Team:Cambridge/Project/Results
From 2012.igem.org


Contents |
Results
Set out below are the developments the team has made over the summer, in tackling our aim and objectives.
RiboSense
Ratiometrica
Instrumentation (Biologger)
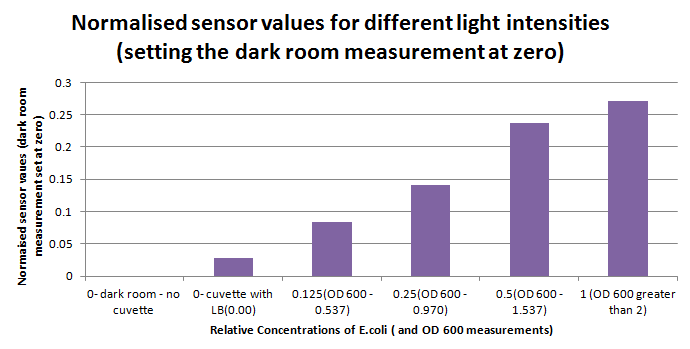
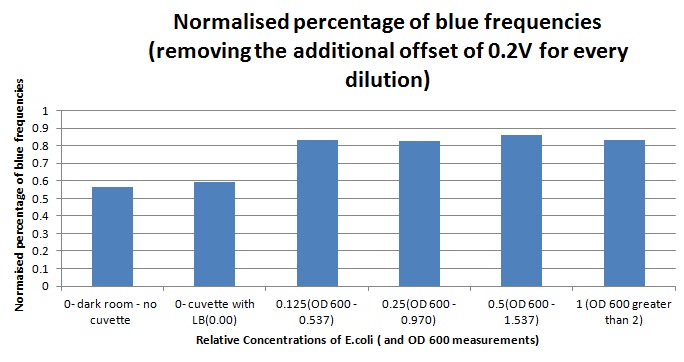
Having our instrumentation completed, the sensitivity of the sensor placed in the right position was tested using a dilution series of luciferase-producing bacteria. 20ml Cultures were grown overnight from single colonies. The cultures were induced with 40ul of 1.5M arabinose (for a final concentration of 3mM). Cultures were left for 2 1/2 hours for full induction. Subsequently, a culture was pelleted and resuspended in 4ml LB. Doubling dilutions, of volume 2ml, were made from this concentrate, down to 1/8th concentration. 1ml of each 2ml dilution was analysed in each cuvette, which was placed in the cuvette holder we made ourselves. The result was very good. An almost linear relationship was obtained when data were normalised with the sensor value taken in the dark room (the latter set at zero) without using the cuvette holder (1-(sensor value/sensor value in absolute dark)), presenting the sensitivity of the sensor to different intensities of light. This behaviour was expected due to the changing offset affecting the luciferase spectrum curve at different light intensities. The offset, using our data, was calculated to be about 0.2V for each dilution. A second graph is shown which takes into account this offset (and removes it), thus showing the presence of blue frequencies. The result was very good as the presence of blue frequencies throughout the dilution series is not only detected, but also found to be approximately constant.
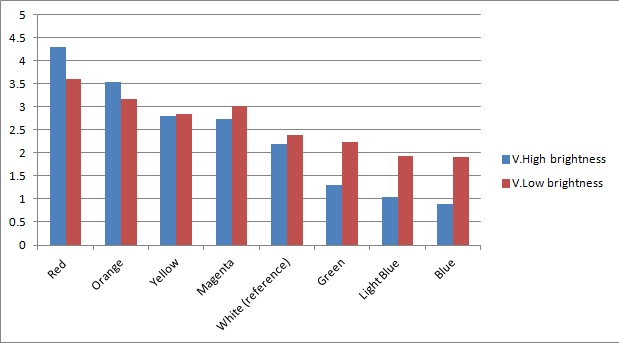
Once the sensor was built and tested for sensitivity, we tested that our circuitry correctly identified different frequencies (colours) of light. As can be seen below, measurements taken from orange and blue light yield values respectively above and below those from white light (our reference point). The data was taken using constant intensity of light for each case (V.High and V.Low brightness, as specified in the application). This was done with the aid of an Android phone and a specialised software application, called Color Flashlight, downloaded from the official Market.
As expected from the potential-divider design of our circuitry, orange and red frequencies caused the resistance of the LDR with the orange filter to decrease, leading to a higher voltage across the LDR with the blue filter. The opposite effect was observed with blue light. The reason that the white reference point is a bit lower than 2.5V (the expected value for a non-biased circuitry with a 5V source), is because we use resistors of total net resistance 1.67 kΩ before the blue LDR. This was done to bias the circuitry towards blue (i.e. decreasing the starting value, thus the sensor identifies always a bit more blue - this can be shown in previous graphs as well) and thus cause orange light to have a larger impact when present. This was used in an attempt to compensate for the fact that the peak at 560nm (Orange) in MOrange/luciferase fusion spectrum is lower than the one at 490 nm (Blue). Even though we did not manage to test the latter with transformed bacteria, the data collected in all the previous experiments makes us confident that the instrumentation is at least adequately functional.
As the major part of the instrumentation, the bio-electronic interface, had been made and tested, now we turned to testing the other parts of our deveoped kit. This included the mechanical chassis of the prototype, the electronic/mechatronic (sensory and motory) components, and of course the software. The overview of the finished hardware/software can be seen in our Intstrumentation-Biologger page. Below, the videos showing our instrumentation in action can be seen.
Further improvements on instrumentation design
Due to the constraints of time and budget, we could not realise all of our aims for the Biologger. What we have developed though is a robust prototype which clearly demonstrates the potential our kit has as a cost-effective and reliable sensory ability. We include the considerations we could not realise below, in order to guide anyone who wishes to turn our prototype into a polished end product.
- The cuvette holders we made could be replaced with alternative cuvettes with bio-containment mechanisms. These cuvettes could have a hollow wall, containing an antibiotic or antiseptic. By applying a small amount of force on the sides with the lid on, the second layer would break, thus releasing the antibiotic inside the cuvette. The cuvette would then pose a greatly diminished biocontainment risk and so this would facilitate disposal.
- Microfluidic technology could be used to deliver the testing sample to the cuvettes. This would both reduce spillage, and potentially reduce the sample volumes required.
- For increased biosecurity, and where cost is not an issue, the device can have a sensor dedicated to each cuvette. In that way, parallel readings could be taken in a non-rotary device, reducing the exposure of the cuvettes not in the detector to the environment.
- Hall-effect sensors or barcode readings could be used to more accurately identify which cuvette was in the detector. Furthermore, these barcodes could also be useful for identifying what analyte is being assayed in the sample by each biosensor.
- GPS data could also be acquired by the Android software, thus facilitating data analysis and detection of possible trends related to geographical position.
Sporage and Distribution
Sporulation of 'Bacillus subtilis' cells happens naturally in nature when the organism is stressed. In the lab sporulation can be artificially triggered by growing on a defined medium. The cells are first transformed with all the necessary parts for sensing and output and then sporulated. The dormant spores can be stored by simply dropping a solution of them onto sterile filter paper disks and air drying them. These disks are attached to the inside of the cuvette thereby immobilising the bacteria. A dessicant is also kept in the cuvette to contribute to the longevity. The cuvette can then be kept until the kit is required. When the kit is required, a provided germination medium is added to the cuvette. After a short incubation time the cuvette can be loaded into the arduino device, test solution added, and used for biosensing.
- work flow
- spova
- results
- containment
Costing
One of the most important motives for our project was a low-cost solution.
Instrumentation Cost:
- Arduino Experimentation Kit
$25.00
- Light - dependent resistors 70kΩ/200kΩ
$2.00
- Colored Gel Filters
$2.00
- Materials for mechanical chassis
$6.00
Total $35.00
Optional: Bluesmirf Gold Bluetooth Modem $50.00
{Template:Team:Cambridge/CAM_2012_TEMPLATE_FOOT}}
 "
"