Team:Bielefeld-Germany/Results/Datapage
From 2012.igem.org

Datapage
How our BioBricks work
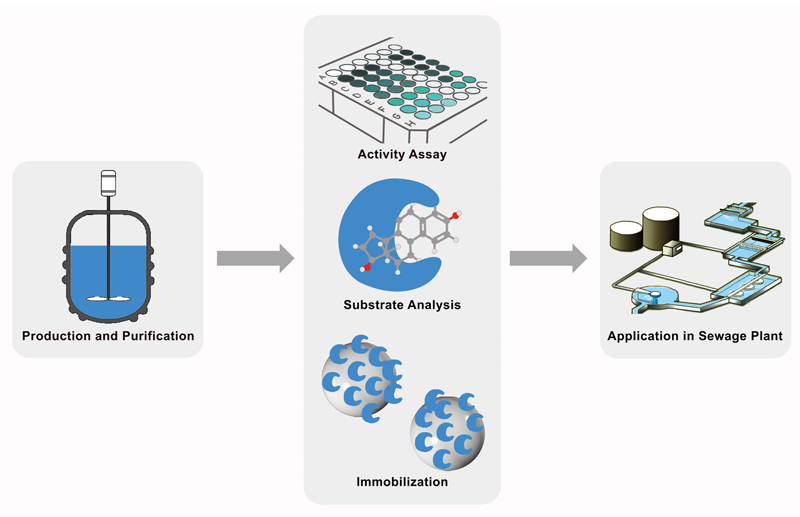
Figure 1: iGEM Team Bielefeld is developing a biological filter using immobilized laccases, enzymes able to radicalize and break down a broad range of aromatic substances. For the production of laccases from different bacteria, fungi and plants, two expression systems are used: Escherichia coli and the yeast Pichia pastoris. Immobilization is carried out either by using CPC-silica beads or by fusing the enzymes to cellulose binding domains. The concept could be extended to other toxic pollutants in drinking and wastewater, as well as to industrial applications in paper and textile industries or even for bioremediation of contaminated soil.
Data for our favorite new parts
- <partinfo>K863000</partinfo> - bpul (laccase from Bacillus pumilus) with T7 promoter, RBS and His-tag: This part is used to overexpress the laccase bpul for further purification followed by characterization of enzyme activity and substrate specificity.
- <partinfo>K863005</partinfo> - ecol (laccase from E. coli) with T7 promoter, RBS and His-tag: This enzyme is overexpressed after induction and can be purified by His-tag. Subsequently the laccase can be characterized regarding to enzyme activity and substrate specificity.
- <partinfo>K863204</partinfo> - shuttle vector pECPP11JS for site-directed recombination of genes of interest in yeast: With this part the production and secretion of a protein of interest like laccase is possible.
We have also characterized the following parts
- <partinfo>BBa_K863012</partinfo> - tthl laccase (from T. thermophilus) with constitutive promoter J23100, RBS and His-tag: This part is used for the constitutive expression of tthl for further purification followed by characterization of enzyme activity and substrate specificity.
- <partinfo>BBa_K863022</partinfo> - bhal laccase (from Bacillus halodurans) with constitutive promoter J23100, RBS and His-tag: This part is used for the constitutive expression of bhal for further purification followed by characterization of enzyme activity and substrate specificity.
| 55px | | | | | | | | | | |
 "
"





