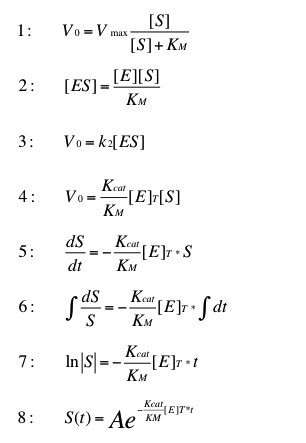Team:Bielefeld-Germany/Modell
From 2012.igem.org
(→The reaction) |
(→The reaction) |
||
| Line 22: | Line 22: | ||
This transformation can be found in "Stryer biochemie". This formula is suitable for very low substrate concentrations. In this case we can estimate the substrate [https://2012.igem.org/wiki/index.php?title=Team:Bielefeld-Germany/Project/Background/Concentrations concentrations] below 0.1 µg L<sup>-1</sup>. | This transformation can be found in "Stryer biochemie". This formula is suitable for very low substrate concentrations. In this case we can estimate the substrate [https://2012.igem.org/wiki/index.php?title=Team:Bielefeld-Germany/Project/Background/Concentrations concentrations] below 0.1 µg L<sup>-1</sup>. | ||
| - | Because we can not find any information about the K<sub>cat</sub> K<sub>M</sub><sup>-1</sup> quotient for the degradation of [[https://2012.igem.org/Team:Bielefeld-Germany/Project/Background#Estradiol: estradiol]] in the enzyme database [http://www.brenda-enzymes.info Brenda] we decided to use different K<sub>cat</sub> K<sub>M</sub><sup>-1</sup> quotients for the degradation of [ | + | Because we can not find any information about the K<sub>cat</sub> K<sub>M</sub><sup>-1</sup> quotient for the degradation of [[https://2012.igem.org/Team:Bielefeld-Germany/Project/Background#Estradiol: estradiol]] in the enzyme database [http://www.brenda-enzymes.info Brenda] we decided to use different K<sub>cat</sub> K<sub>M</sub><sup>-1</sup> quotients for the degradation of [http://www.sigmaaldrich.com/catalog/product/sigma/a1888?lang=de®ion=DE ABTS], a red ox mediator of the laccase. In the following picture we show a few possible reactions of our laccase. |
| - | + | [File:Bielefeld2012_model.jpg|center|800px|thumb|Our Model with different K<sub>cat</sub> K<sub>M</sub><sup>-1</sup> values. The span differs from 0.07 seconds to 1500 seconds.] | |
We use K<sub>cat</sub> K<sub>M</sub><sup>-1</sup> values fro 11 to 643500 and get degradation time points from 0,07s to 1500s. Furthermore we try to integrate different temperatures to our model. In a span from 10°C to 30°C and a K<sub>cat</sub> K<sub>M</sub><sup>-1</sup> value of 1, the degradation differs from 40s to 100s. | We use K<sub>cat</sub> K<sub>M</sub><sup>-1</sup> values fro 11 to 643500 and get degradation time points from 0,07s to 1500s. Furthermore we try to integrate different temperatures to our model. In a span from 10°C to 30°C and a K<sub>cat</sub> K<sub>M</sub><sup>-1</sup> value of 1, the degradation differs from 40s to 100s. | ||
Revision as of 23:59, 26 September 2012
Contents |
Model of a fixed-bed reactor
In our project we plan to construct a fixed-bed reactor, where immobilized laccases degrade synthetic estrogen and other harmful substances. As a small selection we plan to caracterize our different laccases for three estrogens, three analgesics, four PAH´s, one insecticide and three possible redox mediators. If we could model the degradation of one substrate by one Laccase, we could easily replace the specific Kcat KM-1 quotient for another Laccase and the amounts of the other substrates.
We ignore the possible cofactors ABTS, syringaldazine and viuloric acid, because on the one hand thy would cause increase the costs, and one the other hand they are harmful substances too. As shown by Team Substrate Analytic TVEL0 degrades ethinyl estradiol without reox mediator. So the reaction should follow the michaelis menten kinetics.
The reaction
The normal michaelis menten cinetic (1) can be adaptet for low substrate concentrations. To do this formula (2) replaces [ES] in formula (3). The result is formula (4). The velocity of this reaction descibes the cange of the substrate concentration over time (5). Formula (6) and (7) steps to solve the integral. (8) is the resulting formula to determine the time dependent substrate concentration by given starconcentration A.
This transformation can be found in "Stryer biochemie". This formula is suitable for very low substrate concentrations. In this case we can estimate the substrate concentrations below 0.1 µg L-1.
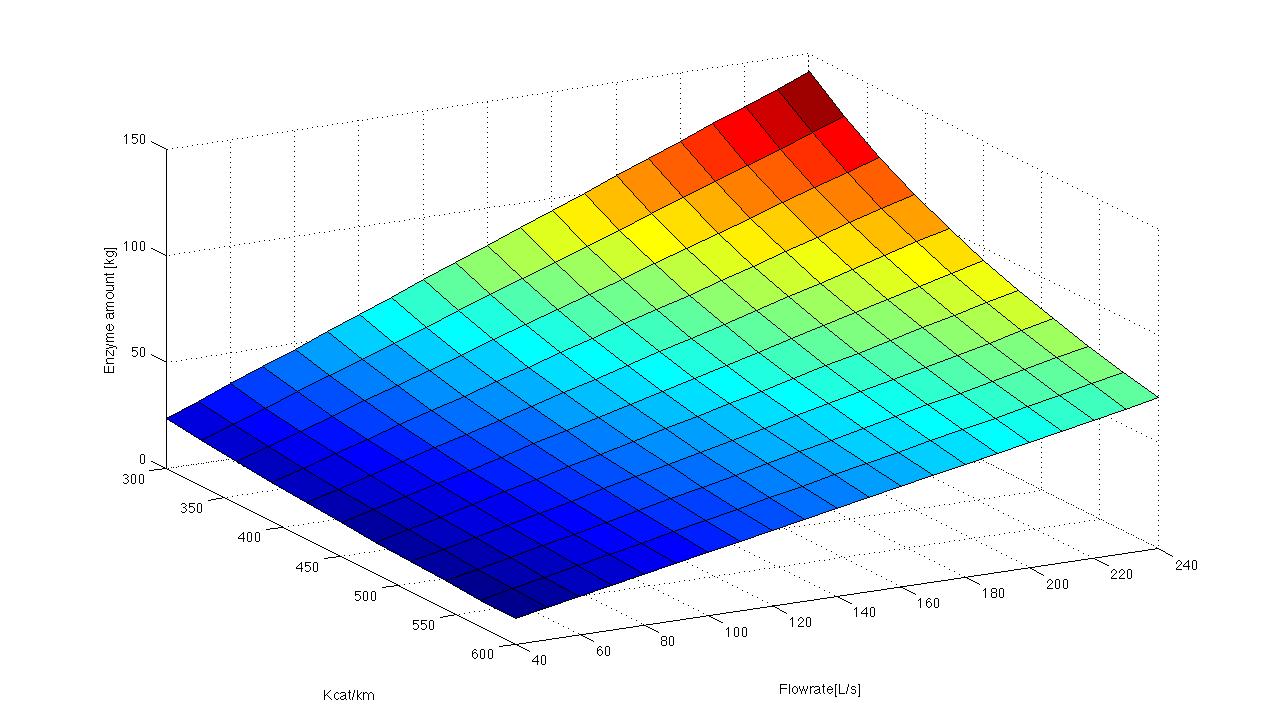
Because we can not find any information about the Kcat KM-1 quotient for the degradation of [estradiol] in the enzyme database Brenda we decided to use different Kcat KM-1 quotients for the degradation of ABTS, a red ox mediator of the laccase. In the following picture we show a few possible reactions of our laccase.
[File:Bielefeld2012_model.jpg|center|800px|thumb|Our Model with different Kcat KM-1 values. The span differs from 0.07 seconds to 1500 seconds.]
We use Kcat KM-1 values fro 11 to 643500 and get degradation time points from 0,07s to 1500s. Furthermore we try to integrate different temperatures to our model. In a span from 10°C to 30°C and a Kcat KM-1 value of 1, the degradation differs from 40s to 100s.
Extend the model with sewageplant data
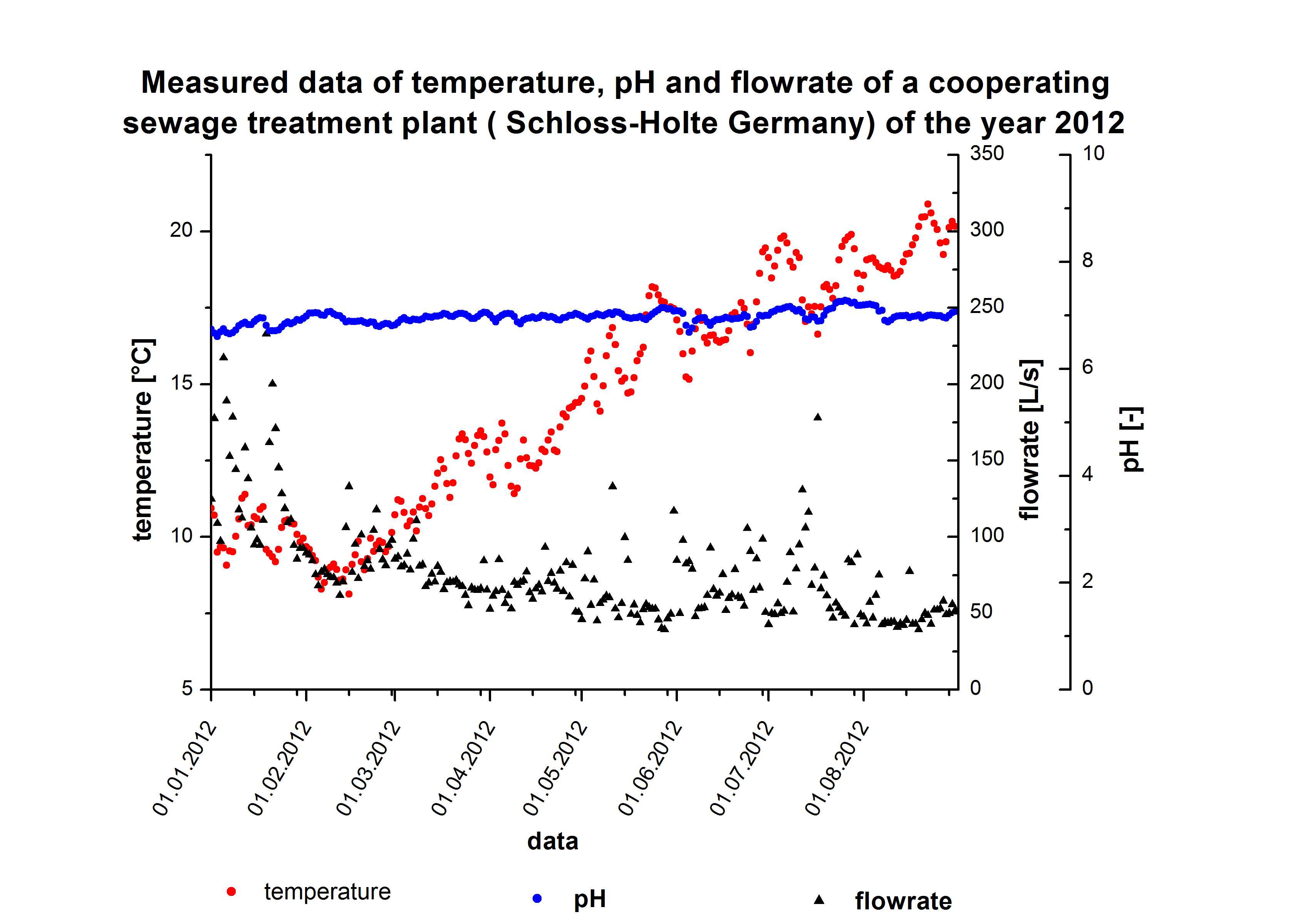
We got discharge water data from Schloss Holte about the water tempreture, pH value and flowrate. The temperature and pH value have a direct influence of enzymatic activity. Dependent on the enzymatic activity we want to calculate the time our fixed-bed reactor will need to degrdate 80 % of the substrates. This will be the dewll time. Combined with the actual flowrate we can determine the reactorsize. To estimate the feasibility we wart to know how much enzyme has to be produced for a wewage plant. A 3D model of the requirde enzme amount dependent of Kcat KM-1 values and flowrate.
Outlook
"Team Substrate Analytic" will determine Kcat KM-1. Additionaly we have the opportunity to work with a lab sewageplant. So we´re going to test our model in defined conditions.
| 55px | | | | | | | | | | |
 "
"