Team:MIT/ResultsFoundational
From 2012.igem.org
DEPRECATED. DO NOT USE OR EDIT. If a page uses this template, relink with MIT-results2.Overview:
Will include cartoon on strand displacement with our best microscopy and link to find out more about how strand displacement works
RNA Strand Displacement In Vitro
Previously:
In 2011, Lulu Qian and Erik Winfree, researchers at Caltech, published a paper entitled "Scaling Up Digital Circuit Computation with DNA Strand Displacement Cascades." This paper demonstrated how scalable logic circuits based on DNA strand displacement cascades in vitro are capable of processes as complicated as the square root function. See our motivation page for more details.
MIT iGEM 2012:
Before our team attempted to bring the mechanism of strand displacement into an in vivo context, we first decided to assay strand displacement in vitro using RNA. We used 2'-O-methylated RNA strands, which had never before been shown to undergo strand displacement in vitro. Before creating our own constructs, we adapted sequences from the Qian/Winfree paper to RNA.
MIT iGEM Foundational Experiment:
Figure A shows a foundational in vitro RNA strand displacement experiment that was performed on a plate reader. The negative control, in black, is a well that received only an annealed reporter complex. The bottom strand of this complex is the gate strand, T*-S6*, with the 3' end tagged with the ROX fluorophore. The top strand of the complex is the output strand, S6. This is complementary to the S6* domain of the gate strand. The 5' end is tagged with the Iowa Black RQ quencher, which absorbs the ROX fluorescence; thus, when the two strands of the reporter are annealed, no fluorescence should be observed. The positive control, in red, is the input strand, T-S6, annealed to the gate strand, T*-S6* tagged with ROX. This is what we would expect the product of a strand displacement reaction to look like. We can see that in the experimental well, when the input is present, it can bind to the exposed T* domain of the reporter and displace the output strand, yielding a fluorescent complex and a waste strand.
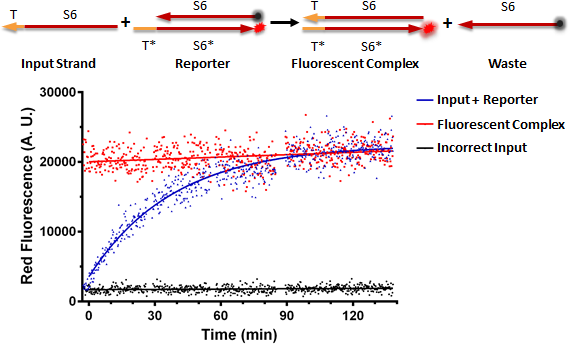
Figure A
Nucleic Acid Delivery
In order to implement RNA strand displacement cascades in vivo, we first demonstrated our ability to deliver nucleic acids to mammalian cells. We have successfully delivered plasmid DNA, single-stranded modified RNA and double-stranded modified RNA to mammalian cells through both lipofection and nucleofection. In this section, we will review three different nucleic acid delivery techniques to mammalian cells, (1) Delivery of plasmid DNA, (2) Delivery of 2'-O-Methyl RNA and (3) Delivery of plasmids with inducible protein control.
(1) Delivery of Plasmid DNA to Mammalian Cells
Through the Gateway method, we have assembled many promoter-gene constructs as detailed on our Biobrick Parts Page. After construction of the plasmid, we deliver the plasmid DNA to Mammalian Cells through the use of transient transfection, lipofection with Lipofectamine 2000 reagent. See our Materials and Methods Page for more detailed Protocol.
Images of eYFP/mKate transfection from Confocal
(2) Delivery of 2'-O-Me RNA to Mammalian Cells
For the purpose of our experiments, RNA oligos with chemical modifications that confer significant overall stability and increase in melting temperature is necessary to prevent spontaneous dissociation and rapid degradation by nucleases under in vivo conditions. One form of chemically modified RNA, 2’O-Methyl RNA, is a naturally occurring and nontoxic RNA variant found in mammalian ribosomal RNAs and transfer RNAs. These modified oligos are in most respects similar to RNA, but the 2' O-Methyl modification increases overall stability as the -OH functional group at the 2' position is replaced with a -OMe group, which can't perform cleavage of the RNA backbone. In addition to significant nuclease stability, the modification seem to confer increases in melting temperature, which minimizes the chance of the RNA strands dissociating upon introduction to a cellular environment. 1
Therefore, we need the ability to deliver 2’-O-Methyl RNA to mammalian cells.
The movie below shows HEK293 cells expressing constitutive eYFP with a 2'-O-Methyl RNA strand labeled with ROX (5-carboxy-x-rhodamine) on the 3' end. As time passes, the complex/vesicles are uptaken by the cell, releasing their payload resulting in whole cell fluorescence. Each frame is 5 minutes, movie encompasses 200 minutes in 9 seconds.
Delivery of ROX-labeled 2'-O-Methyl RNA into HEK cells.
 Time point images taken at t = 0, 2, 3, and 4 hours post-transfection. Images taken at 10X on Zeiss microscope.
Time point images taken at t = 0, 2, 3, and 4 hours post-transfection. Images taken at 10X on Zeiss microscope.
Once we demonstrated ability to deliver 2'-O-Me RNA to mammalian cells, we ran optimization experiments to optimize the ratio of 2'-O-Me RNA delivered to RNAiMAX (transfection reagent used).

Caption
(3) Inducible Control of Protein Expression

The microscopy images above show a brightfield view, the blue filter and the red filter. TagBFP serves as our transfection marker, indicating that cells have taken in foreign DNA. In the red channel, we show that as you increase the concentration of DOX, more cells fluoresce red.

The figure above was generated by transfecting the inducible expression system and varying the concentration of DOX across 16 different data points, and then analyzing using flow cytometry. We demonstrate that as you increase the concentration of DOX, the mean fluorescence increases. At high concentrations of DOX, we eventually see saturation of signal.
1 Behlke MA. (2008) Chemical modification of siRNAs for in vivo use. Oligonucleotides, 18(4):305–320.
In Vivo RNA Strand Displacement
We believe in RNA strand displacement as the ultimate processing medium for mammalian cellular circuits. In order to achieve strand displacement in vivo, we went through five different experimental designs after confirming the ability to deliver and produce many different types of oligos in vivo.
Strategy 1: Lipofectamine 2000 Transfection of RNA version of Reporter from Winfree/QIan 2011 Paper
Our first strategy to implement RNA strand displacement in vivo was to adapt the DNA sequences of inputs, gates and reporters from the Qian/Winfree, "Scaling Up Digital Circuit Computation with DNA Strand Displacement Cascades," 2011 Science Paper to 2’-O-Methyl RNA strands to transfect into mammalian cells. See our Motivation page for more details.
In the first foundational experiment, HEK293 (Human Embryonic Kidney cells) were used that constitutively expressed a yellow fluorescent protein (eYFP) in order to be easily visible in microscopy images. 200,000 HEK293 cells were seeded into four wells of a 24 well plate in supplemented DMEM without phenol red pH indicator. The negative control well did not receive any RNA. As a transfection reagent, each well received 1 uL of Lipofectamine 2000. The positive control well received 5 pmol of a gate strand tagged with a ROX fluorophore annealed to an input strand, to act as a product of a strand displacement reaction. The scrambled input well received 5 pmol annealed double stranded reporter with quenched ROX along with a 5 pmol of an input strand containing the correct toehold domain but the incorrect binding domain. Therefore, when both constructs are inside the cell, a strand displacement reaction should not occur, and the fluorophore remains quenched. In the final well, correct input, the cells received 5 pmol of double stranded reporter as well as 5 pmol of an input strand with the correct toehold domain and hybridization domain. Accordingly, we should expect that the toehold of the input strand binds to the complementary exposed toehold on the double stranded reporter, and will branch migrate and effectively kick off the output strand of the reporter that is tagged with a quencher. Therefore, the fluorophore will no longer be quenched, yielding red fluorescence.
 Refer to this diagram to identify labeled strands
Refer to this diagram to identify labeled strands

In the negative control well, 200,000 HEK293+eYFP cells are healthy and adherent. In the positive control well, we see localized red fluorescence in the form of vesicles as well as distributed, whole cell red fluorescence. In the scrambled input well, we see red vesicles as well as red whole cell fluorescence. In the correct input well, we see only whole cell red fluorescence.
Strategy 2: Switch Transfection reagent to RNAiMAX
From the first foundational experiment, we observed localized red fluorescence in vesicles as well as whole cell fluorescence. This indicates that our reporter complex is either melting, being degraded, being recognized by a specific enzyme etc as well as the reporter is coming apart inside of the lipofectamine vesicles. We researched better transfection reagents for double stranded RNA, and found that Lipofectamine RNAiMAX is designed specifically for the delivery of double stranded RNA, whereas Lipofectamine 200 is specifically designed for the delivery of plasmids.
Once we received the new transfection reagent, we set up experiments similar to the initial experiment but with an optimized protocol for RNAiMAX (See Materials and Methods).
 Caption for images from 6/14 experiment with old reporter and RNAiMAX where we do not see fluorescent vesicles anymore however we do still see fluorescent cells in the control and experimental wells
Caption for images from 6/14 experiment with old reporter and RNAiMAX where we do not see fluorescent vesicles anymore however we do still see fluorescent cells in the control and experimental wells
Strategy 3: Tag RNA strand with an Alexa Fluorophore to act as a transfection marker
Strategy 4: Create DNA plasmids driving transcription of RNA inputs, while transfecting RNA Reporter
Strategy 4: Nucleofect RNA reporter, RNA inputs
[Strategy 5]: Redesign RNA Reporter
 "
"