Team:Evry/Tadpole injection1
From 2012.igem.org
(Difference between revisions)
| Line 9: | Line 9: | ||
The <a href="https://2012.igem.org/Team:Evry/InjectionTuto">injection tutorial</a> explains very simply with diagram how we did injection and how take care about your embryos and tadpole. The experiment carries on 5 days, from the not fertilized egg to a swimming tadpole at stage 48-50. The GFP (or other fluorescent protein) is expressed few hours after the fertilization to the end of the week (see below).<br/> | The <a href="https://2012.igem.org/Team:Evry/InjectionTuto">injection tutorial</a> explains very simply with diagram how we did injection and how take care about your embryos and tadpole. The experiment carries on 5 days, from the not fertilized egg to a swimming tadpole at stage 48-50. The GFP (or other fluorescent protein) is expressed few hours after the fertilization to the end of the week (see below).<br/> | ||
The iGEM-Evry tem say a great thanks to Dr. Nicolas Pollet, Dr. Aurore Thelie and Lena Vouillot (PhD student) who teach us how to inject embryos, take care of tadpoles and how to use their microscope. They are from <a href="http://www.issb.genopole.fr/">Institute of Systems & Synthetic Biology</a> of Evry in the <a href="http://indigene.issb.genopole.fr/">Metarmophosys</a> group.<br/><br/> | The iGEM-Evry tem say a great thanks to Dr. Nicolas Pollet, Dr. Aurore Thelie and Lena Vouillot (PhD student) who teach us how to inject embryos, take care of tadpoles and how to use their microscope. They are from <a href="http://www.issb.genopole.fr/">Institute of Systems & Synthetic Biology</a> of Evry in the <a href="http://indigene.issb.genopole.fr/">Metarmophosys</a> group.<br/><br/> | ||
| - | |||
<h2>Plasmids injected:</h2><br/><br/> | <h2>Plasmids injected:</h2><br/><br/> | ||
| Line 76: | Line 75: | ||
The GFP is still present in specific tissue, but also the GFP is decreasing. Plasmids could be ruined by cells, and/or the quantity of plasmids could decrease in each cells which involded the diminution of GFP in each cells, after that it is more difficult to see GFP. | The GFP is still present in specific tissue, but also the GFP is decreasing. Plasmids could be ruined by cells, and/or the quantity of plasmids could decrease in each cells which involded the diminution of GFP in each cells, after that it is more difficult to see GFP. | ||
| - | picture with the LSM 510 META Laser Scanning Microscope from Zeiss | + | picture with the LSM 510 META Laser Scanning Microscope from Zeiss.<br/> A great thank to Dr Daniel Stockholm and <a href="http://www.genethon.fr/en/">Genethon</a> for using this microscope.<br/> |
<img src="/wiki/images/c/ce/Picture_genethon_GFP-aid_7.09.jpg" alt="perdu" width="900px" /> | <img src="/wiki/images/c/ce/Picture_genethon_GFP-aid_7.09.jpg" alt="perdu" width="900px" /> | ||
<h2><li>pCS2+ citrine</li> </h2><br/><br/> | <h2><li>pCS2+ citrine</li> </h2><br/><br/> | ||
| - | Tadpole have 4 days (stage 45-46), because our lab does not have YFP filter the LSM 510 META Laser Scanning Microscope from Zeiss was used. A great thank to Dr Daniel Stockholm and <a href="http://www.genethon.fr/en/">Genethon</a> for using this microscope.<br/> | + | Tadpole have 4 days (stage 45-46), because our lab does not have YFP filter the LSM 510 META Laser Scanning Microscope from Zeiss was used.<br/> A great thank to Dr Daniel Stockholm and <a href="http://www.genethon.fr/en/">Genethon</a> for using this microscope.<br/> |
<img src="/wiki/images/b/b6/Picture_citrine_7.09.jpg" alt="perdu" width="900px" /> <br/> | <img src="/wiki/images/b/b6/Picture_citrine_7.09.jpg" alt="perdu" width="900px" /> <br/> | ||
The expression of mCitrine is localized in tail's muscles. | The expression of mCitrine is localized in tail's muscles. | ||
Revision as of 13:41, 23 September 2012
Characterization of injected plasmid into Xenopus tropicalis eggs
Embryos/tadpoles was not fed during all the week of experiment, they grew up with their own vitellus.
Pictures was taken with the Zeiss stereomicroscope: SteREO Lumar V12 with the camera AxioCamMR3.
The injection tutorial explains very simply with diagram how we did injection and how take care about your embryos and tadpole. The experiment carries on 5 days, from the not fertilized egg to a swimming tadpole at stage 48-50. The GFP (or other fluorescent protein) is expressed few hours after the fertilization to the end of the week (see below).
The iGEM-Evry tem say a great thanks to Dr. Nicolas Pollet, Dr. Aurore Thelie and Lena Vouillot (PhD student) who teach us how to inject embryos, take care of tadpoles and how to use their microscope. They are from Institute of Systems & Synthetic Biology of Evry in the Metarmophosys group.
Plasmids injected:
We injected 2.3 nl of plasmid at 100ng.µl-1 - embryos were stored at 21°C during all the experiment.
- pCS2+ GFP-aid: contains the constitutive promoter CMV and the aid sequenced of the aid system fusionned to GFP (Green Fluorescent Protein)(Nishimura et al., 2009), this Biobrick created by our team is BBa_K812010, and it was integrated into our Eucaryotic plasmid BBa_K812000 . Number of Plasmids injected: ~ 3.78E+7
- pCS2+ citrine: contains the constitutive promoter CMV and the fluorescent protein citrin (yellow), the plasmid BBa_K812130 (ready to use in Xenopus) is from our pCS2+ euckaryotic plasmid BBa_K812000 with the YFP (with the Kozak sequence) BBa_K812030. Number of Plasmids injected: ~ 4.38E+7
- pCS2+ mCFP: contains the constitutive promoter CMV and the fluorescent protein mCFP (Cyan Fluorescent Protein). the plasmid BBa_K812132 (ready to use in Xenopus) is from our pCS2+ euckaryotic plasmid BBa_K812000 with the YFP (with the Kozak sequence) BBa_K812032. Number of Plasmids injected: ~ 4.37E+7
- pCS2+ GFP-aid
- pCS2+ citrine
- pCS2+ mCFP
24h after injection
Eggs are near stage 20, neural fold is visible and the size of tadpol is near 1 mm

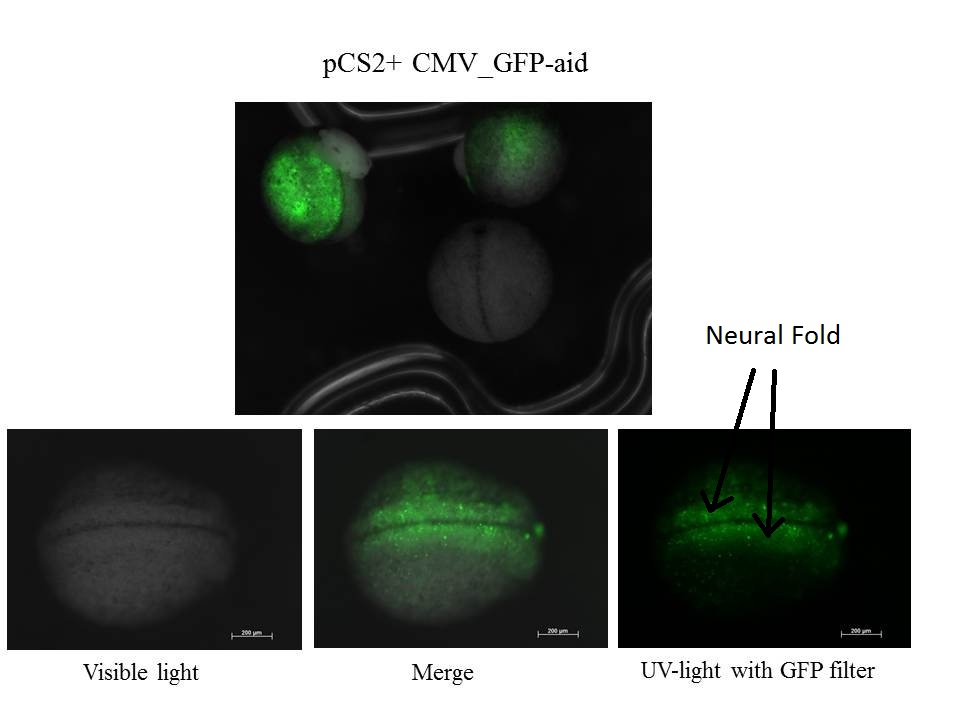

z-stack of the embryo
48h after injection
Embryos are at stage 34-38 and move by intermittence, the size of tadpol is near 2.5 mm
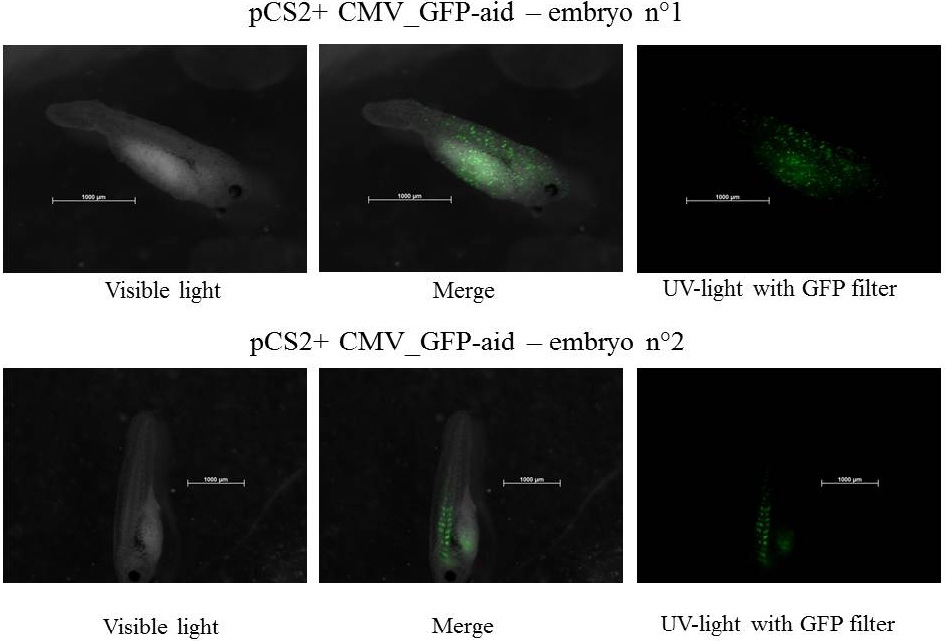
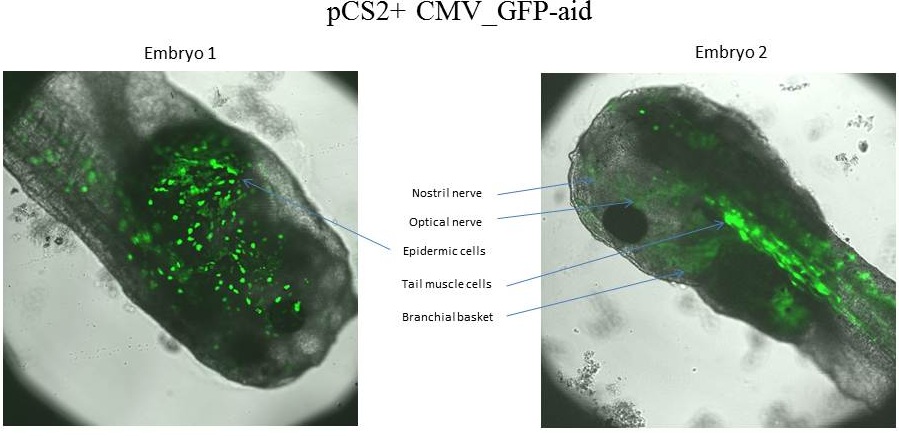
The expression of GFP-aid is localized in different tissue for each tadpole, in spite of the promoter is constitutive (CMV). We can think that the plasmid does not diffuse in the eggs because of the vitellus viscosity. This question was raised in one of the modelling part
3 days after injection
Embryos are at stage 41-42 and swim, the size of tadpole is near 4 mm.
From this tadpole stage an anaesthetic is required to take pictures of tadpoles, otherwise the light teases tadpoles, and it is impossible to take a picture

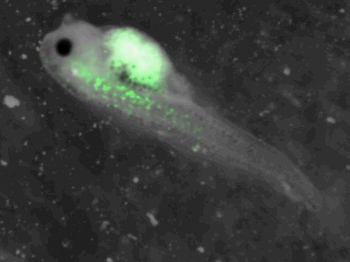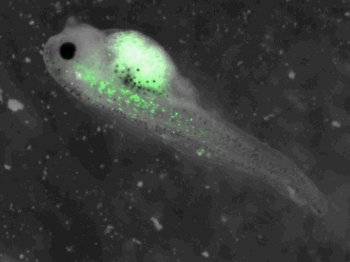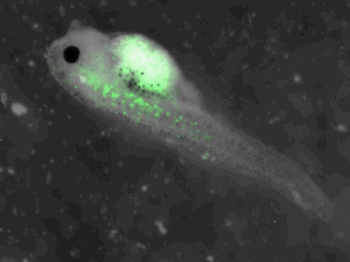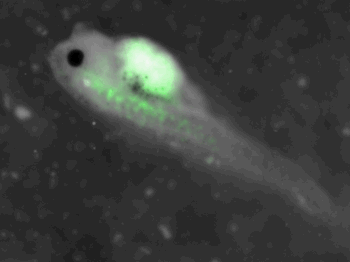
z-stack pictures: pCS2+ CMV_GFP-aid, GFP expression is not in same tissue between tadpoles: for example the tadpole on the left picture bones of the tail produced GFP, on the right picture the GFP expression is localized in the skin. The only part of the tadpole moving is the heart beatting (between the head and the stomac)
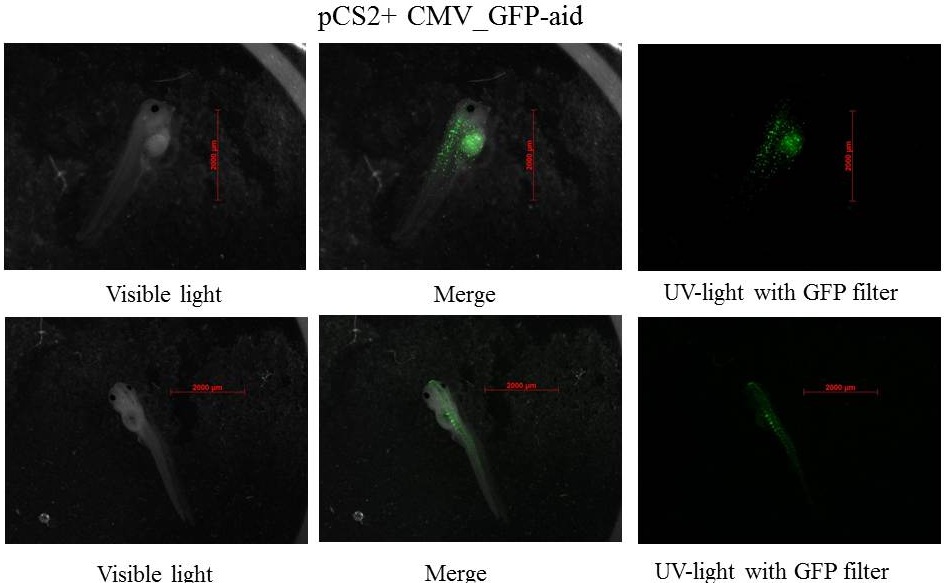
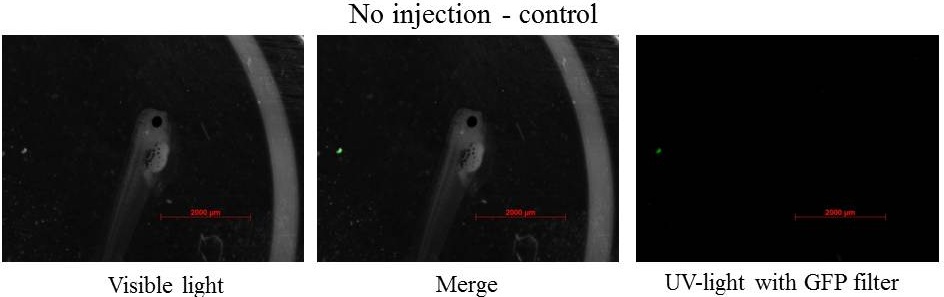
The expression of GFP-aid is localized in different tissue for each tadpole, like the day before. GFP is present in same tissue, it means that the plasmid stays in the same cells.
4 days after injection
Embryos are at stage 45-46 and swim, the size of tadpole is near 5 mm.
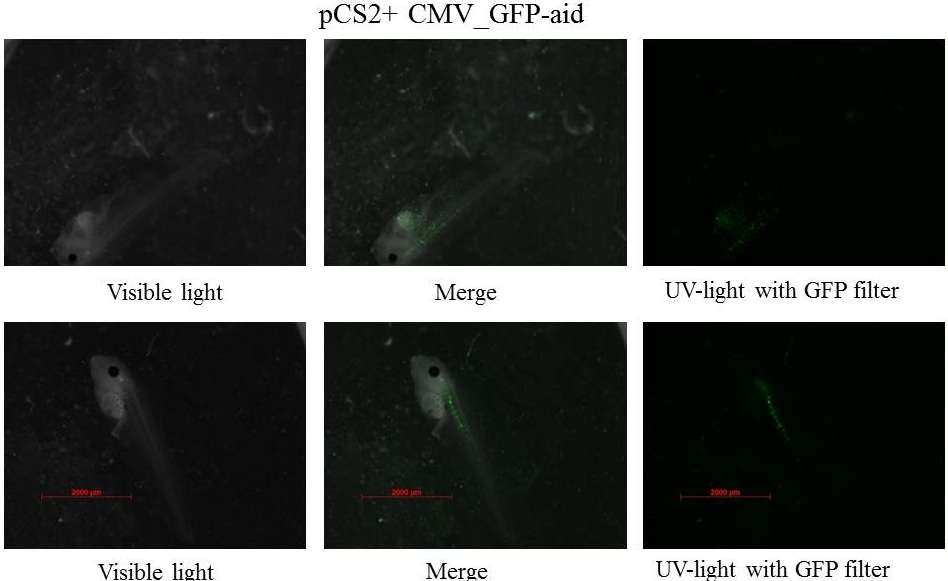
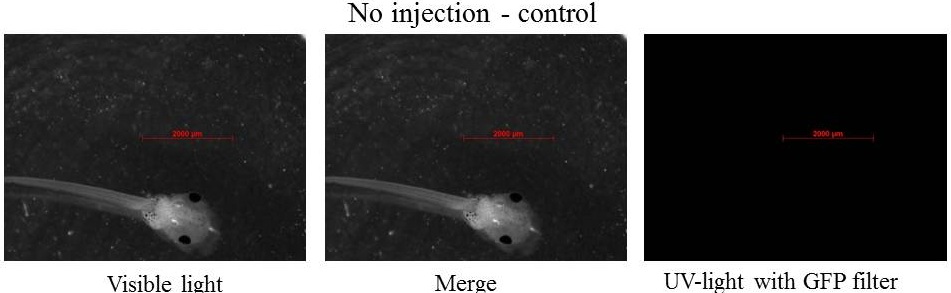
The GFP is still present in specific tissue, but also the GFP is decreasing. Plasmids could be ruined by cells, and/or the quantity of plasmids could decrease in each cells which involded the diminution of GFP in each cells, after that it is more difficult to see GFP. picture with the LSM 510 META Laser Scanning Microscope from Zeiss.
A great thank to Dr Daniel Stockholm and Genethon for using this microscope.
Tadpole have 4 days (stage 45-46), because our lab does not have YFP filter the LSM 510 META Laser Scanning Microscope from Zeiss was used.
A great thank to Dr Daniel Stockholm and Genethon for using this microscope.
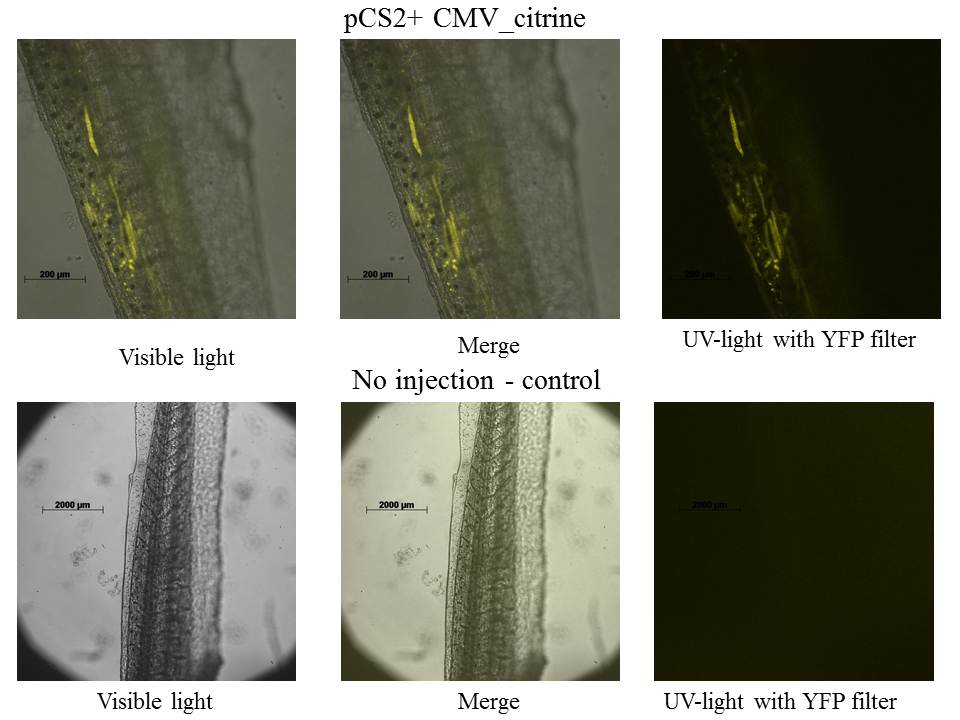
The expression of mCitrine is localized in tail's muscles.
References:
Nishimura, K. et al., 2009. An auxin-based degron system for the rapid depletion of proteins in nonplant cells. Nature Methods, 6(12), p.917-922.
 "
"