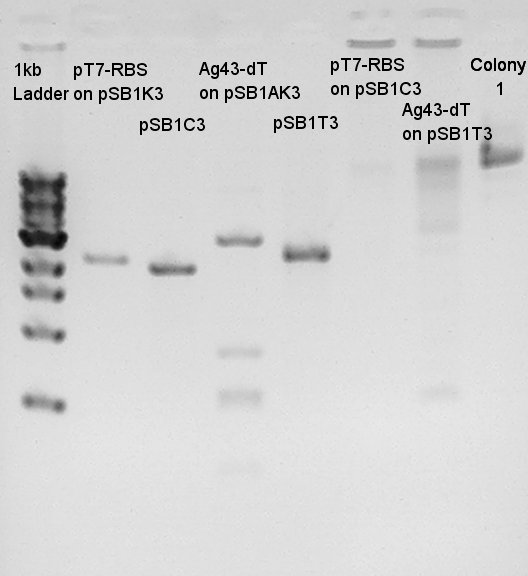
Digestion results
pT7+RBS
Ag43+dT
Insert DNA were correct we thought. We tried digestion for 3A Assembly[pT7-RBS + Ag43-dT + pSB1C3]
Digestion
Digestion for 3A Assembly. pT7-RBS
| DNA | 17 ul |
| EcoRI | 1 ul |
| SpeI | 1 ul |
| 10xH buffer | 3 ul |
| DW | 8 ul |
| Total | 30 ul |
Ag43-dT
| DNA | 12.5 ul |
| XbaI | 1 ul |
| PstI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 3.5 ul |
| Total | 20 ul |
pSB1C3
| DNA | 20 ul |
| EcoRI | 1 ul |
| PstI | 1 ul |
| 10xH buffer | 3 ul |
| DW | 5 ul |
| Total | 30 ul |
Reacted in 2 hrs at 37c.
July 11th
Ligation
Ligation of pT7-RBS + Ag43-dT + pSB1C3(3A Assembly) Ligation recipe
| pT7-RBS | 2 ul |
| Ag43-dT | 2 ul |
| pSB1C3 | 3 ul |
| Ligation Mighty Mix(TAKARA) | 8 ul |
| Total | 16 ul |
Ligation reaction time was written below.
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
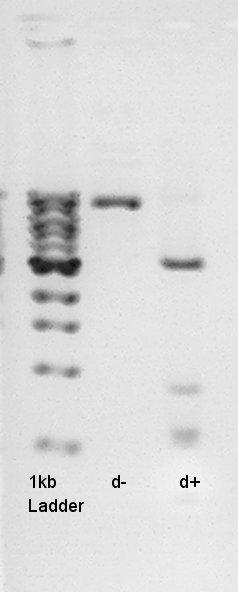
Electrophoresis result
Transformation
Transformation for pT7-RBS + Ag43-dT + pSB1C3 into BL21(DE3)(E.coli which have T7 polymerase coadin site in their genomic DNA).
- Added DNA soltions (Ligation products) 1 ul to BL21(DE3) compitent cell.
- Stood on ice in 30 min.
- Heatshock for 1min at 42c.
- Added 200 ul of LB to transformed BL21(DE3) solution.
- Pre-cultivate in 2 hrs
- Splead 200 ul of LB&BL21(DE3) solution supernant to LBC.
- 50ul of LB&BL21(DE3) solution were added to 200ul LB solution then spread 200 ul to LBC plate.
- Cultivated.
July 12th
Colony PCR
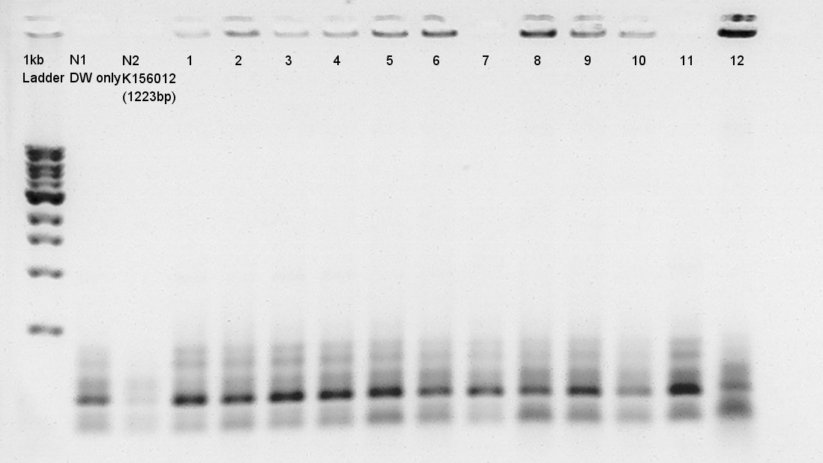
Colony PCR for ligation product.Each products were reacted in recipes written below.
- picked up each 16 colonies from LB plates by Autoclaved toothpicks.
- Dipped into 10 ul DW in 1.5 ml eppendorf tubes.
- from 10 ul DW, 4 ul was added in PCR reaction solution (below), 6 ul was mixed with 200 ul LB.
- Ran PCR machine in recipe below.
- Electrophoresis for confirmation of PCR results.
| DNA solution | 4 ul |
| KapaTaq ready mix | 5 ul |
| BioBrick prefix forward primer | 0.5 ul |
| BioBrick suffix reverse primer | 0.5 ul |
| Total | 10 ul |
PCR recipe
(pT7 + RBS)
| Number | Degree | Second |
| 1 | 94 | 120 |
| 2 | 94 | 30 |
| 3 | 68 | 90 |
| 4 | 4 | HOLD |
Cycle:2~3 x 40
Liquid culturing
Liquid culture for some colonies used in colony PCR.
- Prepared 200 ul LB solutions.
- To these LB solutions, added 6 ul of LB solutions (colony PCR solutions were pre-cultivated in about 3 hrs) and added 2 ml LB and antibiotic(Cp).
- Cultivated 18 hrs and 30 min.
July 13th
July 14th
Ligation
Ligation for digestion fragments written above. Ligation recipe Each DNA fragments (pT7-RBS + pSB1C3 and Ag43-dT + pSB1T3) were reacted in recipe written below.
| Insert | 5 ul |
| Vector | 1 ul |
| Ligation Mighty Mix(TAKARA) | 6 ul |
| Total | 12 ul |
| Degree | Minute |
| 16 | 30 |
| 65 | 10 |
| 4 | Hold |
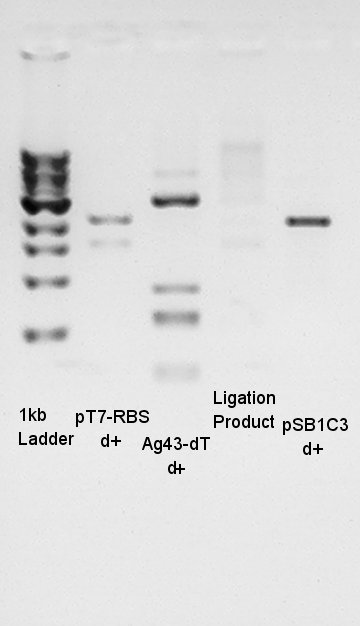
Ligation result with colony no. 1 (see colony pcr result in 9th)
Transformation
Transformation for ligation products written above.
- Added DNA soltions (Ligation products) 1 ul to DH5α compitent cell.
- Stood on ice in 30 min.
- Added 600 ul of LB to transformed DH5α solution.
- Pre-cultivate in 2 hrs
- Splead 300 ul of LB&DH5α solution to LBC and LBT , 100 ul added into 900 ul of LB.
- Splead 300 ul of LB&DH5α solution from 1000 ul LB (100 ul added into 900 ul) to LBC and LBT.
- Cultivated in 21 hrs.
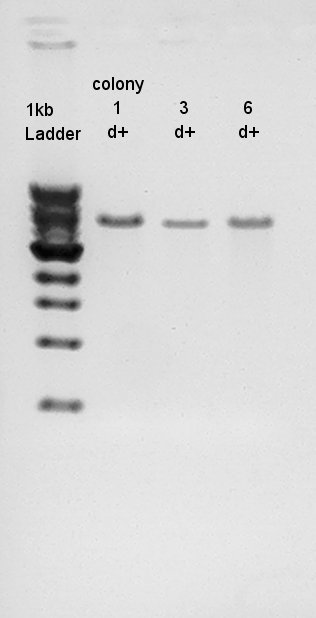
Liquid culture
Liquid culture for Ag43(BBa_K346007)
- Picked up one colony from single colony isolated plate.
- Dipped into LBC.
- cultivated.
July 15th
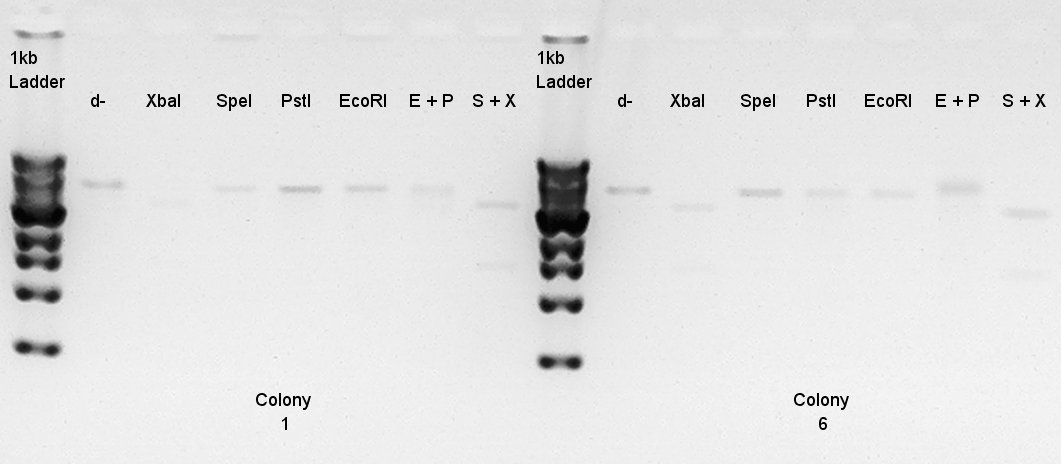
Gel extraction
We used Gel Extraction Kit (FastGene Gel/PCR extraction kit:NipponGenetics) to purify digestion products (see 14th).
Digestion
Digestion to confirm what kind of restriction enzyme cutting sites these DNA (colony 1 and 6) have. EcoRI
| DNA solution | 6 ul |
| EcoRI | 1 ul |
| 10xH buffer | 1 ul |
| DW | 2 ul |
| Total | 10 ul |
XbaI
| DNA solution | 6 ul |
| XbaI | 1 ul |
| 10xM buffer | 1 ul |
| BSA | 1 ul |
| DW | 1 ul |
| Total | 10 ul |
PstI
| DNA solution | 6 ul |
| PstI | 1 ul |
| 10xH buffer | 1 ul |
| DW | 2 ul |
| Total | 10 ul |
SpeI
| DNA solution | 6 ul |
| SpeI | 1 ul |
| 10xM buffer | 1 ul |
| DW | 2 ul |
| Total | 10 ul |
EcoRI + PstI
| DNA solution | 6 ul |
| EcoRI | 1 ul |
| PstI | 1 ul |
| 10xH buffer | 1 ul |
| DW | 11 ul |
| Total | 20 ul |
XbaI + SpeI
| DNA solution | 6 ul |
| XbaI | 1 ul |
| SpeI | 1 ul |
| 10xM buffer | 1 ul |
| DW | 11 ul |
| Total | 20 ul |
Ethanol precipitation
Ethanol precipitation for digestion products.
- Added 2 ul of NaoAc, 1.5 ul of glycogen and 50 ul of 100% ethanol.
- Centrifuged in 15000 rpm, 10 min at 4C.
- Remove supernatant and added 220 ul of 70% ethanol.
- Centrifuged in 15000 rpm, 5 min at 4C.
- Remove supernatant and air drying in room temperature then added 5 ul of DW.
Electrophoresis
Electrophoresis for digest-ethanol precipitation products.
- added 5 ul of EtBr.
- Migrated in 30 min.
Single colony isolation
Single colony isolation for Transformation products synthesized yesterday.
- one colony picked up from cultivated LBC and LBT plate.
- Spread on LBC and LBT.
- Cultivated.
Digestion
Digestion of Ag43(with S,P), dT(With X,P) and pT7-RBS(With E). Ag43
| DNA solution | 9 ul |
| SpeI | 1 ul |
| PstI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 7 ul |
| Total | 20 ul |
dT
| DNA solution | 1.1 ul |
| XbaI | 1 ul |
| PstI | 1 ul |
| 10xM buffer | 2 ul |
| DW | 14.9 ul |
| Total | 20 ul |
pT7-RBS
| DNA solution | 6 ul |
| EcoRI | 1 ul |
| 10xH buffer | 2 ul |
| DW | 11 ul |
| Total | 20 ul |
 "
"